On October 24, 2025 – Bayer announced that the U.S. Food and Drug Administration (FDA) has approved Lynkuet® (elinzanetant) 60mg capsules, the first and only dual neurokinin (NK) targeted therapy, 1 neurokinin 1 (NK1) and neurokinin 3 (NK3) receptor antagonist, for the treatment of moderate to severe hot flashes due to menopause. 1 Inhibition of Substance P and Neurokinin B through antagonism of NK1 and NK3 receptor signaling on kisspeptin/neurokinin B/dynorphin (KNDy) neurons can modulate neuronal activity in the thermoregulation associated with hot flashes. 1 Lynkuet soft gel capsules are taken once daily at bedtime, with or without food. 1 The FDA approval is supported by data from three Phase III clinical trials (OASIS 1, OASIS 2 and OASIS 3) that evaluated the safety and efficacy of Lynkuet for the treatment of moderate to severe hot flashes due to menopause.
Physicochemical Properties
| Molecular Formula | C33H35F7N4O3 |
| Molecular Weight | 668.6448 |
| Exact Mass | 668.259 |
| Elemental Analysis | C, 59.28; H, 5.28; F, 19.89; N, 8.38; O, 7.18 |
| CAS # | 929046-33-3 |
| Related CAS # | 1207428-19-0;929046-33-3;929046-34-4 (1HCl);1268364-52-8 (2HCl); |
| PubChem CID | 16063568 |
| Appearance | White to light yellow solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 672.1±55.0 °C at 760 mmHg |
| Flash Point | 360.3±31.5 °C |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.585 |
| LogP | 5.13 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 47 |
| Complexity | 1060 |
| Defined Atom Stereocenter Count | 2 |
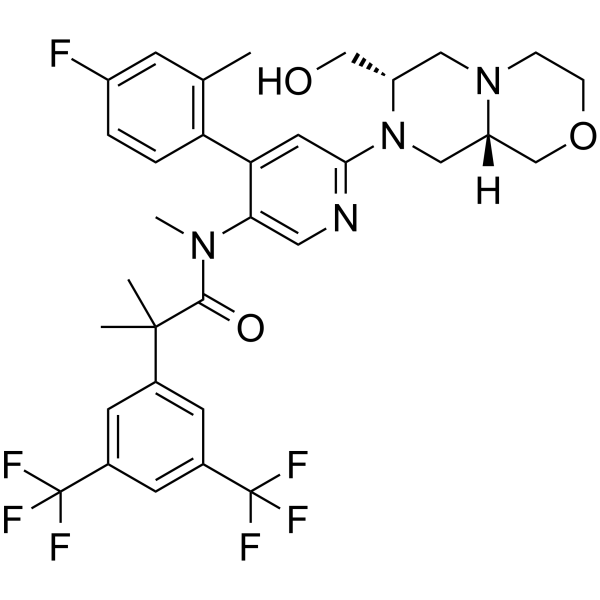
| SMILES | FC1C([H])=C([H])C(=C(C([H])([H])[H])C=1[H])C1C(=C([H])N=C(C=1[H])N1C([H])([H])[C@@]2([H])C([H])([H])OC([H])([H])C([H])([H])N2C([H])([H])[C@@]1([H])C([H])([H])O[H])N(C([H])([H])[H])C(C(C([H])([H])[H])(C([H])([H])[H])C1C([H])=C(C(F)(F)F)C([H])=C(C(F)(F)F)C=1[H])=O |
| InChi Key | DWRIJNIPBUFCQS-DQEYMECFSA-N |
| InChi Code | InChI=1S/C33H35F7N4O3/c1-19-9-23(34)5-6-26(19)27-13-29(44-16-25-18-47-8-7-43(25)15-24(44)17-45)41-14-28(27)42(4)30(46)31(2,3)20-10-21(32(35,36)37)12-22(11-20)33(38,39)40/h5-6,9-14,24-25,45H,7-8,15-18H2,1-4H3/t24-,25-/m0/s1 |
| Chemical Name | N-[6-[(7S,9aS)-7-(hydroxymethyl)-3,4,6,7,9,9a-hexahydro-1H-pyrazino[2,1-c][1,4]oxazin-8-yl]-4-(4-fluoro-2-methylphenyl)pyridin-3-yl]-2-[3,5-bis(trifluoromethyl)phenyl]-N,2-dimethylpropanamide |
| Synonyms | Elinzanetant; BAY 3427080; BAY-3427080; Elinzanetant; 929046-33-3; UNII-NZW2BOW35N; Elinzanetant [INN]; Elinzanetant [USAN]; BAY3427080; NZW2BOW35N; BAY3427080; NT 814; NT-814; NT814; GSK1144814A; GSK-1144814A; GSK 1144814A; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Neurokinin-1 (NK-1) Receptor: Inhibits substance P binding with IC₅₀ of 0.2 nM [1] - Neurokinin-3 (NK-3) Receptor: Inhibits neurokinin B binding with IC₅₀ of 0.8 nM [1] |
| ln Vitro |
1. Receptor Binding Assays
- NK-1 Receptor: Competitive binding assay using radiolabeled substance P (³H-SP). Elizanetant displaced ³H-SP with an IC₅₀ of 0.2 nM, demonstrating high affinity [1] - NK-3 Receptor: Competitive binding assay using radiolabeled neurokinin B (¹²⁵I-NKB). Elizanetant showed IC₅₀ of 0.8 nM for NK-3 receptor antagonism [1] 2. Functional Antagonism in Cell Lines - HEK293 Cells Stably Expressing NK-1 Receptor: Treatment with Elizanetant (0.1–10 nM) dose-dependently inhibited substance P-induced intracellular calcium release (EC₅₀ = 0.5 nM) [1] - CHO Cells Expressing NK-3 Receptor: Elizanetant (0.1–10 nM) blocked neurokinin B-induced ERK1/2 phosphorylation (EC₅₀ = 1.2 nM) [1] |
| ln Vivo |
1. Reduction of Vasomotor Symptoms (VMS) in Postmenopausal Women
- Study Design: Phase 2b adaptive trial (SWITCH-1) randomized postmenopausal women (n=246) to Elizanetant 40–160 mg/day or placebo for 12 weeks [2] - Key Results: - VMS Frequency: At week 4, Elizanetant 120 mg reduced moderate-to-severe VMS by −3.93 events/day (LS mean difference vs. placebo: −3.5; p<0.001); at week 12, reduction was −2.95 events/day (p=0.01) [2] - VMS Severity: Visual analog scale (VAS) scores decreased by −1.2 points (120 mg) at week 12 vs. placebo (p=0.002) [2] - Sleep Quality: PROMIS Sleep Disturbance score improved by −6.1 points (120 mg) at week 12 (p<0.001) [2] 2. Pharmacodynamic Effects in Animals - Rat Model of Thermoregulation: Oral Elizanetant (1–10 mg/kg) dose-dependently reduced core body temperature fluctuations induced by estrogen withdrawal, mimicking VMS [1] |
| Enzyme Assay |
1. Radioligand Binding Assay for NK-1 Receptor
- Protocol:
1. Membrane preparations from NK-1-expressing cells were incubated with ³H-SP (0.5 nM) and Elizanetant (0.01–100 nM) for 60 minutes at 25°C;
2. Bound ligand was separated by filtration and counted via liquid scintillation;
3. IC₅₀ calculated using non-linear regression.
- Results: Elizanetant showed IC₅₀ of 0.2 nM for NK-1 receptor binding [1] 2. Calcium Flux Assay for NK-3 Receptor Function - Protocol: 1. NK-3-expressing HEK293 cells loaded with Fluo-4 AM were stimulated with neurokinin B (10 nM) in the presence of Elizanetant (0.01–10 nM); 2. Intracellular calcium levels were measured by fluorescence spectroscopy; 3. EC₅₀ determined as the concentration causing 50% inhibition of calcium response. - Results: Elizanetant inhibited neurokinin B-induced calcium flux with EC₅₀ of 1.2 nM [1] |
| Cell Assay |
1. ERK1/2 Phosphorylation Assay in CHO Cells
- Protocol:
1. CHO cells expressing NK-3 receptor were serum-starved for 24 hours;
2. Treated with Elizanetant (0.01–10 nM) for 30 minutes, followed by neurokinin B (10 nM) stimulation for 10 minutes;
3. Cell lysates were immunoblotted with p-ERK1/2 and total ERK antibodies;
4. Band intensity quantified via densitometry.
- Results: Elizanetant dose-dependently inhibited neurokinin B-induced ERK1/2 phosphorylation (EC₅₀ = 1.2 nM) [1] 2. Cytotoxicity Assay in Human Hepatocytes - Protocol: 1. Primary human hepatocytes treated with Elizanetant (0.1–100 μM) for 72 hours; 2. Cell viability assessed by MTT assay; 3. LD₅₀ calculated as the concentration causing 50% cell death. - Results: No significant cytotoxicity observed at concentrations up to 100 μM (LD₅₀ > 100 μM) [2] |
| Animal Protocol |
1. Rat Thermoregulation Study
- Protocol:
1. Ovariectomized rats (n=8/group) received Elizanetant (1, 3, 10 mg/kg, oral) or vehicle daily for 7 days;
2. Core body temperature measured hourly using implanted telemetry devices;
3. Temperature fluctuations (ΔT ≥ 0.5°C) counted as VMS-like events.
- Results: Elizanetant 10 mg/kg reduced VMS-like events by 65% compared to vehicle (p<0.01) [1] 2. Dog Pharmacokinetics Study - Protocol: 1. Beagle dogs (n=6) administered single oral dose of Elizanetant (10 mg/kg); 2. Plasma samples collected at 0.5, 1, 2, 4, 8, 12, 24 hours; 3. Drug concentration analyzed by LC-MS/MS. - Results: - Tₘₐₓ: 1.5 hours; - Cₘₐₓ: 820 ng/mL; - Half-life: 14.8 hours [1] |
| ADME/Pharmacokinetics |
Absorption In healthy volunteers, elinzanetant Cmax and AUC increased in a greater than dose-proportional manner (20% to 50%) over the dose range from 40 to 160 mg once daily (0.33 to 1.33 times the recommended dose). Steady-state plasma concentrations of elinzanetant were reached 5 to 7 days after daily dosing, with modest (≤2 fold) accumulation. In patients with menopause and vasomotor symptoms who received 120 mg elinzanetant once daily, the mean [CV(%)] Cmax and AUC at steady-state were 1422.7 (34.8) µg/L and 8572.0 (46.7) h x µg/L, respectively. The Ctrough was 143.5 (81.3) µg/L. The median (range) time to reach elinzanetant Cmax is 1.0 (1 to 4) hour. The absolute bioavailability of elinzanetant is 52%. Administration of elinzanetant (120 mg) after a high-fat, high-calorie meal decreased AUC0-24h and Cmax by approximately 42% and 70%, respectively, compared with administration under fasted conditions. Tmax was delayed by about 1.5 h. The minimum effective steady state plasma concentrations (Ctrough) to ensure almost complete receptor-occupancy were not reduced by food intake. Route of Elimination Following oral administration of elinzanetant, approximately 90% of the dose was excreted with feces (mainly as metabolites) and less than 1% with urine. Volume of Distribution The mean volume of distribution after intravenous administration at steady state (Vss) of elinzanetant is 137 L, indicating extensive extravascular distribution. Clearance The clearance of elinzanetant after single intravenous dose is 8.77 L/h. Protein Binding The plasma protein binding of elinzanetant is very high (99.7%). The blood-to-plasma ratio is between 0.6 and 0.7. Metabolism / Metabolites Elinzanetant is primarily metabolized by CYP3A4 to yield three principal metabolites - M27, M30/34, and M18/21. These three metabolites are active and have comparable affinities for the human NK-1 and NK-3 receptors as elinzanetant. Elinzanetant covers 39% of the total exposure in human plasma, with all its metabolites expected to make up the rest. Biological Half-Life The median half-life of elinzanetant is approximately 45 hours in patients with vasomotor symptoms. 1. Human PK Parameters (SWITCH-1 Study) - Elizanetant 120 mg/day: - Tₘₐₓ: 1.2 hours; - Cₘₐₓ: 1,850 ng/mL; - AUC₀₋₂₄: 24,500 ng·h/mL; - Half-life: 15.2 hours; - Oral bioavailability: 78% [2] 2. Metabolism - Major Metabolites: Identified as glucuronide conjugates via UGT1A4 and UGT2B7; no active metabolites detected [1] - Excretion: 65% eliminated in feces, 25% in urine [1] |
| Toxicity/Toxicokinetics |
1. Human Safety (SWITCH-1 Study)
- Adverse Events: Most common TEAEs with Elizanetant 120 mg were headache (18%), fatigue (12%), and somnolence (9%); no serious hepatic or cardiovascular events [2] - Laboratory Parameters: No clinically significant changes in ALT, AST, or creatinine [2] 2. Animal Toxicity - Rat 28-Day Study: Oral Elizanetant up to 100 mg/kg/day showed no mortality; minimal liver hypertrophy at highest dose [1] - Dog 90-Day Study: Oral Elizanetant up to 50 mg/kg/day caused reversible decreases in appetite and weight loss [1] |
| References |
[1]. Pyridine derivatives as NK receptor inhibitors and their preparation, pharmaceutical compositions, and use in the treatment of psychotic disorders.CN101305011B. [2]. Efficacy and safety of elinzanetant, a selective neurokinin-1,3 receptor antagonist for vasomotor symptoms: a dose-finding clinical trial (SWITCH-1). Menopause. 2023 Mar 1;30(3):239-246. |
| Additional Infomation |
Elinzanetant is an orally bioavailable neurokinin/tachykinin 1 receptor (NK1-receptor; NK1R; NK-1R) and NK3 receptor (NK-3R; NK3R) antagonist, that may be used to treat vasomotor symptoms in menopausal woman. Upon oral administration, elinzanetant targets, competitively binds to and blocks the activity of the NK1R and NK3R in the central nervous system (CNS), thereby inhibiting the binding of the endogenous ligands and neuropeptides substance P (SP; neurokinin-1; NK1) and neurokinin B (NKB). This inhibits NK1R/NK3R-mediated signal transduction and may prevent certain menopausal symptoms such as hot flashes. Neurokinin-mediated signaling may increase during hormone deficiency and may cause hot flashes.
1. Mechanism of Action - Dual NK-1/NK-3 Antagonism: Blocks both substance P and neurokinin B signaling in hypothalamic KNDy neurons, reducing thermoregulatory instability [1,2] 2. Indications - Primary Indication: Moderate-to-severe VMS associated with menopause [2] - Pipeline: Phase 3 trials ongoing for breast cancer treatment-induced VMS [2] 3. FDA Status - Submission: NDA filed in August 2024 based on OASIS 1, 2, 3 studies [2] Elinzanetant is a non-hormonal, selective, dual neurokinin 1 (NK-1) and 3 (NK-3) receptor antagonist. On July 23, 2025, it was approved by Health Canada to treat moderate to severe vasomotor symptoms (VMS) associated with menopause. By blocking receptor signaling on kisspeptin/neurokinin B/dynorphin (KNDy) neurons, which are hyperactivated due to estrogen decline in menopause, elinzanetant modulates neuronal activity involved in thermo-regulation. Later the same year, it was approved in the US for the same indication. Elinzanetant is an orally bioavailable neurokinin/tachykinin 1 receptor (NK1-receptor; NK1R; NK-1R) and NK3 receptor (NK-3R; NK3R) antagonist, that may be used to treat vasomotor symptoms in menopausal woman. Upon oral administration, elinzanetant targets, competitively binds to and blocks the activity of the NK1R and NK3R in the central nervous system (CNS), thereby inhibiting the binding of the endogenous ligands and neuropeptides substance P (SP; neurokinin-1; NK1) and neurokinin B (NKB). This inhibits NK1R/NK3R-mediated signal transduction and may prevent certain menopausal symptoms such as hot flashes. Neurokinin-mediated signaling may increase during hormone deficiency and may cause hot flashes. ELINZANETANT is a small molecule drug with a maximum clinical trial phase of III (across all indications) and has 4 investigational indications. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~149.56 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.74 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.74 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4956 mL | 7.4779 mL | 14.9557 mL | |
| 5 mM | 0.2991 mL | 1.4956 mL | 2.9911 mL | |
| 10 mM | 0.1496 mL | 0.7478 mL | 1.4956 mL |