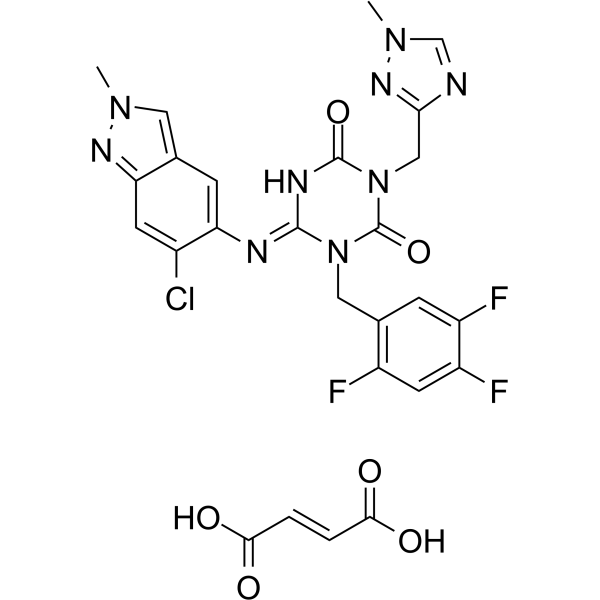
Ensitrelvir fumarate (S-217622 fumarate; Xocova) is a novel and orally bioavailable SARS-CoV-2 3CL protease inhibitor (IC50=13 nM) approval in Japan under the emergency regulatory approval system (EUA) for COVID-19 treatment. It is an antiviral drug developed by Shionogi in partnership with Hokkaido University. It became the first Japanese domestic pill to treat COVID-19, third to be regulatorally approved in Japan; in February 2022.
Physicochemical Properties
| Molecular Formula | C26H21CLF3N9O6 |
| Molecular Weight | 647.949853658676 |
| Exact Mass | 647.125 |
| Elemental Analysis | C, 48.20; H, 3.27; Cl, 5.47; F, 8.80; N, 19.46; O, 14.81 |
| CAS # | 2757470-18-9 |
| Related CAS # | 2647530-73-0;2757470-18-9 (fumarate); |
| PubChem CID | 162623410 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 45 |
| Complexity | 1040 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CN1C=C2C=C(C(=CC2=N1)Cl)NC3=NC(=O)N(C(=O)N3CC4=CC(=C(C=C4F)F)F)CC5=NN(C=N5)C.C(=C/C(=O)O)\C(=O)O |
| InChi Key | FBOCUALVLIWPNQ-WLHGVMLRSA-N |
| InChi Code | InChI=1S/C22H17ClF3N9O2.C4H4O4/c1-32-7-12-4-18(13(23)5-17(12)30-32)28-20-29-21(36)35(9-19-27-10-33(2)31-19)22(37)34(20)8-11-3-15(25)16(26)6-14(11)245-3(6)1-2-4(7)8/h3-7,10H,8-9H2,1-2H3,(H,28,29,36)1-2H,(H,5,6)(H,7,8)/b2-1+ |
| Chemical Name | (E)-6-((6-chloro-2-methyl-2H-indazol-5-yl)imino)-3-((1-methyl-1H-1,2,4-triazol-3-yl)methyl)-1-(2,4,5-trifluorobenzyl)-1,3,5-triazinane-2,4-dione fumaric acid |
| Synonyms | S 217622; S217622; Xocova;S-217622 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | SARS-CoV-2 3CL protease | |
| ln Vitro |
|
|
| ln Vivo |
|
|
| Enzyme Assay |
3CL Protease Inhibition Assay[3] The 3CL protease inhibition assay was conducted in 384-well plates. The substance solution (10 mM dimethyl sulfoxide [DMSO] solution) was diluted to 250 μmol/L stepwise with a threefold dilution with DMSO. Finally, the solutions were mixed with 20 mmol/L Tris-HCl (pH 7.5) as a compound solution. Ten microliters of compound solution was added manually to each well, and then 5 μL of 16 μM substrate in inhibition buffer (2 mM EDTA, 20 mM DTT, 0.02% BSA, and 20 mM Tris-HCl, pH 7.5) was added. The reaction was initiated by adding 5 μL of 12 nM 3CL protease) in an inhibition buffer and incubated at room temperature for 3 h. The following operations were the same as those described in the Biological Screening. |
|
| Cell Assay |
Cellular Antiviral Activity[3] Antiviral activity against SARS-CoV-2, SARS-CoV, MERS-CoV, and HCoV-229E was assessed by monitoring the cell viability; that against HCoV-OC43 was assessed by monitoring viral RNA in a cell suspension. EC50 values were determined by plotting the compound concentration versus inhibition and fitting data with a four-parameter logistical fit. EC90 values against HCoV-OC43 were determined from the resulting dose–response curves and calculated with the two-point method. Antiviral activities against SARS-CoV-2 were evaluated using VeroE6/TMPRSS2 cells. VeroE6/TMPRSS2 cells (1.5 × 104/well) suspended in minimum essential medium (MEM) supplemented with heat-inactivated 2% FBS were seeded into 96-well plates with diluted compounds in each well. Cells were infected with each SARS-CoV-2 at 30–3000 TCID50/well and cultured at 37 °C with 5% CO2 for 3 days or 4 days. Cell viability was assessed using a CellTiter-Glo 2.0 assay. The CC50 was assessed in the absence of viruses after being cultured for 3 days. |
|
| Animal Protocol |
In Vivo SARS-CoV-2 Infection and Treatment Studies[3] In vivo SARS-CoV-2 infection experiments were conducted in accordance with the guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). The animal study protocol was approved by the director of the institute based on the report of the Institutional Animal Care and Use Committee of Shionogi Research Laboratories. Mouse in vivo SARS-CoV-2 infection studies were done at Shionogi Pharmaceutical Research Center. Five-week-old female BALB/cAJcl mice (n = 5 or 10 per group) were intranasally inoculated with SARS-CoV-2 Gamma strain (hCoV-19/Japan/TY7-501/2021) (10000 TCID50/mouse) under anesthesia. Immediately after infection, the mice were orally administered S-217622 fumaric acid (2, 8, 16, or 32 mg/kg q12h; n = 5 per group) or vehicle (0.5 w/v% methyl cellulose in aqueous solution q12h; n = 10 per group) for 1 day. Twenty-four hours postinfection, the mice were euthanized via cervical dislocation under anesthesia; their lungs were removed, and the viral titers in the lung homogenates were determined using VeroE6/TMPRSS2 cells. Viral titers are expressed as log10 TCID50/mL. |
|
| References |
[1]. COVID-19, Influenza and RSV: Surveillance-informed prevention and treatment - Meeting report from an isirv-WHO virtual conference. Antiviral Res. 2022;197:105227. [2]. Discovery of S-217622, a Non-Covalent Oral SARS-CoV-2 3CL Protease Inhibitor Clinical Candidate for Treating COVID-19. bioRxiv 2022.01.26.477782. |
|
| Additional Infomation | The coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has resulted in millions of deaths and threatens public health and safety. Despite the rapid global spread of COVID-19 vaccines, effective oral antiviral drugs are urgently needed. Here, we describe the discovery of S-217622, the first oral noncovalent, nonpeptidic SARS-CoV-2 3CL protease inhibitor clinical candidate. S-217622 was discovered via virtual screening followed by biological screening of an in-house compound library, and optimization of the hit compound using a structure-based drug design strategy. S-217622 exhibited antiviral activity in vitro against current outbreaking SARS-CoV-2 variants and showed favorable pharmacokinetic profiles in vivo for once-daily oral dosing. Furthermore, S-217622 dose-dependently inhibited intrapulmonary replication of SARS-CoV-2 in mice, indicating that this novel noncovalent inhibitor could be a potential oral agent for treating COVID-19.[3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~77.17 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.86 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (3.86 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (3.86 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5433 mL | 7.7166 mL | 15.4333 mL | |
| 5 mM | 0.3087 mL | 1.5433 mL | 3.0867 mL | |
| 10 mM | 0.1543 mL | 0.7717 mL | 1.5433 mL |