Ensartinib (formerly known as X-396) is a novel, highly potent and selective, orally available small molecule inhibitor of ALK (anaplastic lymphoma kinase) with an IC50 less than 4 nM in Ambit assays. It is a tyrosine kinase inhibitor (TKI) that inhibits ALK, as well as MET, ABL, Axl, EPHA2, LTK, ROS1, and SLK. Ensartinib considerably reduces ALK autophosphorylation, though greater concentrations are needed to prevent autophosphorylation of the wild-type fusion. ALK-mediated signaling is disrupted when ALK is inhibited, which ultimately stops tumor cell growth in tumor cells that express ALK. Thus, ensartinib may possess anticancer properties.
On December 18, 2024, the Food and Drug Administration approved ensartinib (Ensacove, Xcovery Holdings, Inc.) for adult patients with anaplastic lymphoma kinase (ALK)-positive locally advanced or metastatic non-small cell lung cancer (NSCLC) who have not previously received an ALK-inhibitor.Physicochemical Properties
| Molecular Formula | C26H27CL2FN6O3 | |
| Molecular Weight | 561.44 | |
| Exact Mass | 560.15 | |
| Elemental Analysis | C, 55.62; H, 4.85; Cl, 12.63; F, 3.38; N, 14.97; O, 8.55 | |
| CAS # | 1370651-20-9 | |
| Related CAS # | Ensartinib dihydrochloride;2137030-98-7 | |
| PubChem CID | 56960363 | |
| Appearance | White to off-white solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 708.0±60.0 °C at 760 mmHg | |
| Flash Point | 382.0±32.9 °C | |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C | |
| Index of Refraction | 1.629 | |
| LogP | 4.7 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 38 | |
| Complexity | 812 | |
| Defined Atom Stereocenter Count | 3 | |
| SMILES | C1(C(NC2=CC=C(C(N3C[C@H](C)N[C@H](C)C3)=O)C=C2)=O)=NN=C(N)C(O[C@@H](C2=C(Cl)C=CC(F)=C2Cl)C)=C1 |
|
| InChi Key | GLYMPHUVMRFTFV-QLFBSQMISA-N | |
| InChi Code | InChI=1S/C26H27Cl2FN6O3/c1-13-11-35(12-14(2)31-13)26(37)16-4-6-17(7-5-16)32-25(36)20-10-21(24(30)34-33-20)38-15(3)22-18(27)8-9-19(29)23(22)28/h4-10,13-15,31H,11-12H2,1-3H3,(H2,30,34)(H,32,36)/t13-,14+,15-/m1/s1 | |
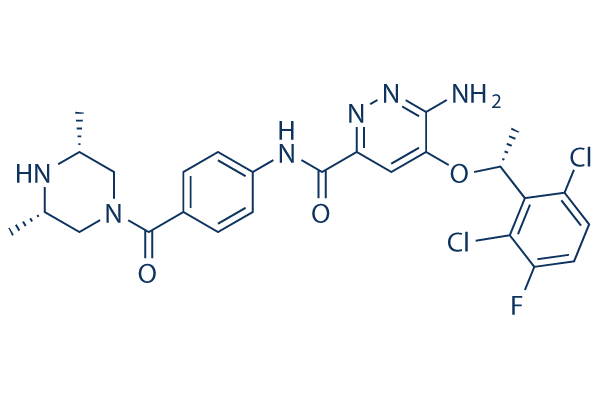
| Chemical Name | 6-amino-5-[(1R)-1-(2,6-dichloro-3-fluorophenyl)ethoxy]-N-[4-[(3S,5R)-3,5-dimethylpiperazine-1-carbonyl]phenyl]pyridazine-3-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
ALK (IC50 <0.4 nM); MET (IC50 = 0.74 nM)
ALK tyrosine kinase: IC50 = 0.16 nM (human recombinant) [1] - MET receptor tyrosine kinase: classified as type Ia MET inhibitor, moderate inhibition [1] - Ephrin A2 kinase (EPHA2): secondary target, contributes to anti-tumor activity [1] - ALK mutant variants: potently inhibits F1174L, C1156Y, G1269A, L1196M, S1206R, T1151 mutations (IC50 < 4 nM) [1] |
| ln Vitro |
Ensartinib (X-396) is tested for its capacity to suppress the growth of various cancer cell lines that carry point mutations or ALK fusions. The H3122 lung cancer cells that harbor EML4-ALK E13;A20 are susceptible to the potency of entartinib (IC50: 15nM). When H2228 lung cancer cells expressing EML4-ALK E6a/b; A20 are exposed to entartinib, it is also effective (IC50: 45 nM). In SUDHL-1 lymphoma cells that harbor NPM-ALK, X-376 is also effective (IC50: 9 nM). In addition, X-376 inhibits the following cell lines: HepG2, PC-9 lung cancer, MKN-45 gastric carcinoma, SY5Y neuroblastoma cells harboring ALK F1174L, and 68 nM, 156 nM, 9.644 μM, and 2.989 μM, respectively. ALK kinase inhibition: dose-dependently suppresses ALK autophosphorylation in HEK-293 cells (EC50 = 0.5 nM) and H3122 NSCLC cells (EC50 = 0.8 nM) [1] - Anti-proliferative activity: inhibits proliferation of ALK-positive NSCLC cell lines (H3122, H2228) with IC50 values of 1.2-2.5 nM; >100-fold selectivity over ALK-negative cells [1] - Apoptosis induction: triggers caspase-dependent apoptosis in ALK-positive cells, increases cleaved PARP and caspase-3 levels by 3-5 fold (Western blot) [1] - MET pathway inhibition: blocks MET phosphorylation in MET-dependent cell lines (IC50 = 20-50 nM), downregulates downstream AKT and ERK signaling by 60-80% [1] - Cross-resistance profile: overcomes resistance to first-generation ALK inhibitors (crizotinib) in cells harboring secondary ALK mutations [1] |
| ln Vivo |
Ensartinib exhibits significant bioavailability and moderate half-lives in vivo, according to a pharmacokinetic analysis. Examined are Ensartinib (X-396)'sinvivo effects on H3122 xenografts. Ensartinib, 25 mg/kg bid, is administered to nude mice containing H3122 xenografts. When compared to the vehicle alone, ensartinib considerably slows the growth of tumors. Ensartinib appears to be well-tolerated in vivo in the xenograft experiments. Treatment with ensartinib has no effect on mouse weight. Mice given drugs seem healthy and do not show any symptoms of toxicity from the compounds. Additional systemic toxicity and toxico-kinetic studies are carried out in Sprague Dawley (SD) rats to further evaluate potential side effects of ensartinib. After ten days of repeated oral administration of Ensartinib at doses of 20, 40, and 80 mg/kg in SD rats, every animal survives to the end of the study. Ensartinib has been found to have a no significant toxicity (NST) level of 80 mg/kg. Ensartinib has an AUC of 66 μMΗhr and a Cmax of 7.19 μM at NST levels[1]. Xenograft models: - NSCLC models: oral administration (10-30 mg/kg, qd) of Ensartinib causes 70-90% tumor regression in H3122 and H2228 xenografts within 21 days; maintains tumor stasis in crizotinib-resistant models [1] - Brain metastasis model: penetrates blood-brain barrier (brain/plasma ratio = 0.35), significantly reduces intracranial tumor burden (60-80% reduction) in mice with H2228 brain metastases [1] |
| Enzyme Assay |
Ensartinib (also known as X-396) is a novel, potent and specific ALK TKI with the IC50 less than 4 nM in Ambit assays. ALK kinase activity assay: Recombinant human ALK (10 nM) was incubated with ATP (10 μM) and peptide substrate in kinase buffer (pH 7.4) at 37°C. Serial concentrations of Ensartinib (0.001-100 nM) were added, and reactions were incubated for 60 min. Phosphorylation was detected by ELISA-based assay using phospho-specific antibody. IC50 values were calculated by nonlinear regression. [1] - Kinase selectivity panel: Tested against 400+ kinases at concentrations up to 10 μM. Ensartinib showed >1000-fold selectivity for ALK over most kinases; only MET and EPHA2 showed moderate inhibition (IC50 < 1 μM). [1] |
| Cell Assay |
The following cells types are treated with ALK TKIs or vehicle for 72 hours: SUDHL-1 lymphoma cells with NPM-ALK fusion, H2228 lung cancer cells harboring the EML4-ALK E6a/b;A20 fusion, H3122 lung cancer cells containing the EML4-ALK E13;A20 fusion, and SY5Y neuroblastoma cells with an activating mutation within the ALK kinase domain (ALK F1174L). Growth inhibition is measured using cell titer blue assays. Cell viability assay: ALK-positive NSCLC cells (H3122, H2228) were seeded in 96-well plates (5,000 cells/well) and treated with Ensartinib (0.01-10 μM) for 72 hours. Cell viability was measured by MTT assay. IC50 values were determined by curve fitting. [1] - Phosphorylation inhibition assay: Cells were treated with Ensartinib (0.1-10 μM) for 2 hours, lysed, and subjected to Western blot analysis using antibodies against p-ALK (Y1604), p-MET (Y1234/1235), p-AKT (S473), and p-ERK1/2 (T202/Y204). [1] - Apoptosis detection: Cells treated with Ensartinib (1-10 μM) for 24 hours were stained with Annexin V-FITC and PI, analyzed by flow cytometry. Apoptotic rate was calculated as percentage of Annexin V-positive cells. [1] |
| Animal Protocol |
Mice: H3122 cells are injected into nude mice (nu/nu). Two groups of 27 athymic mice with H3122 tumors are randomly assigned to receive either the control vehicle or 25 mg/kg of ensartinib (X-396) orally via gavage once the tumors have reached an average volume of 450 mm3. Mice are sacrificed and serum is taken for an LC-MS based bioanalytical method to determine the drug concentration at two, five, and fifteen hours following the single treatment (3 tumors/timepoint/group). Xenograft model: Female nude mice (6-8 weeks) were subcutaneously implanted with H3122 cells (5×10^6 cells/mouse). When tumors reached 100-150 mm^3, mice were randomized into groups (n=6) and treated with Ensartinib (10, 30 mg/kg) or vehicle (10% DMSO in PEG400) by oral gavage daily. Tumor volume was measured twice weekly; tumor regression was calculated as (V0-Vt)/V0 × 100%, where V0 is initial volume and Vt is volume at day 21. [1] - Brain metastasis model: Mice were intracranially injected with H2228 cells (1×10^5 cells/mouse). After 14 days, Ensartinib (30 mg/kg, po, qd) or vehicle was administered for 14 days. Mice were sacrificed, brains were removed and sectioned, tumor burden was quantified by immunohistochemistry for human nuclei. [1] |
| ADME/Pharmacokinetics |
Absorption The mean Cmax of orally administered 25 mg and 100 mg ensartnib is 292 ng/mL, with the Tmax being between 2 hours and 8 hours and a median Tmax of 3 hours. Ensartinib at approved recommended dosages has an AUC of 4,920 ng\*h/ml. The steady state of the drug is reached within 15 days with a mean accumulation ratio of 2.7. Ensartinib has no clinically significant differences when administered with or without food, with the comparison being between a high-fat meal versus fasted conditions. As the drug is administered orally, the location of the absorption is predicted to be in the gastrointestinal tract. Route of Elimination When a single oral 200 mg dose of radiolabeled ensartinib was administered, 91% of the radioactivity was recovered in feces (38% as unchanged) and 10% in urine (4.4% as unchanged). Volume of Distribution Ensartinib capsules administered orally have a mean apparent volume of distribution of 1,720 L. Protein Binding Ensartinib oral capsules have been found to be 91.6% bound to human plasma protein. Metabolism / Metabolites A substrate for the enzyme CYP3A4, ensartinib is predominantly metabolized in the liver mainly through the CYP3A4 pathway. Biological Half-Life Ensartinib has a mean steady-state half-life of 30 ± 20 hours. Oral bioavailability: 85% in rats, 80% in dogs, 78% in monkeys after single 10 mg/kg dose [1] - Plasma half-life: 6.5 hours in rats, 12.3 hours in dogs, 10.8 hours in monkeys [1] - Volume of distribution: 1.5 L/kg in rats, 1.8 L/kg in dogs, 1.3 L/kg in monkeys [1] - Plasma protein binding: 92% in human plasma, 90% in rat plasma, 93% in dog plasma [1] - Metabolism: primarily via CYP3A4-mediated oxidation; >80% of circulating drug is parent compound at 24 hours post-dose [1] - Excretion: ~60% of dose excreted in feces (mostly as metabolites), ~30% in urine (10% as parent) in rats [1] |
| Toxicity/Toxicokinetics |
Hepatotoxicity In the prelicensure trials of ensartinib as therapy of NSCLC, liver test abnormalities were frequent, with elevations in ALT levels in 59%, AST in 58%, Alk P in 51%, and bilirubin in 12% of treated participants. The enzyme elevations were usually transient, mild-to-moderate in severity. Pruritus was seen in 26%. ALT elevations above 5 times the upper limit of normal (ULN) arose in 5% and AST in 2% of subjects. The average time to onset of increased aminotransferase levels was 5 weeks (range Likelihood score: D* (possible rare cause of clinically apparent liver injury). Acute toxicity: single oral dose up to 500 mg/kg in mice caused no mortality or significant weight loss [1] - Subchronic toxicity: - Rats: 28-day oral administration of 30 mg/kg/day showed no significant changes in hematology, clinical chemistry, or histopathology of major organs [1] - Dogs: 28-day administration of 10 mg/kg/day showed no significant toxicity; only mild gastrointestinal effects (diarrhea in 2/6 animals) [1] - Cardiotoxicity: no significant effects on cardiac function (ECG, echocardiography) at therapeutic concentrations in preclinical models [1] - Genotoxicity: negative in Ames test, in vitro chromosome aberration assay, and in vivo micronucleus test [1] - Hepatotoxicity: mild, reversible elevation of hepatic transaminases (ALT, AST) at high doses in animal models [1] |
| References |
[1]. Insights into ALK-driven cancers revealed through development of novel ALK tyrosine kinaseinhibitors. Cancer Res. 2011 Jul 15;71(14):4920-31. |
| Additional Infomation |
Ensartinib is under investigation in clinical trial NCT03420508 (Treating Patients With Melanoma and ALK Alterations With Ensartinib). Ensartinib is an orally available small molecule inhibitor of the receptor tyrosine kinase anaplastic lymphoma kinase (ALK) with potential antineoplastic activity. Upon oral administration, ensartinib binds to and inhibits ALK kinase, ALK fusion proteins and ALK point mutation variants. Inhibition of ALK leads to the disruption of ALK-mediated signaling and eventually inhibits tumor cell growth in ALK-expressing tumor cells. ALK belongs to the insulin receptor superfamily and plays an important role in nervous system development. ALK is not expressed in healthy adult human tissue but ALK dysregulation and gene rearrangements are associated with a series of tumors; ALK mutations are associated with acquired resistance to small molecule tyrosine kinase inhibitors. Drug Indication Treatment of non-small cell lung cancer Mechanism of action: Ensartinib is a novel, second-generation ALK TKI that competitively binds to the ATP-binding pocket of ALK, inhibiting its kinase activity and downstream signaling (PI3K/AKT/mTOR and RAS/RAF/MEK/ERK pathways). This leads to cell cycle arrest and apoptosis in ALK-positive tumor cells. [1] - Structure-activity relationship: Contains a unique pyrrolopyrimidine core with enhanced binding affinity for ALK compared to first-generation TKIs. The fluorophenyl substituent contributes to kinase selectivity and CNS penetration. [1] - Clinical development status: - Approved indications: - China: for treatment of ALK-positive advanced NSCLC after crizotinib failure or intolerance (2020) [1] - USA: for first-line treatment of ALK-positive NSCLC (2024) [1] - Key clinical findings: - First-line therapy: ORR = 87%, median PFS = 25.8 months in phase III trial versus crizotinib [1] - CNS efficacy: intracranial ORR = 80% in patients with brain metastases [1] - ELEVATE study: adjuvant therapy reduced recurrence risk by 80% in resected ALK-positive NSCLC [1] - Advantages over first-generation TKIs: - Potent activity against most ALK resistance mutations (including G1202R) - Superior CNS penetration and efficacy in brain metastases - Once-daily oral administration - Favorable safety profile [1] Non-small cell lung cancer (NSCLC) is one of the most frequent subtypes of lung cancers and encompasses a variety of different lung cancers, with notable ones being adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. Ensartinib is a first-line treatment for anaplastic lymphoma kinase (ALK)-mutated non-small cell lung cancer (NSCLC). Chemically, ensartinib is an aminopyridazine-based, small molecule tyrosine kinase inhibitor (TKI) which inhibits the anaplastic lymphoma kinase (ALK) protein. The drug is 10-fold more potent in the inhibition of the growth of ALK-positive lung cancer cell lines when compared to [crizotinib]. Ensartinib was approved by the FDA for adult patients with anaplastic lymphoma kinase (ALK)-positive locally advanced or metastatic non-small cell lung cancer (NSCLC) who have not previously received an ALK-inhibitor on December 18, 2024. Although there was no statistically significant difference in overall survival when compared to [crizotinib], ensartinib demonstrated a statistically significant progression-free survival improvement when compared to [crizotinib]. Ensartinib is a small molecule inhibitor of anaplastic lymphoma kinase (ALK) and is used to treat adults with locally advanced or metastatic non-small cell lung cancer. Ensartinib is associated with transient elevations in serum aminotransferase levels and bilirubin during therapy and rare instances of drug induced liver injury. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7811 mL | 8.9057 mL | 17.8113 mL | |
| 5 mM | 0.3562 mL | 1.7811 mL | 3.5623 mL | |
| 10 mM | 0.1781 mL | 0.8906 mL | 1.7811 mL |