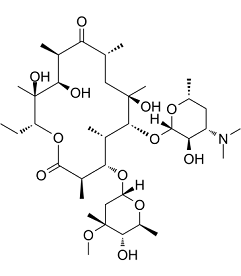
Erythromycin is a potent and broad-spectrum antibiotic belonging to a group of drugs called macrolide antibiotics, it is produced by actinomycete Streptomyces erythreus and is an inhibitor of protein translation and mammalian mRNA splicing. It acts by binding to bacterial 50S ribosomal subunits and inhibits RNA-dependent protein synthesis by blockage of transpeptidation and/or translocation reactions, without affecting synthesis of nucleic acid, thus inhibiting growth of gram negative and gram positiove bacteria. Erythromycin is used to treat certain infections caused by bacteria, such as infections of the respiratory tract, including bronchitis, pneumonia, Legionnaires' disease (a type of lung infection), and pertussis (whooping cough; a serious infection that can cause severe coughing); diphtheria (a serious infection in the throat); sexually transmitted diseases (STD), including syphilis; and ear, intestine, gynecological, urinary tract, and skin infections.
Physicochemical Properties
| Molecular Formula | C37H67NO13 |
| Molecular Weight | 733.9268 |
| Exact Mass | 733.461 |
| Elemental Analysis | C, 60.55; H, 9.20; N, 1.91; O, 28.34 |
| CAS # | 114-07-8 |
| Related CAS # | Erythromycin-d6;959119-25-6;Erythromycin-d3;959119-26-7;Erythromycin Ethylsuccinate;1264-62-6;Erythromycin stearate;643-22-1;Erythromycin lactobionate;3847-29-8;Erythromycin (aspartate);30010-41-4;Erythromycin thiocyanate;7704-67-8;Erythromycin A dihydrate;59319-72-1;Erythromycin-13C,d3;2378755-50-9 |
| PubChem CID | 12560 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 818.4±65.0 °C at 760 mmHg |
| Melting Point | 138-140ºC |
| Flash Point | 448.8±34.3 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.535 |
| Source | Streptomyces erythreHs |
| LogP | 2.83 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 51 |
| Complexity | 1180 |
| Defined Atom Stereocenter Count | 18 |
| SMILES | O([C@@]1([H])[C@@]([H])([C@]([H])(C([H])([H])[C@@]([H])(C([H])([H])[H])O1)N(C([H])([H])[H])C([H])([H])[H])O[H])[C@@]1([H])[C@@](C([H])([H])[H])(C([H])([H])[C@@]([H])(C([H])([H])[H])C([C@]([H])(C([H])([H])[H])[C@]([H])([C@@](C([H])([H])[H])([C@@]([H])(C([H])([H])C([H])([H])[H])OC([C@]([H])(C([H])([H])[H])[C@]([H])([C@]1([H])C([H])([H])[H])O[C@@]1([H])C([H])([H])[C@](C([H])([H])[H])([C@]([H])([C@]([H])(C([H])([H])[H])O1)O[H])OC([H])([H])[H])=O)O[H])O[H])=O)O[H] |
| InChi Key | ULGZDMOVFRHVEP-RWJQBGPGSA-N |
| InChi Code | InChI=1S/C37H67NO13/c1-14-25-37(10,45)30(41)20(4)27(39)18(2)16-35(8,44)32(51-34-28(40)24(38(11)12)15-19(3)47-34)21(5)29(22(6)33(43)49-25)50-26-17-36(9,46-13)31(42)23(7)48-26/h18-26,28-32,34,40-42,44-45H,14-17H2,1-13H3/t18-,19-,20+,21+,22-,23+,24+,25-,26+,28-,29+,30-,31+,32-,34+,35-,36-,37-/m1/s1 |
| Chemical Name | (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-6-(((2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-14-ethyl-7,12,13-trihydroxy-4-(((2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyltetrahydro-2H-pyran-2-yl)oxy)-3,5,7,9,11,13-hexamethyloxacyclotetradecane-2,10-dione |
| Synonyms | Emycin; HSDB 3074; HSDB-3074; HSDB3074; Eryc-125; Eryc-250; Erythromycin |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Macrolide antibiotic |
| ln Vitro |
P. falciparum cannot grow when erythromycin is present; its IC50 and IC90 values are 58.2 μM and 104.0 μM, respectively[1]. Erythromycin (10 μM, 100 μM; 24 h, 72 h) exhibits anti-inflammatory and antioxidant properties. It also suppresses the accumulation of 4-HNE (p<0.01) and 8-OHdG (p<0.01) and significantly lowers the expression of TNF-α (p<0.01) and Iba-1 (p<0.01)[4]. |
| ln Vivo | Erythromycin (gastric intubation; 0.1–50 mg/kg; 30-120 days) slows the growth of tumors and increases the amount of time that mice survive after receiving a dose of 5 mg/kg.When given at a dose of 50 mg/kg, erythromycin (gastric intubation; 5 mg/kg) shortens the mean survival time in tumor-bearing mice by 4-5 days. However, it protects mice alive even 120 days after inoculation.[3]. A single injection of erythromycin (i.h.; 50 mg/kg) protects against cerebral ischemia reperfusion injury in a rat model[4]. |
| Enzyme Assay | Erythromycin inhibited growth of P. falciparum with IC50 and IC90 values of 58.2+/-7.7 microM and 104.0+/-10.8 microM, respectively. The activity of antimalarial drugs in combination with azithromycin or erythromycin against P. falciparum K1 were compared. Combinations of chloroquine with azithromycin or erythromycin showed synergistic effects against parasite growth in vitro. Combinations of quinine-azithromycin and quinine-erythromycin showed potentiation. Additive effects were observed in mefloquine-azithromycin and mefloquine-erythromycin combinations. Similar results were also produced by pyronaridine in combination with azithromycin or erythromycin. However, artesunate-azithromycin and artesunate-erythromycin combinations had antagonistic effects. The in vitro data suggest that azithromycin and erythromycin will have clinical utility in combination with chloroquine and quinine. The worldwide spread of chloroquine-resistant P. falciparum might inhibit the ability to treat malaria patients with chloroquine-azithromycin and chloroquine-erythromycin in areas of drug-resistant. The best drug combinations against multidrug-resistant P. falciparum are quinine-azithromycin and quinine-erythromycin [4]. |
| Cell Assay |
Cell Line: Primary cortical neuron of embryos (derived from the cerebral cortices of Sprague-Dawley rats 17 days old) Concentration: 10, 100 μM Incubation Time: 24, 72 hours Result: increased the cultivated neuronal cells' viability in vitro following three hours of oxygen-glucose deprivation (OGD). |
| Animal Protocol | Animal Model: Six-week-old female ddY mice with EAC cellsor six-week-old CDF mice with P388 cells[3] Dosage: 0.1 mg/kg; 0.5 mg/kg; 10 mg/kg; 30 mg/kg; 50 mg/kg Administration: Gastric intubation; 30-120 days Result: reduced tumor growth and extended the mice's mean survival time (5 mg/kg); in contrast, the 50 mg/kg dose caused the MST in tumor-bearing mice to be shorter. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Orally administered erythromycin is readily absorbed. Food intake does not appear to exert effects on serum concentrations of erythromycin. Some interindividual variation exists in terms of erythromycin absorption, which may impact absorption to varying degrees. The Cmax of erythromycin is 1.8 mcg/L and the Tmax is 1.2 hours. The serum AUC of erythromycin after the administration of a 500mg oral dose was 7.3±3.9 mg.h/l in one pharmacokinetic study. Erythromycin is well known for a bioavailability that is variable (18-45%) after oral administration and its susceptibility to broken down under acidic conditions. In patients with normal liver function, erythromycin concentrates in the liver and is then excreted in the bile.Under 5% of the orally administered dose of erythromycin is found excreted in the urine. A high percentage of absorbed erythromycin is not accounted for, but is likely metabolized. Erythromycin is found in most body fluids and accumulates in leucocytes and inflammatory liquid. Spinal fluid concentrations of erythromycin are low, however, the diffusion of erythromycin through the blood-brain barrier increases in meningitis, likely due to the presence of inflamed tissues which are easily penetrated. Erythromycin crosses the placenta. The clearance of erythromycin in healthy subjects was 0.53 ± 0.13 l/h/kg after a 125mg intravenous dose. In a clinical study of healthy patients and patients with liver cirrhosis, clearance of erythromycin was significantly reduced in those with severe liver cirrhosis. The clearance in cirrhotic patients was 42.2 ± 10.1 l h–1 versus 113.2 ± 44.2 l h-1 in healthy patients. Absorption of orally administered erythromycins occurs mainly in the duodenum. The bioavailability of the drugs is variable and depends on several factors including the particular erythromycin derivative, the formulation of the dosage form administered, acid stability of the derivative, presence of food in the GI tract, and gastric emptying time. Erythromycin is rather slowly absorbed after oral administration. peak serum concentrations ranged from 0.1 to 4.8 ug/mL according to the form and the coating of erythromycin administered. The oral absorption is less that 50% and erythromycin is degraded by gastric acid. It is absorbed in the small intestine (mainly in duodenum for humans) as erythromycin base. Erythromycin diffuses readily into intracellular fluids, achieving antibacterial activity in essentially all sites except the brain and CSF. Erythromycin penetrates into prostatic fluid, achieving concentrations approximately 40% of those in plasma. Concentrations in middle ear exudate reach only 50% of serum concentrations and thus may be inadequate for the treatment of otitis media caused by H. influenzae. Protein binding is approximately 70% to 80% for erythromycin base and even higher, 96%, for the estolate. Erythromycin traverses the placenta, and drug concentrations in fetal plasma are about 5% to 20% of those in the maternal circulation. Concentrations in breast milk are 50% of those in serum. In an in vitro model using human skin, erythromycin was absorbed into the stratum corneum following topical application of 10-20 mg of the drug in a vehicle containing dimethylacetamide and 95% alcohol. The drug does not appear to be absorbed systemically following twice daily application of a 2% solution of the drug in a vehicle containing 77% alcohol and polyethylene glycol and acetone. It is not known if erythromycin is absorbed from intact or denuded skin, wounds, or mucous membranes following topical application of an ointment containing the drug. For more Absorption, Distribution and Excretion (Complete) data for Erythromycin (13 total), please visit the HSDB record page. PEAK CONCN IN PLASMA...0.3-0.5 UG/ML 4 HR AFTER ORAL ADMIN OF 250 MG OF BASE & ARE 0.3-1.9 UG/ML AFTER...500-MG TABLET. VARIOUS ESTERS OF ERYTHROMYCIN HAVE BEEN PREPARED TO...IMPROVE STABILITY & FACILITATE ABSORPTION. ...CONCN OF ERYTHROMYCIN IN PLASMA ARE LITTLE DIFFERENT IF STEARATE IS GIVEN ORALLY. ...DIFFUSES READILY INTO INTRACELLULAR FLUIDS, & ANTIBACTERIAL ACTIVITY... ACHIEVED AT...ALL SITES EXCEPT BRAIN & CSF. ...ONE OF FEW ANTIBIOTICS THAT PENETRATES INTO PROSTATIC FLUID, CONCN ARE APPROX 40% OF...PLASMA. EXTENT OF BINDING...TO PLASMA PROTEINS VARIES...PROBABLY EXCEEDS 70% IN ALL.../FORMS OF DRUG/. /ERYTHROMYCIN/ ERYTHROMYCIN BASE IS ADEQUATELY ABSORBED FROM UPPER PART OF SMALL INTESTINE; IT IS INACTIVATED BY GASTRIC JUICE... FOOD IN STOMACH DELAYS ITS ULTIMATE ABSORPTION. /ERYTHROMYCIN/ ERYTHROMYCIN TRAVERSES PLACENTAL BARRIER; & CONCN OF DRUG IN FETAL PLASMA ARE ABOUT 5-20% OF THOSE IN MATERNAL CIRCULATION. /ERYTHROMYCIN/ For more Absorption, Distribution and Excretion (Complete) data for ERYTHROMYCIN STEARATE (11 total), please visit the HSDB record page. Metabolism / Metabolites Hepatic first-pass metabolism contributes significantly to erythromycin metabolism after an oral dose. Erythromycin is partially metabolized by CYP3A4 enzyme to N-desmethylerythromycin. Erythromycin is also hydrolyzed to _anhydro_ forms (anhydroerythromycin [AHE] and other metabolites), and this process is promoted by acidic conditions. AHE is inactive against microbes but inhibits hepatic drug oxidation and is therefore considered to be an important contributor to erythromycin drug-drug interactions. Twenty hours after an oral administration of 10 mg erythromycin to rats, about 37-43% of the administered radioactivity was recovered in the intestinal tract plus feces, 27.2 to 36.1% in the urine, 21-29% in the expired air. It was rapidly metabolized in the liver, mainly through demethylation process, and excreted in the bile as des-N-methyl-erythromycin, the major metabolite present only in the bile and in the intestinal contents of rats. The isotropic methyl group was eliminated in the expired air as CO2. IT IS HYDROLYZED IN SMALL INTESTINE & IN TISSUES TO YIELD ERYTHROMYCIN. Hepatic. Extensively metabolized - after oral administration, less than 5% of the administered dose can be recovered in the active form in the urine. Erythromycin is partially metabolized by CYP3A4 resulting in numerous drug interactions. Half Life: 0.8 - 3 hours Biological Half-Life The elimination half-life of oral erythromycin was 3.5 hours according to one study and ranged between 2.4-3.1 hours in another study. Repetitive dosing of erythromycin leads to increased elimination half-life. ... The serum elimination half-life of erythromycin is approximately 1.6 hours. The serum half-life in normal subjects is 2 hours and in anuric subjects, 4-6 hours. |
| Toxicity/Toxicokinetics |
Toxicity Summary Erythromycin acts by penetrating the bacterial cell membrane and reversibly binding to the 50 S subunit of bacterial ribosomes or near the “P” or donor site so that binding of tRNA (transfer RNA) to the donor site is blocked. Translocation of peptides from the “A” or acceptor site to the “P” or donor site is prevented, and subsequent protein synthesis is inhibited. Erythromycin is effective only against actively dividing organisms. The exact mechanism by which erythmromycin reduces lesions of acne vulgaris is not fully known: however, the effect appears to be due in part to the antibacterial activity of the drug. Interactions Erythromycin is metabolized by CYP3A and concomitant use with drugs that inhibit the CYP3A isoenzyme may result in increased erythromycin plasma concentrations. There is some evidence that concomitant use of oral erythromycin with drugs that inhibit CYP3A (i.e., fluconazole, ketoconazole, itraconazole, diltiazem, verapamil) is associated with an increased incidence of sudden death from cardiac causes, presumably because of increased plasma erythromycin concentrations resulting in an increased risk of QT prolongation (a dose-associated effect of erythromycin) and serious ventricular arrhythmias. Therefore, it has been suggested that concomitant use of erythromycin and drugs that are potent inhibitors of CYP3A should be avoided. Erythromycin may interact with astemizole and terfenadine (both drugs no longer commercially available in the US), resulting in potentially serious adverse cardiovascular effects. Some evidence indicates that erythromycin may alter the metabolism of astemizole and terfenadine, probably via inhibition of the cytochrome P-450 microsomal enzyme system. While erythromycin has been shown to decrease markedly the clearance of the active carboxylic acid metabolite of terfenadine, the effect of the macrolide on unchanged terfenadine concentrations has not been fully elucidated, but appears to show interindividual variation. In studies in extensive metabolizers of dextromethorphan or debrisoquin, erythromycin markedly impaired clearance of the active metabolite of terfenadine in all such individuals but produced measurable effects on unchanged terfenadine in only one-third of these individuals. In addition, erythromycin is known to inhibit the enzyme system responsible for astemizole's metabolism. Prolongation of the QT interval and ventricular tachycardia, including torsades de pointes, have been reported in some patients receiving astemizole or terfenadine concomitantly with erythromycin or the structurally related macrolide troleandomycin (no longer commercially available in the US). Rarely, cardiac arrest and death have been reported in patients receiving erythromycin and terfenadine concomitantly. Therefore, when terfenadine and astemizole were commercially available in the US, these antihistamines were contraindicated in patients receiving erythromycin, clarithromycin, or troleandomycin. In addition, concomitant administration of astemizole or terfenadine and azithromycin also was not recommended, although limited data suggested that azithromycin did not alter the metabolism of terfenadine. Although in vitro studies have shown varying degrees of additive or synergistic effects against some organisms when erythromycin was used in conjunction with penicillins, streptomycin, sulfonamides, rifampin, or chloramphenicol, the clinical importance of these reports has not been established. Antagonism of bactericidal activity has been observed between erythromycin and clindamycin in vitro. In addition, antagonism has been reported when a bacteriostatic drug was administered with a bactericidal drug, but antagonism has not been convincingly documented clinically. Concomitant use of erythromycin in patients receiving high dosage of theophylline has resulted in decreased clearance of theophylline, elevated serum theophylline concentrations, and a prolonged serum half-life of the bronchodilator. An interaction may be most likely to occur in patients receiving an erythromycin dosage greater than 1.5 g daily for more than 5 days. Patients receiving theophylline should be closely monitored for signs of theophylline toxicity when erythromycin is administered concomitantly; serum theophylline concentrations should be monitored and dosage of the bronchodilator reduced if indicated. Although further study is needed and the clinical importance has not been determined to date, there is some evidence that concomitant administration of erythromycin and theophylline can also result in decreased serum erythromycin concentrations and subtherapeutic concentrations of erythromycin may occur. For more Interactions (Complete) data for Erythromycin (22 total), please visit the HSDB record page. A 77-YR-OLD WOMAN IS REPORTEDLY MAINTAINED ON 7.5 MG OF WARFARIN DAILY IN WHOM THE ADMIN OF ORAL ERYTHROMYCIN STEARATE, 500 MG 4 TIMES A DAY, RESULTED IN A PROTHROMBIN TIME OF 64 SECONDS (CONTROL, 11 SECONDS). Non-Human Toxicity Values LD50 Rat oral 9272 mg/kg LD50 Mouse ip 463 mg/kg LD50 Mouse sc 1800 mg/kg LD50 Mouse im 426 mg/kg For more Non-Human Toxicity Values (Complete) data for Erythromycin (6 total), please visit the HSDB record page. |
| References |
[1]. Erythromycin. Med Clin North Am. 1982 Jan;66(1):79-89. [2]. Activity of azithromycin or erythromycin in combination with antimalarial drugs against multidrug-resistant Plasmodium falciparum in vitro. Acta Trop. 2006 Dec;100(3):185-91. Epub 2006 Nov 28. [3]. Antitumor effect of erythromycin in mice. Chemotherapy. 1995 Jan-Feb. 41(1):59-69. [4]. Neuroprotective effects of erythromycin on cerebral ischemia reperfusion-injury and cell viability after oxygen-glucose deprivation in cultured neuronal cells. Brain Res. 2014 Nov 7. 1588:159-67. |
| Additional Infomation |
Therapeutic Uses Antibiotics, Macrolide; Gastrointestinal Agents; Protein Synthesis Inhibitors MEDICATION (VET): In veterinary medicine, /erythromycin/ is used the treatment of clinical and subclinical mastitis in lactating cows, for the treatment of infectious diseases due to erythromycin-sensitive bacteria (cattle, sheep, swine, poultry) and for the treatment of chronic respiratory diseases due to mycoplasma in poultry. Erythromycin is used as an alternative agent in the treatment of anthrax. Parenteral penicillins generally have been considered the drugs of choice for the treatment of naturally occurring or endemic anthrax caused by susceptible strains of Bacillus anthracis, including clinically apparent GI, inhalational, or meningeal anthrax and anthrax septicemia, although IV ciprofloxacin or IV doxycycline also are recommended. Erythromycin is suggested as an alternative to penicillin G for the treatment of naturally occurring or endemic anthrax in patients hypersensitive to penicillins. ./NOT included in US product label/ Erythromycin is used topically in the treatment of acne vulgaris. Therapy of acne vulgaris must be individualized and frequently modified depending on the types of acne lesions which predominate and the response to therapy. Topical anti-infectives, including erythromycin, are generally effective in the treatment of mild to moderate inflammatory acne. However, use of topical anti-infectives as monotherapy may lead to bacterial resistance; this resistance is associated with decreased clinical efficacy. Topical erythromycin is particularly useful when used with benzoyl peroxide or topical retinoids. Results of clinical studies indicate that combination therapy results in a reduction in total lesion counts of 50 to 70%. /Included in US product label/ For more Therapeutic Uses (Complete) data for Erythromycin (23 total), please visit the HSDB record page. ITS ACTIONS & USES ARE IDENTICAL TO THOSE OF ERYTHROMYCIN. ERYTHROMYCIN MAY BE USEFUL FOR DISSEMINATED GONOCOCCAL DISEASE IN PREGNANT PT WHO IS ALLERGIC TO PENICILLIN... 13 PT...TREATED WITH 500 MG OF ERYTHROMYCIN... STEARATE, GIVEN ORALLY EVERY 6 HR FOR 5 DAYS, SHOWED RAPID CLINICAL & BACTERIOLOGICAL RESPONSES. ANTIBACTERIAL AGENT MEDICATION (VET): ANTIBACTERIAL AGENT Drug Warnings Some commercially available formulations of erythromycin lactobionate powder for injection contain benzyl alcohol as a preservative. Although a causal relationship has not been established, administration of injections preserved with benzyl alcohol has been associated with toxicity in neonates. Toxicity appears to have resulted from administration of large amounts (i.e., about 100-400 mg/kg daily) of benzyl alcohol in these neonates. Although use of drugs preserved with benzyl alcohol should be avoided in neonates whenever possible, the American Academy of Pediatrics states that the presence of small amounts of the preservative in a commercially available injection should not proscribe its use when indicated in neonates. /Erythromycin lactobionate/ In several neonates with infections caused by Ureaplasma urealyticum who received IV administration of erythromycin lactobionate, adverse cardiac effects (e.g., bradycardia, hypotension, cardiac arrest, arrhythmias) requiring cardiopulmonary resuscitation have been reported. Some clinicians state that these adverse effects may depend on serum concentration and/or infusion rate of the drug. It has been suggested that prolonged IV infusion of erythromycin lactobionate (e.g., over 60 minutes) may reduce such adverse cardiac effects. However, it has been suggested that certain individuals may be at increased risk of developing erythromycin-induced adverse cardiac effects and that decreasing the rate of IV infusion may decrease but not eliminate the risk of such effects. Further study is needed to determine the pharmacokinetics and safety of erythromycin lactobionate in neonates. /Erythromycin lactobionate/ Maternal Medication usually Compatible with Breast-Feeding: Erythromycin: Reported Sign or Symptom in Infant or Effect on Lactation: None. /From Table 6/ POTENTIAL ADVERSE EFFECTS ON FETUS: None known. POTENTIAL SIDE EFFECTS ON BREAST-FED INFANT: None known, although theoretically could cause diarrhea in infant. COMMENTS: Crosses placenta in high doses to fetal level 24% of maternal; breast milk may exceed maternal serum concentration. FDA Category: B (B = Studies in laboratory animals have not demonstrated a fetal risk, but there are no controlled studies in pregnant women; or animal studies have shown an adverse effect (other than a decrease in fertility), but controlled studies in pregnant women have not demonstrated a risk to the fetus in the first trimester and there is no evidence of a risk in later trimesters.) /From Table II/ For more Drug Warnings (Complete) data for Erythromycin (17 total), please visit the HSDB record page. ...ERYTHROMYCIN & ITS DERIV SELDOM CAUSE SERIOUS ADVERSE REACTIONS. Pharmacodynamics Macrolides, such as erythromycin, stop bacterial growth by inhibiting protein synthesis and translation, treating bacterial infections. Erythromycin does not exert effects on nucleic acid synthesis. This drug has been shown to be active against most strains of the following microorganisms, effectively treating both in vitro and clinical infections. Despite this, it is important to perform bacterial susceptibility testing before administering this antibiotic, as resistance is a common issue that may affect treatment. **A note on antimicrobial resistance, pseudomembranous colitis, and hepatotoxicity** Many strains of Haemophilus influenzae are resistant to erythromycin alone but are found to be susceptible to erythromycin and sulfonamides used in combination. It is important to note that Staphylococci that are resistant to erythromycin may emerge during erythromycin and/or sulfonamide therapy. Pseudomembranous colitis has been reported with most antibacterial agents, including erythromycin, and may range in severity from mild to life-threatening. Therefore, the physician should consider this diagnosis in patients with diarrhea after the administration of antibacterial agents. Erythromycin can cause hepatic dysfunction, cholestatic jaundice, and abnormal liver transaminases, particularly when erythromycin estolate is administered. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 100 mg/mL (136.25 mM) Ethanol : ~100 mg/mL H2O : 1 mg/mL (1.36 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (2.83 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (2.83 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (2.83 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 10% DMSO+40% PEG300+5% Tween-80+45% Saline: ≥ 2.08 mg/mL (2.83 mM) Solubility in Formulation 5: 5 mg/mL (6.81 mM) in 0.5% CMC-Na 0.1% Tween-80 (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3625 mL | 6.8126 mL | 13.6253 mL | |
| 5 mM | 0.2725 mL | 1.3625 mL | 2.7251 mL | |
| 10 mM | 0.1363 mL | 0.6813 mL | 1.3625 mL |