Physicochemical Properties
| Molecular Formula | C13H18O5S |
| Molecular Weight | 286.34 |
| Exact Mass | 286.087 |
| CAS # | 26225-79-6 |
| PubChem CID | 33360 |
| Appearance | White crystaline solid |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 409.1±45.0 °C at 760 mmHg |
| Melting Point | 100ºC |
| Flash Point | 201.2±28.7 °C |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.546 |
| LogP | 2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 19 |
| Complexity | 411 |
| Defined Atom Stereocenter Count | 0 |
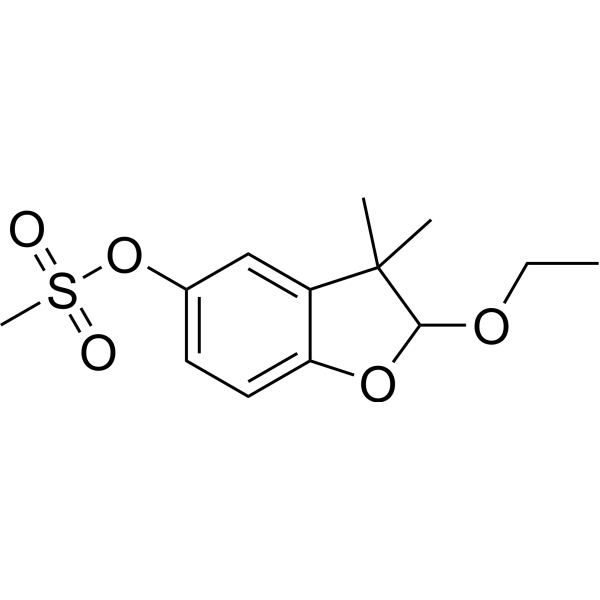
| SMILES | CC1(C)C(OCC)OC2=CC=C(OS(C)(=O)=O)C=C12 |
| InChi Key | IRCMYGHHKLLGHV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C13H18O5S/c1-5-16-12-13(2,3)10-8-9(18-19(4,14)15)6-7-11(10)17-12/h6-8,12H,5H2,1-4H3 |
| Chemical Name | (2-ethoxy-3,3-dimethyl-2H-1-benzofuran-5-yl) methanesulfonate |
| Synonyms | ETHOFUMESATE; 26225-79-6; Tramat; Nortron (new); (2-ethoxy-3,3-dimethyl-2H-1-benzofuran-5-yl) methanesulfonate; Nortran; 2-Ethoxy-3,3-dimethyl-2,3-dihydro-1-benzofuran-5-yl methanesulfonate; NC 8438; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Herbicidal agent |
| ln Vitro | The commercial herbicide formulation Betanal® Expert and its active ingredients (a.i.s) ethofumesate, phenmedipham and desmedipham were focused in this study. Following questions yielding from a previous study, an in-depth analysis of the reproductive toxicity of the pesticide was made. Long-term exposures of Daphnia magna and Daphnia longispina to Betanal® Expert, to each a.i. and to a customised mixture matching the a.i.s ratio within the commercial formulation were carried out, and deleterious effects in the offspring were recorded. This intended to clarify whether (1) the tested compounds induce reproductive injury; (2) there is interspecific variation in daphnids tolerance to the compounds; (3) there is an interaction between chemicals in combined treatments; and (4) the so-called inert ingredients added to the commercial formulation contribute to the toxicity of the herbicide. Generally, developmental impair was observed in both species (egg abortion and release of undeveloped embryos or dead offspring) at concentrations of any of the a.i.s below 1 mg L(-1). Ethofumesate was invariably the least toxic pesticide, and D. magna tended to be of slightly higher sensitivity to the exposures compared to D. longispina. Joint exposures indicated that the a.i.s can interact, inducing more than and less than additive effects for Betanal® Expert and the customised a.i. mixture, respectively. This indicates that inert ingredients co-formulating the commercial pesticide (which are absent from the customised a.i. mixture) actually contribute to its overall toxicity. This study constitutes an add-on to the discussion on the ecotoxicological framework required for authorisation of pesticide trade and usage. The results support the need to consider test species, long-term hazardous potential and toxicity of commercial formulations rather than solely that of active ingredients, as relevant variables in pesticide regulation[Environ Sci Pollut Res Int. 2016 Jul;23(13):13276-87]. |
| ln Vivo | Ethofumesate (ETO) is a chiral herbicide that is marketed as a racemic mixture in the European Union and the United States. The growing consumption of pesticides in the world, along with their presence in water and food, has increased human exposure to these chemicals. Another issue concerning these compounds is that each enantiomer of a chiral pesticide may interact with biomolecules differently. For this reason, this study aimed to investigate the in vitro metabolism of ethofumesate (the racemic mixture as well as the isolated enantiomers) by human liver microsomes (HLM) and to explore the in vitro-in vivo correlation. Before the kinetics was determined, the method was fully validated by evaluating its selectivity, linearity, precision, accuracy, carryover, and stability. All the evaluated parameters agreed with the European Medicines Agency guideline. The enzyme kinetic parameters and the in vitro-in vivo correlation demonstrated that there was no enantioselective difference for the metabolism and bioavailable fraction of each enantiomer. The enzyme kinetics was biphasic; the KM1 values were 15, 5.8, and 5.6 for rac-ETO, (+)-ETO, and (-)-ETO, respectively. The total in vitro intrinsic clearance was 0.10 mg mL min-1 mg-1 for rac-ETO and its enantiomers. The enantiomer (-)-ETO was only metabolized by CYP2C19, while (+)-ETO was metabolized by both CYP2C19 and CYP3A4. CYP2C19 polymorphism and/or inhibition may represent a risk for humans exposed to this pesticide[1]. |
| Enzyme Assay |
Ennzyme kinetics[1] Enantioselective metabolism was assessed by incubating rac-ETO, (+)-ETO, or (–)-ETO with HLMs. The initial reaction rate (V0) of rac-ETO-2-OH formation was monitored. To this end, an achiral GC-MS method was employed to quantify the metabolite rac-ETO-2-OH in the incubation medium. Initially, the V0 conditions were determined according to protein concentration (0.2 mg mL−1) and incubation time (10, 20, 30, or 40 min); three different rac-ETO concentrations (1.50, 100, or 200 μmol L−1) were employed. Linearity in the formation of the metabolite rac-ETO-2-OH was investigated for the four incubation times. The enzyme kinetic parameters after rac-ETO, (+)-ETO, or (–)-ETO metabolism by HLM was determined by using substrate concentrations ranging from 0.6 to 180 μmol L−1 (n = 5). The results were plotted as V0 versus the substrate concentration. The Eadie-Hofstee plot was employed to determine the enzyme kinetic model. Finally, the first component of the enzyme kinetics was fitted with the Michaelis Menten model, and the second component was fitted by linear regression to determine the slope of the enzyme kinetic curve in this phase [15]. The kinetic parameters (Michaelis Menten constant (KM) and maximum rate (VMAX)) were obtained by nonlinear regression analysis with the GraphPad Prism 6 software (San Diego, CA, USA), and the intrinsic clearance of the first component (CLINT1) was calculated by using Eq. 1 [15].(1) Where CLINT1 is the intrinsic clearance, KM1 is the Michaelis-Menten constant of the first component, and VMAX1 is the maximum rate of the first component. The intrinsic clearance of the second component (CLINT2) was obtained by the slope of the kinetic curve in the second phase of the enzyme kinetics. The second component was distinguished from the first component by using the Eadie-Hofstee graph. The inflection point indicated the boundary between both components. |
| ADME/Pharmacokinetics |
Metabolism / Metabolites /In animals the/ major metabolite is the lactone or free acid form of the respective 2-oxo compound. |
| Toxicity/Toxicokinetics |
Toxicity Data LC50 (rat) >3,970 mg/m3/4H Antidote and Emergency Treatment Basic treatment: Establish a patent airway (oropharyngeal or nasopharyngeal airway, if needed). Suction if necessary. Watch for signs of respiratory insufficiency and assist ventilations if needed. Administer oxygen by nonrebreather mask at 10 to 15 L/min. Monitor for pulmonary edema and treat if necessary ... . Monitor for shock and treat if necessary ... . Anticipate seizures and treat if necessary ... . For eye contamination, flush eyes immediately with water. Irrigate each eye continuously with 0.9% saline (NS) during transport ... . Do not use emetics. For ingestion, rinse mouth and administer 5 ml/kg up to 200 ml of water for dilution if the patient can swallow, has a strong gag reflex, and does not drool ... . Cover skin burns with dry sterile dressings after decontamination ... . / Advanced treatment: Consider orotracheal or nasotracheal intubation for airway control in the patient who is unconscious, has severe pulmonary edema, or is in severe respiratory distress. Positive-pressure ventilation techniques with a bag valve mask device may be beneficial. Consider drug therapy for pulmonary edema ... . Consider administering a beta agonist such as albuterol for severe bronchospasm ... . Monitor cardiac rhythm and treat arrhythmias as necessary ... . Start IV administration of D5W /SRP: "To keep open", minimal flow rate/. Use 0.9% saline (NS) or lactated Ringer's if signs of hypovolemia are present. For hypotension with signs of hypovolemia, administer fluid cautiously. Watch for signs of fluid overload ... . Treat seizures with diazepam or lorazepam ... . Use proparacaine hydrochloride to assist eye irrigation ... . Toxicity Data LC50 (rat) >3,970 mg/m3/4H Non-Human Toxicity Values LD50 Rat oral 1130 mg/kg LD50 Mouse oral >5000 mg/kg LD50 Rat percutaneous >2000 mg/kg LC50 Rat inhalation >3.97 mg/L air/4 hr |
| References | [1]. Perovani IS, et al. Enantioselective in vitro metabolism and in vitro-in vivo correlation of the herbicide ethofumesate in a human model. J Pharm Biomed Anal. 2020;187:113349. |
| Additional Infomation |
2-ethoxy-3,3-dimethyl-2,3-dihydro-1-benzofuran-5-yl methanesulfonate is a methanesulfonate ester that is methanesulfonic acid in which the hydrogen of the hydroxy group has been replaced by a 2-ethoxy-3,3-dimethyl-2,3-dihydro-1-benzofuran-5-yl group. It is an ether, a methanesulfonate ester and a member of 1-benzofurans. Ethofumesate is a pre- and post-emergence herbicide for grasses and broad-leaved weed control in various crops. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4924 mL | 17.4618 mL | 34.9235 mL | |
| 5 mM | 0.6985 mL | 3.4924 mL | 6.9847 mL | |
| 10 mM | 0.3492 mL | 1.7462 mL | 3.4924 mL |