Physicochemical Properties
| Molecular Formula | C19H21FN2O2 |
| Molecular Weight | 328.38 |
| Exact Mass | 328.158 |
| Elemental Analysis | C, 69.49; H, 6.45; F, 5.79; N, 8.53; O, 9.74 |
| CAS # | 130581-13-4 |
| Related CAS # | Eplivanserin hemifumarate;130580-02-8;Eplivanserin;130579-75-8 |
| PubChem CID | 135515430 |
| Appearance | Off-white to pink solid powder |
| LogP | 4.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 24 |
| Complexity | 418 |
| Defined Atom Stereocenter Count | 0 |
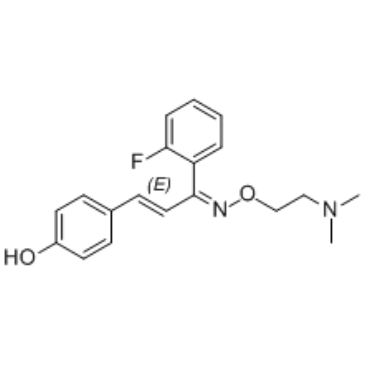
| SMILES | CN(C)CCON=C(C=CC1=CC=C(C=C1)O)C2=CC=CC=C2F |
| InChi Key | VAIOZOCLKVMIMN-FOUXOUMPSA-N |
| InChi Code | InChI=1S/C19H21FN2O2/c1-22(2)13-14-24-21-19(17-5-3-4-6-18(17)20)12-9-15-7-10-16(23)11-8-15/h3-12,23H,13-14H2,1-2H3/b12-9+,21-19+ |
| Chemical Name | 4-[(E,3E)-3-[2-(dimethylamino)ethoxyimino]-3-(2-fluorophenyl)prop-1-enyl]phenol |
| Synonyms | Eplivanserin; 130579-75-8; SR-46349; SR46349; 3CO94WO6DJ; 130581-13-4; (E)-1-(2-fluorophenyl)-3-(4-hydroxyphenyl)-2-propen-1-one O-[2-(dimethylamino)ethyl]oxime; 4-[(E,3Z)-3-[2-(dimethylamino)ethoxyimino]-3-(2-fluorophenyl)prop-1-enyl]phenol; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product is not stable in solution, please use freshly prepared working solution for optimal results. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 5-HT2A Receptor |
| ln Vitro | Eplivanserin (SR 46349B) has mild inhibitory effects on other 5-HT kinases with IC50 of 0.12 μM (porcine cortical 5-HT1C), 14 μM (rat hippocampus 5-HT1A), and 16 μM (rat stnatum 5-HT1B , Ox) caudate nucleus 5-HT1D). Eplivanserin also inhibits rat cortical epinephrine α1 and α2, rat whole brain histamine H1, Na+ channels, and rat striatum dopamine D1 and D2, with IC50 of 3.4 μM, 1.0 μM, 5.0 μM, and 39 μM correspondingly. , 9 μM and 28 μM, respectively [1]. |
| ln Vivo | The 5-HT2 receptor binding of [3H]ketanserin is inhibited by eplivanserin halffumarate (SR 46349B), with an ED50 of 0.087 mg/kg following intraperitoneal injection and 0.097 mg/kg following oral dosing in mice [1]. Following repeated cocaine treatment, SR 46349B (0.25–1 mg/kg; i.p.) inhibits the hyperactivity caused by cocaine in rats [2]. |
| Enzyme Assay | A new potent, selective and p.o. active serotonergic [5-hydroxytryptamine (5-HT2)] receptor antagonist, Eplivanserin (SR-46349) [trans, 4-([3Z)3-(2-dimethylaminoethyl)oxyimino-3(2-flurophenyl++ +)propen-1-yl]phenol hemifumarate) has been characterized by a series of "in vitro" and "in vivo" methods. Based upon binding studies with 5-HT2 receptors in rat brain cortical membranes and blockade of 5-HT-induced contractions in isolated tissues (rabbit thoracic aorta, rat jugular vein, rat caudal artery, rat uterus and guinea pig trachea), Eplivanserin (SR-46349) showed high affinity for 5-HT2 receptors. Furthermore, Eplivanserin (SR-46349) displayed moderate affinity for the 5-HT1C receptor and had no affinity for the other 5-HT1 subclass (5-HT1A, 5-HT1B or 5-HT1D), dopamine (D1 or D2), "alpha" adrenergic (alpha-1 or alpha-2), sodium and calcium channel and histamine (H1) receptors. It did not interact with histamine (H1), alpha-1 adrenergic and 5-HT3 receptors in smooth muscle preparations. No inhibition of the uptake of norepinephrine, dopamine or 5-HT was seen. Based upon blockade of pressor responses to 5-HT in pithed rats and in vivo binding studies in mice, SR 46349B was found to be a potent and p.o. active 5-HT2 receptor antagonist with a relatively long duration of action. Behavioral experiments, including mescaline- and 5-hydroxytryptophan-induced head twitches and learned helplessness, as well as sleep-waking cycle and EEG spectral parameter studies, indicated that SR 46349B has a classical 5-HT2 psychopharmacological antagonist profile.[1] |
| Animal Protocol | The 5-HT2AR antagonist Eplivanserin (SR-46349) also blocked cocaine-evoked hyperactivity following repeated cocaine treatment, whereas the other 5-HT2R ligands were ineffective. When any of the 5-HT2R ligands was coadministered with cocaine during the treatment regimen (10 mg/kg/day for 5 days), the development of sensitization was unchanged as measured by the level of cocaine-evoked hyperactivity upon challenge 5 days after termination of the treatment. The present study implies that 5-HT2AR and 5-HT2CR exert oppositional influence upon hyperactivity evoked by acute administration of cocaine; this balance is altered following repeated cocaine administration. |
| References |
[1]. Biochemical and pharmacological properties of SR 46349B, a new potent and selective 5-hydroxytryptamine2 receptor antagonist. J Pharmacol Exp Ther. 1992 Aug;262(2):759-68. [2]. Contribution of serotonin (5-hydroxytryptamine; 5-HT) 5-HT2 receptor subtypes to the hyperlocomotor effects of cocaine: acute and chronic pharmacological analyses. J Pharmacol Exp Ther. 2004 Sep;310(3):1246-54. [3]. COMPOSITIONS CONTAINING A SEROTONIN SELECTIVE REUPTAKE INHIBITOR AND A 5-HT2A RECEPTOR ANTAGONIST. WO 2005/002578 A1. |
| Additional Infomation |
Eplivanserin has been used in trials studying the treatment of Sleep, Insomnia, Chronic Pain, Fibromyalgia, and Primary Insomnia, among others. Drug Indication Insomnia The role of serotonin (5-hydroxytryptamine; 5-HT) 5-HT2 receptor subtypes (5-HT2AR, 5-HT2BR, and 5-HT2CR) in acute cocaine-evoked hyperactivity was compared with their contribution to the development and expression of locomotor sensitization upon repeated, intermittent treatment with cocaine (10 mg/kg/day for 5 days) in male Wistar rats. Cocaine-evoked hyperactivity was significantly enhanced by pretreatment with the preferential 5-HT2AR agonist 1-(2,5-dimethoxy-4-iodophenyl)-2-aminopropane (DOI) and the 5-HT2CR antagonist SDZ SER-082 [(+)-cis-4,5,7a,8,9,10,11,11a-octahydro-7H-10-methylindolo(1,7-BC)(2,6) naphthyridine fumarate]. The 5-HT2AR antagonist SR 46349B [1(Z)-[2-(dimethylamino)ethoxyimino]-1(2-fluorophenyl)-3-(4-hydroxyphenyl)-2(E)-propene] and the preferential 5-HT2CR agonist MK 212 [6-chloro-2-(1-piperazinyl)pyrazine HCl] (2 mg/kg) significantly attenuated acute cocaine-evoked hyperactivity; however, a lower dose of MK 212 (0.3 mg/kg) enhanced cocaine-evoked hyperactivity. The 5-HT2BR agonist BW 723C86 (1-[5-(2-thienylmethoxy)-1H-3-indolyl]propan-2-amine HCl) and the 5-HT2BR antagonist SB 204741 [N-(1-methyl-5-indolyl)-N'-(3-methyl-5-isothiazolyl) urea] had no effect on cocaine-evoked hyperactivity. Repeated treatment with cocaine alone resulted in a 2-fold increase in hyperactivity upon challenge with cocaine 5 days after termination of the cocaine regimen (sensitization). The 5-HT2AR antagonist SR 46349B also blocked cocaine-evoked hyperactivity following repeated cocaine treatment, whereas the other 5-HT2R ligands were ineffective. When any of the 5-HT2R ligands was coadministered with cocaine during the treatment regimen (10 mg/kg/day for 5 days), the development of sensitization was unchanged as measured by the level of cocaine-evoked hyperactivity upon challenge 5 days after termination of the treatment. The present study implies that 5-HT2AR and 5-HT2CR exert oppositional influence upon hyperactivity evoked by acute administration of cocaine; this balance is altered following repeated cocaine administration. [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~125 mg/mL (~380.66 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.33 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.33 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (6.33 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0453 mL | 15.2263 mL | 30.4525 mL | |
| 5 mM | 0.6091 mL | 3.0453 mL | 6.0905 mL | |
| 10 mM | 0.3045 mL | 1.5226 mL | 3.0453 mL |