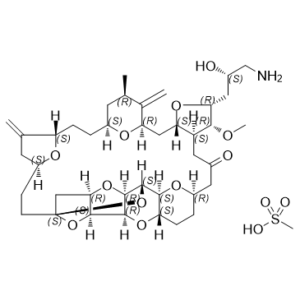
Eribulin mesylate (E7389 mesylate) is the mesylate salt of Eribulin (E-7389; Halaven; B1939; ER-086526; Eisai), which a synthetic analogue of the marine-originated natural product halichondrin B, is an antitubulin/microtubule targeting agent (MTA)/mitotic/tubulin inhibitor approved for use in the treatment of metastatic breast cancer. Eribulin (E7389) inhibits the proliferation of cancer cells by binding microtubule proteins and microtubules.
Physicochemical Properties
| Molecular Formula | C40H59NO11.CH4O3S |
| Molecular Weight | 826.002220000001 |
| Exact Mass | 825.396 |
| Elemental Analysis | C, 59.62; H, 7.69; N, 1.70; O, 27.12; S, 3.88 |
| CAS # | 441045-17-6 |
| Related CAS # | Eribulin;253128-41-5;Eribulin-d3 mesylate |
| PubChem CID | 17755248 |
| Appearance | White to off-white solid powder |
| LogP | 4.723 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 57 |
| Complexity | 1470 |
| Defined Atom Stereocenter Count | 19 |
| SMILES | C=C1C[C@@](CC[C@@]2(C[C@@]3([H])O4)O[C@]([C@](O[C@](C5)([H])CC6)([H])[C@@]6([H])O7)([H])[C@@]4([H])[C@]7([H])[C@@]3([H])O2)([H])O[C@@]1([H])CC[C@](C[C@@H](C)C8=C)([H])O[C@]8([H])C[C@@](O[C@H](C[C@H](O)CN)[C@@H]9OC)([H])[C@]9([H])CC5=O.CS(=O)(O)=O |
| InChi Key | QAMYWGZHLCQOOJ-WRNBYXCMSA-N |
| InChi Code | InChI=1S/C40H59NO11.CH4O3S/c1-19-11-24-5-7-28-20(2)12-26(45-28)9-10-40-17-33-36(51-40)37-38(50-33)39(52-40)35-29(49-37)8-6-25(47-35)13-22(42)14-27-31(16-30(46-24)21(19)3)48-32(34(27)44-4)15-23(43)18-41;1-5(2,3)4/h19,23-39,43H,2-3,5-18,41H2,1,4H3;1H3,(H,2,3,4)/t19-,23+,24+,25-,26+,27+,28+,29+,30-,31+,32-,33-,34-,35+,36+,37+,38-,39+,40+;/m1./s1 |
| Chemical Name | (1S,3S,6S,9S,12S,14R,16R,18S,20R,21R,22S,26R,29S,31R,32S,33R,35R,36S)-20-[(2S)-3-amino-2-hydroxypropyl]-21-methoxy-14-methyl-8,15-dimethylidene-2,19,30,34,37,39,40,41-octaoxanonacyclo[24.9.2.13,32.13,33.16,9.112,16.018,22.029,36.031,35]hentetracontan-24-one;methanesulfonic acid |
| Synonyms | E7389 mesylate; E-7389; Halaven; B1939; ER-086526; E-7389 mesylate; E 7389 mesylate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Eribulin mesylate (1-100 nM; 72 h) suppresses cell growth with IC50 values of 22.8 and 21.5 nM for LM8 and Dunn cells respectively[1]. Eribulin mesylate (10-50 nM; 12-72 hours) significantly promotes early apoptosis in LM8 cells treated at a dosage of 50 nM for 24 hours [1]. In LM8 cells, eribulin mesylate (10-50 nM; 12-72 hours) induced G2/M phase arrest at 50 nM for 12 hours, while long-term treatment at 10 nM (72 hour) will not induce G2/M phase arrest[1]. Eribulin mesylate (1-50 nM; 12 hours) does not promote senescence in LM8 cells [1]. Eribulin mesylate (1-10 nM; 16 hours) produces morphological alterations and inhibits cell migration in LM8 cells at low concentrations [1]. |
| ln Vivo | In mice with osteosarcoma, eribulin mesylate (1 mg/kg; intravenously given once weekly for two weeks) decreases the growth of the primary tumor and lung metastases [1]. At low concentrations, eribulin mesylate (1 mg/kg; intravenously administered once) inhibits the emergence of circulating tumor cells (CTCs) [1]. |
| Cell Assay |
Cell proliferation assay[1] Cell Types: LM8 cells and Dunn cells Tested Concentrations: 0, 1, 10, 100 nM Incubation Duration: 72 hrs (hours) Experimental Results: Inhibition of cell proliferation in a dose-dependent manner. Apoptosis analysis [1] Cell Types: LM8 cells Tested Concentrations: 0, 10, 50 nM Incubation Duration: 12, 24, 48, 72 hrs (hours) Experimental Results: 50 nM concentration induced early apoptosis after 12 hrs (hours). No apoptosis was detected at 10 nM concentration. Cell cycle analysis[1] Cell Types: LM8 Cell Tested Concentrations: 0, 10, 50 nM Incubation Duration: 12, 24, 48, 72 hrs (hours) Experimental Results: Treatment with 50 nM for 12 hrs (hours) induces G2/M arrest. 10 nM treatment did not induce G2/M arrest. |
| Animal Protocol |
Animal/Disease Models: C3H/HeN mice (4 weeks old) were injected with LM8 cells [1] Doses: 1 mg/kg Route of Administration: intravenous (iv) (iv)injection once a week for 2 weeks Experimental Results: Inhibited primary tumor growth and induced tumor cell apoptosis Death. Reduce lung metastasis. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of eribulin during breastfeeding. The manufacturer recommends that breastfeeding be discontinued during eribulin therapy and for 2 weeks after the last dose. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References |
[1]. Low-dose eribulin reduces lung metastasis of osteosarcoma in vitro and in vivo. Oncotarget. 2019 Jan 4; 10(2): 161-174. [2]. Eribulin binds at microtubule ends to a single site on tubulin to suppress dynamic instability. Biochemistry, 2010. 49(6): p. 1331-7. [3]. Inhibition of centromere dynamics by eribulin (E7389) during mitotic metaphase. Mol Cancer Ther, 2008. 7(7): p. 2003-11. [4]. Eribulin induces irreversible mitotic blockade: implications of cell-based pharmacodynamics for in vivo efficacy under intermittent dosing conditions. Cancer Res, 2011. 71(2): p. 496-505. |
| Additional Infomation |
Eribulin mesylate is a methanesulfonate salt obtained by reaction of eribulin with one equivalent of methanesulfonic acid. A fully synthetic macrocyclic ketone analogue of marine sponge natural products. Inhibits growth phase of microtubules via tubulin-based antimitotic mechanism, which leads to G2/M cell-cycle block, disruption of mitotic spindles, and, ultimately, apoptotic cell death after prolonged mitotic blockage. It has a role as an antineoplastic agent and a microtubule-destabilising agent. It contains an eribulin(1+). Eribulin Mesylate is the mesylate salt of a synthetic analogue of halichondrin B, a substance derived from a marine sponge (Lissodendoryx sp.) with antineoplastic activity. Eribulin binds to the vinca domain of tubulin and inhibits the polymerization of tubulin and the assembly of microtubules, resulting in inhibition of mitotic spindle assembly, induction of cell cycle arrest at G2/M phase, and, potentially, tumor regression. See also: Eribulin (has active moiety). |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 100 mg/mL (~121.07 mM) Ethanol :≥ 100 mg/mL (~121.07 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.03 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.03 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (3.03 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: ≥ 2.5 mg/mL (3.03 mM) (saturation unknown) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear EtOH stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 5: ≥ 2.5 mg/mL (3.03 mM) (saturation unknown) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear EtOH stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 6: ≥ 2.5 mg/mL (3.03 mM) (saturation unknown) in 10% EtOH + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear EtOH stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2107 mL | 6.0533 mL | 12.1065 mL | |
| 5 mM | 0.2421 mL | 1.2107 mL | 2.4213 mL | |
| 10 mM | 0.1211 mL | 0.6053 mL | 1.2107 mL |