Physicochemical Properties
| Molecular Formula | C55H86O24 |
| Molecular Weight | 1131.26 |
| Exact Mass | 1130.55 |
| CAS # | 123748-68-5 |
| PubChem CID | 6476030 |
| Appearance | White to off-white solid powder |
| Density | 1.46 |
| Boiling Point | 1140.6±65.0 °C at 760 mmHg |
| Flash Point | 311.8±27.8 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.628 |
| LogP | 1.88 |
| Hydrogen Bond Donor Count | 13 |
| Hydrogen Bond Acceptor Count | 24 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 79 |
| Complexity | 2300 |
| Defined Atom Stereocenter Count | 27 |
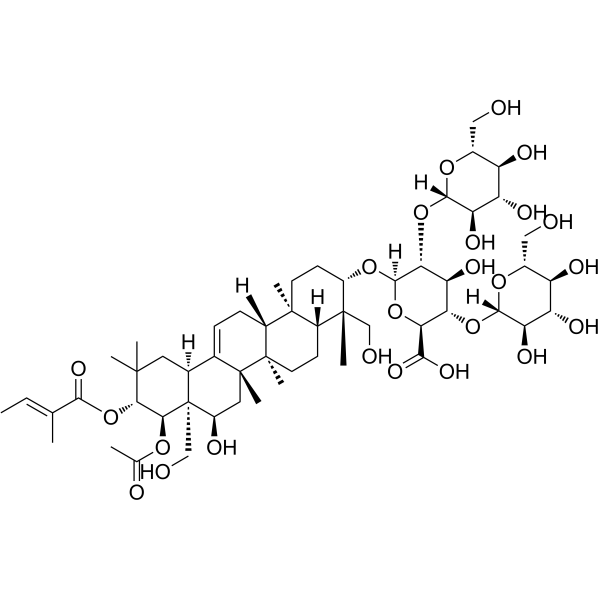
| SMILES | C/C=C(\C)/C(=O)O[C@H]1[C@@H]([C@@]2([C@@H](C[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H]([C@]5(C)CO)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)C(=O)O)O[C@H]7[C@@H]([C@H]([C@@H]([C@H](O7)CO)O)O)O)O)O[C@H]8[C@@H]([C@H]([C@@H]([C@H](O8)CO)O)O)O)C)C)[C@@H]2CC1(C)C)C)O)CO)OC(=O)C |
| InChi Key | AXNVHPCVMSNXNP-IVKVKCDBSA-N |
| InChi Code | InChI=1S/C55H86O24/c1-10-23(2)46(71)79-43-44(72-24(3)60)55(22-59)26(17-50(43,4)5)25-11-12-30-51(6)15-14-32(52(7,21-58)29(51)13-16-53(30,8)54(25,9)18-31(55)61)75-49-41(77-48-38(67)36(65)34(63)28(20-57)74-48)39(68)40(42(78-49)45(69)70)76-47-37(66)35(64)33(62)27(19-56)73-47/h10-11,26-44,47-49,56-59,61-68H,12-22H2,1-9H3,(H,69,70)/b23-10+/t26-,27+,28+,29+,30+,31+,32-,33+,34+,35-,36-,37+,38+,39-,40-,41+,42-,43-,44-,47-,48-,49+,51-,52+,53+,54+,55-/m0/s1 |
| Chemical Name | (2S,3S,4S,5R,6R)-6-[[(3S,4S,4aR,6aR,6bS,8R,8aR,9R,10R,12aS,14aR,14bR)-9-acetyloxy-8-hydroxy-4,8a-bis(hydroxymethyl)-4,6a,6b,11,11,14b-hexamethyl-10-[(E)-2-methylbut-2-enoyl]oxy-1,2,3,4a,5,6,7,8,9,10,12,12a,14,14a-tetradecahydropicen-3-yl]oxy]-4-hydroxy-3,5-bis[[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy]oxane-2-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
HIV-1 protease (IC50 = 35 μM)[1] |
| ln Vitro |
Escin Ia showed inhibitory activity against HIV-1 protease. At a concentration of 100 μM, a mixture of escins (which includes Escin Ia and other compounds) inhibited 86.1 ± 0.2% of the HIV-1 protease activity. Specifically, the isolated compound Escin Ia exhibited an IC50 value of 35 μM against HIV-1 protease.[1] |
| Enzyme Assay |
The inhibitory activity of compounds against HIV-1 protease was assayed. A volume of 25 μL of assay buffer (50 mM NaOAc, pH 4.9) containing 2.5 μg of the peptide substrate was mixed with 2.5 μL of the compound solution (dissolved in DMSO). Then, 6.25 μL of recombinant HIV-1 protease was added to initiate the reaction. The mixture was incubated at 37°C for 15 minutes, after which the reaction was terminated by adding 2.5 μL of 10% trifluoroacetic acid (TFA). The resulting hydrolysate and the remaining substrate were quantitatively analyzed using reversed-phase HPLC. The HPLC analysis employed a gradient of acetonitrile (20-40% over 26 minutes) in 0.1% TFA at a flow rate of 1.0 mL/min, with detection at 280 nm. The peak areas of the substrate and the hydrolysate were measured, and the inhibitory activity was calculated based on the reduction in the hydrolysate peak area compared to the control.[1] |
| References |
[1]. Anti-HIV-1 protease triterpenoid saponins from the seeds of Aesculus chinensis. J Nat Prod. 1999 Nov;62(11):1510-3. [2]. Escin Ia suppresses the metastasis of triple-negative breast cancer by inhibiting epithelial-mesenchymal transition via down-regulating LOXL2 expression. Oncotarget. 2016 Apr 26;7(17):23684-99. |
| Additional Infomation |
escin Ia has been reported in Aesculus chinensis and Aesculus hippocastanum with data available. See also: Horse Chestnut (part of); beta-ESCIN (annotation moved to). Escin Ia is a major bioactive triterpenoid saponin isolated from the seeds of Aesculus chinensis.[1] The structure of Escin Ia was elucidated as 21β-tigloyl-22α-acetylprotoaescigenin-3β-O-[β-D-glucopyranosyl (1→2)] [β-D-glucopyranosyl (1→4)]-β-D-glucopyranosiduronic acid.[1] Its absolute configuration was further supported by single-crystal X-ray analysis.[1] This study reports for the first time the anti-HIV-1 protease activity of escins, including Escin Ia.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~88.40 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (2.21 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8840 mL | 4.4199 mL | 8.8397 mL | |
| 5 mM | 0.1768 mL | 0.8840 mL | 1.7679 mL | |
| 10 mM | 0.0884 mL | 0.4420 mL | 0.8840 mL |