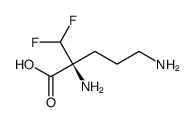
Eflornithine (DFMO; α-difluoromethylornithine; MDL-71782; RMI-71782; trade names: Vaniqa among others) is an approved medication used to treat African trypanosomiasis and excessive hair growth on the face in women. Specifically, it is used for the 2nd stage of sleeping sickness caused by T. b. gambiense and may be used in combination with nifurtimox.
Physicochemical Properties
| Molecular Formula | C6H12N2O2F2 |
| Molecular Weight | 182.16848 |
| Exact Mass | 182.087 |
| CAS # | 67037-37-0 |
| Related CAS # | Eflornithine;70052-12-9;L-Eflornithine monohydrochloride;69955-42-6;Eflornithine hydrochloride;68278-23-9;L-Eflornithine;66640-93-5; 96020-91-6 (HCl hydrate) 68278-23-9 (HCl); 70050-55-4 (R-isomer); 66640-93-5 (L-isomer); 69955-42-6 (S-isomer); |
| PubChem CID | 3009 |
| Appearance | White to light yellow solid powder |
| Density | 1.293g/cm3 |
| Boiling Point | 347ºC at 760 mmHg |
| Flash Point | 163.7ºC |
| Index of Refraction | 1.462 |
| LogP | 1.173 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 12 |
| Complexity | 166 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | VLCYCQAOQCDTCN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H12F2N2O2/c7-4(8)6(10,5(11)12)2-1-3-9/h4H,1-3,9-10H2,(H,11,12) |
| Chemical Name | 2,5-diamino-2-(difluoromethyl)pentanoic acid |
| Synonyms | EFLORNITHINE; 70052-12-9; dfmo; Difluoromethylornithine; Ornidyl; 2-(Difluoromethyl)ornithine; 2,5-diamino-2-(difluoromethyl)pentanoic acid; ...; 67037-37-0; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Ornithine decarboxylase |
| ln Vitro |
Eflornithine is a specific, irreversible inhibitor of the enzyme ornithine decarboxylase which is thought to slow hair growth by inhibiting this enzyme in hair follicles[2]. In vitro permeation study of eflornithine was performed using Franz diffusion cell. The hair growth inhibitory activity of eflornithine was significantly enhanced when the eflornithine cream was applied onto a mouse skin area pretreated with microneedles, most likely because the micropores created by microneedles allowed the permeation of eflornithine into the skin, as confirmed in an in vitro permeation study. Immunohistochemistry data revealed that cell proliferation in the skin and hair follicles was also significantly inhibited when the eflornithine cream was applied onto a skin area pretreated with microneedles.[3] |
| ln Vivo | The only novel drug approved in the last fifty years for the treatment of human African trypanosomiasis is eflornithine. It is mostly used as a stand-by medication for Trypanosoma brucei gambiense infections that do not respond to melarsoprol [1]. When it came to decreasing the growth of facial hair in participants with excess hair, eflornithine 15% cream outperformed a placebo. Following a 24-week course of treatment, face hirsutism improved at least somewhat in 58% of eflornithine patients and 34% of placebo subjects [2]. When eflornithine cream was administered to mouse skin areas that had been microneedled beforehand, the hair growth inhibitory activity of eflornithine was markedly increased [3]. After 14 days of hypertension, eflornithine treatment of constrictive hypertensive rats led to the normalization of KCI and norepinephrine contractile strength as well as the relaxing of acetylcholine [4]. |
| Enzyme Assay |
In in vitro studies using Salmonella and two strains of Saccharomyces, eflornithine did not induce mutagenic changes [1]. In vitro permeation of eflornithine hydrochloride through mouse skin[3] In vitro permeation assay using Franz diffusion cell apparatus was completed as previously described (Kumar et al. 2012; Kumar et al. 2011; Naguib, Kumar, & Cui 2014) using the lower dorsal skin of C57BL/6 mice. Hair was trimmed using an electric clipper 24 h before the collection of the skin. Skin was harvested, wrapped in aluminum foil, and stored at −20°C for a maximum period of one month and used whenever needed. Freezing of the skin at −20°C (without a cryo-protectant) is commonly applied in literature, and such skin samples have been used frequently for permeability studies (Stahl, Wohlert, & Kietzmann 2012). Dennerlein et al. showed that freezing and storing of freshly excised human skin for up to 30 days at −20°C does not affect the skin permeability (Dennerlein et al. 2013). Other researchers showed that when human skin was wrapped in aluminum foil and stored at −26°C, the skin retained its barrier properties for up to 6 months (Badran, Kuntsche, & Fahr 2009). After the fat layer was removed, the skin was mounted onto the Franz diffusion cells with dorsal side facing upward. The receiver compartment contained 5 ml of water and was maintained at 37°C with a Haake SC 100 Water Circulator (ThermoScientific, Wellington, NH). The hair-trimmed skin was treated with a Dermaroller® microneedle roller as previously described before it was mounted onto the Franz diffusion cells (Kumar et al. 2011; Naguib, Kumar, & Cui 2014). The skin sample was placed onto the flat surface of a balance, and the microneedle roller was rolled in four perpendicular directions over the skin surface, 5 times each for a total of 20 times, with an applying pressure of 350–400 g, which was constantly measured using the balance while the roller was rolled. The diffusion area of the skin was 0.64 cm2. The donor compartment was loaded with 4 mg of eflornithine hydrochloride in 500 μl water and covered with parafilm to prevent evaporation. After 0, 1, 3, 6, 8, and 24 h, samples (150 μl) were withdrawn from the receiver compartment and immediately replenished with fresh medium. The samples were analyzed using HPLC following a method described previously with modifications (Saravanan et al. 2009). Chromatographic analysis was carried out with an Agilent 1260 Infinity HPLC station equipped with ZORBAX Eclipse Plus C18 (5 μm, 4.6 × 150 mm) column using a acetonitrile-buffer mixture (70%:30%, v/v) as the mobile phase. The buffer was prepared by dissolving 0.68 g of potassium phosphate monobasic in 1 l of water. The flow rate was 0.8 ml/min. The detector wavelength was 210 nm. |
| Cell Assay | Skin tissues were fixed with a buffered formalin (10%) solution for 24 h, washed with 0.1 M of sodium phosphate buffer (pH 7.4), dehydrated in graded ethanol, embedded in paraffin, and sectioned vertically. The sections were stained using hematoxylin-eosin (H&E) or an antibody against 5-bromo-2′-deoxyuridine (BrdU) in the Histology and Tissue Processing facility in the Dell Pediatric Research Institute at the University of Texas at Austin. Mice were injected intraperitoneally with BrdU in phosphate buffered saline (PBS, pH 7.4, 10 mM) at the dose of 100 μg/g body weight, 30 min prior to euthanasia. All skin sections were examined under an Olympus BX53 microscope [3]. |
| Animal Protocol |
In vivo efficacy study was performed in a mouse model by monitoring the re-growth of hair in the lower dorsal skin of mice after the eflornithine cream was applied onto an area pretreated with microneedles. The skin and the hair follicles in the treated area were also examined histologically[3]. Female C57BL/6 mice (8–10 weeks old) were are ideal for examining the physiological actions during different hair cycle phases due to the occurrence of naturally synchronized hair cycles with cyclic pigmentation (Slominski, Paus, & Costantino 1991). Each experimental group was composed of 3–4 mice. Hair in the lower dorsal skin of anesthetized mice was either trimmed using an electric clipper, plucked using GiGi® Honee warm wax as previously described (Xiao et al. 2012), or chemically removed using Nair® lotion. The skin area where the hair was removed was then treated with the eflornithine hydrochloride 13.9% cream (~50 mg per mouse per treatment) using a spatula two times a day in an interval of at least 8 h for a maximum period of 36 days. A group of mice whose hair in the application site was trimmed using a clipper were also treated with the microneedle roller every time before the application of eflornithine cream as previously described (Kumar et al. 2012). Briefly, mice were placed onto the flat surface of a balance, and the microneedle roller was rolled over the marked skin surface, 10 times parallel to mouse length, with an applying pressure of 350–400 g as indicated on the balance. In control groups, the hair in mouse dorsal skin was removed by trimming, plucking, or chemical depilation with Nair®, but the area was not treated with the eflornithine cream. The hair re-growth was evaluated by taking digital photographs of the mouse skin areas for a maximum period of 36 days after the first application of the eflornithine cream. On the last day of the study, animals were euthanized, and skin samples were collected from the treated areas for immunohistochemical studies.[3] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Following oral administrations of eflornithine, peak plasma concentrations of eflornithine (Cmax) were achieved (Tmax) 3.5 hours post-dosing. The Cmax and AUC (area under the concentration-time curve) of eflornithine were not affected by food (high fat and high calories). Administration of crushed tablets in a standard pudding admixture had no effect on eflornithine exposure (Cmax and AUC6h). The mean percutaneous absorption of eflornithine in women with unwanted facial hair, from a 13.9% w/w cream formulation, is < 1% of the radioactive dose, following either single or multiple doses under conditions of clinical use, that included shaving within 2 hours before radiolabeled dose application in addition to other forms of cutting or plucking and tweezing to remove facial hair. Steady state was reached within four days of twice-daily application. Following twice-daily application of 0.5 g of the cream (total dose 1.0 g/day; 139 mg as anhydrous eflornithine hydrochloride), under conditions of clinical use in women with unwanted facial hair (n=10), the steady-state Cmax, Ctrough and AUC12hr were approximately 10 ng/mL, 5 ng/mL, and 92 ng hr/mL, respectively, expressed in terms of the anhydrous free base of eflornithine hydrochloride. At steady state, the dose-normalized peak concentrations (Cmax) and the extent of daily systemic exposure (AUC) of eflornithine following twice-daily application of 0.5 g of the cream (total dose 1.0 g/day) is estimated to be approximately 100- and 60-fold lower, respectively, when compared to 370 mg/day once-daily oral doses. This compound is not known to be metabolized and is primarily excreted unchanged in the urine. Eflornithine volume of distribution (Vz/F) is 24.3 L. The clearance (CL/F) of eflornithine is 5.3 L/h. The mean percutaneous absorption of eflornithine in women with unwanted facial hair, from a 13.9% w/w cream formulation, is < 1% of the radioactive dose, following either single or multiple doses under conditions of clinical use, that included shaving within 2 hr before radiolabeled dose application in addition to other forms of cutting or plucking and tweezing to remove facial hair. Following twice daily application of 0.5 g of the cream (total dose 1.0 g/day; 139 mg as anhydrous eflornithine hydrochloride), under conditions of clinical use in women with unwanted facial hair (n=10), the steady-state Cmax, Ctrough and AUC12hr were approximately 10 ng/mL, 5 ng/mL, and 92 nghr/mL, respectively, expressed in terms of the anhydrous free base of eflornithine hydrochloride. At steady state, the dose-normalized peak concentrations (Cmax) and the extent of daily systemic exposure (AUC) of eflornithine following twice-daily application of 0.5 g of the cream (total dose 1.0 g/day) is estimated to be approximately 100- and 60-fold lower, respectively, when compared to 370 mg/day once-daily oral doses. Eflornithine is not metabolized and is excreted unchanged in urine. For more Absorption, Distribution and Excretion (Complete) data for Eflornithine (8 total), please visit the HSDB record page. Metabolism / Metabolites This compound is not known to be metabolized and is primarily excreted unchanged in the urine. Biological Half-Life The terminal plasma elimination half-life of eflornithine was 3.5 hours, and the apparent steady-state plasma half-life of eflornithine was approximately 8 hours. The apparent steady-state plasma t1/2 of eflornithine was approximately 8 hours. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Maternal intravenous eflornithine 400 mg/kg daily for 7 days did not cause any adverse serious effects in breastfed infants. After topical application, eflornithine is poorly absorbed so it is not likely to reach the bloodstream of the infant or cause any adverse effects in breastfed infants. ◉ Effects in Breastfed Infants A cohort of 33 infants who were breastfed (extent not stated) by hospitalized mothers taking nifurtimox was followed in the Democratic Republic of the Congo. Thirty mothers took a full course of 30 doses of oral nifurtimox 15 mg/kg daily and all received 14 doses of intravenous eflornithine 400 mg/kg daily for 7 days for human African trypanosomiasis. (sleeping sickness). Nursing mothers also took a median of 4 other concomitant medications, including amoxicillin, ciprofloxacin, metronidazole, trimethoprim-sulfamethoxazole, aspirin, and diclofenac (1 patient each); hydrocortisone, promethazine and quinine (2 patients each); levamisole (6 patients); sulfadoxine-pyrimethamine (8 patients); dipyrone (13 patients); acetaminophen (16 patients); and mebendazole (17 patients). No serious adverse events were reported in any of the breastfed infants. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Eflornithine does not specifically bind to human plasma proteins. |
| References |
[1]. Eflornithine for the treatment of human African trypanosomiasis. Parasitol Res. 2003 Jun;90 Supp 1:S49-52. [2]. Topical eflornithine. Am J Clin Dermatol. 2001;2(3):197-201; discussion 202. [3]. A method to improve the efficacy of topical eflornithine hydrochloride cream. Drug Deliv. 2016 Jun;23(5):1495-501. [4]. Eflornithine alters changes in vascular responsiveness associated with coarctation hypertension. Clin Exp Hypertens. 1997 Apr;19(3):297-312. |
| Additional Infomation |
Therapeutic Uses Antineoplastic Agents; Enzyme Inhibitors; Trypanocidal Agents Eflornithine is indicated for the reduction of unwanted facial hair in women. Eflornithine has only been studied on the face and adjacent involved areas under the chin of affected individuals. Usage should be limited to these areas of involvement. /Included in US product label/ /EXPERIMENTAL THERAPY:/ D,L-alpha-difluoromethylornithine (DFMO) was synthesized over 20 years ago. It was hoped that this enzyme-activated, irreversible inhibitor of ornithine decarboxylase, the first enzyme in polyamine synthesis, would be effective as a chemotherapy for hyperproliferative diseases, including cancer and/or infectious processes. DFMO was generally found to exert cytostatic effects on mammalian cells and tissues, and its effectiveness as a therapeutic agent has been modest. DFMO was also found to cause treatment-limiting (but reversible) ototoxicity at high doses. This side effect, along with its minimal therapeutic activity, contributed to the loss of interest by many clinicians in further developing DFMO as a cancer therapeutic agent. However, DFMO was subsequently shown to inhibit carcinogen-induced cancer development in a number of rodent models, and interest in developing this compound as a preventive agent has increased. The rationale for the inhibition of ornithine decarboxylase as a cancer chemopreventive agent has been strengthened in recent years because this enzyme has been shown to be transactivated by the c-myc oncogene in certain cell/tissue types and to cooperate with the ras oncogene in malignant transformation of epithelial tissues. Recent clinical cancer chemoprevention trials, using dose de-escalation designs, indicate that DFMO can be given over long periods of time at low doses that suppress polyamine contents in gastrointestinal and other epithelial tissues but cause no detectable hearing loss or other side effects. Current clinical chemoprevention trials are investigating the efficacy of DFMO to suppress surrogate end point biomarkers (e.g., colon polyp recurrence) of carcinogenesis in patient populations at elevated risk for the development of specific epithelial cancers, including colon, esophageal, breast, cutaneous, and prostate malignancies. The usual first-line treatment for Trypanosoma brucei gambiense sleeping sickness is melarsoprol, but when that fails the outlook has hitherto been grim. The polyamine synthesis inhibitor eflornithine (difluoromethylornithine, DFMO) has emerged as an alternative therapy. 207 patients with late-stage T b gambiense sleeping sickness were treated in rural Zaire with three different regimens of DFMO in an open-trial design. During treatment, trypanosomes disappeared from the CSF of all 87 patients in whom parasites had been seen before DFMO administration, and there was a sharp fall in CSF white cell count from a mean of 186/microliters to 21/microliters. 152 patients have been followed for at least a year after DFMO treatment, and only 13 (9%) have relapsed. Treatment failures were more common in children less than 12 years, among patients treated with oral DFMO only, and among patients who received DFMO as the initial treatment of their recently diagnosed trypanosomiasis. Toxicity was acceptable. Only 4 patients died during or shortly after treatment. Bone marrow suppression resulting in anaemia (43%) or leucopenia (53%) was common but bore little consequence. This open trial shows that DFMO is as active as and possibly less toxic than melarsoprol. For economic and logistic reasons DFMO may not be the first-choice therapy in rural Africa but for the vast majority of patients who relapse after melarsoprol DFMO will be curative. ... A polyamine biosynthesis inhibitor, eflornithine (chemical name, DL-alpha-difluoromethylornithine, supplied as monohydrochloride monohydrate), was used to treat a 3 1/2-year-old child with newly diagnosed severe trypanosomiasis that had been acquired more than two years previously in Zaire or the Congo. Treatment consisted of 300 to 400 mg/kg/d of eflornithine by continuous intravenous infusion for 25 days followed by 300 mg/kg/d of eflornithine by mouth divided in four equal doses daily for 17 days. The child's recovery was dramatic, with eradication of blood and cerebrospinal fluid parasites in the first week. Cerebrospinal fluid pleocytosis resolved completely. Her generalized adenopathy and fever gradually resolved. Severe ataxia, inability to walk or to change posture on her own, marked language regression, and lethargy all improved during and after her therapy. The drug was well tolerated; the only noted adverse effect was transient thrombocytopenia during the fourth week of therapy. Eflornithine was a safe and effective agent for treatment of trypanosomiasis with central nervous system involvement in this child. Drug Warnings Contraindications /include/ known hypersensitivity to eflornithine or any ingredient in the formulation. Eflornithine hydrochloride topical cream should be discontinued if hypersensitivity reactions occur. Transient stinging or burning may occur if applied to abraded or broken skin. Skin irritation may occur in susceptible patients or in those using higher than recommended dosages. No laboratory test abnormalities have been consistently found to be associated with eflornithine. In an open labeled study, some patients showed an increase in their transaminases; however, the clinical significance of these findings is not known. For more Drug Warnings (Complete) data for Eflornithine (11 total), please visit the HSDB record page. Pharmacodynamics Inhibition of polyamine synthesis by eflornithine restored the balance of the LIN28/Let-7 metabolic pathway, which is involved in the regulation of cancer stem cells and glycolytic metabolism, by decreasing expression of the oncogenic drivers MYCN and LIN28B in MYCN-amplified neuroblastoma. In vitro, eflornithine induced senescence and suppressed neurosphere formation in MYCN-amplified and MYCN non-amplified neuroblastoma cells, indicating a cytostatic effect. Treatment with eflornithine prevented or delayed tumor formation in mice injected with limiting dilutions of MYCN-amplified neuroblastoma cells. Additionally, polyamines are also involved in keratin synthesis, and inhibition of polyamines can decrease the proliferation of hair matrix cells and thus inhibit the anagen phase of hair production. |
Solubility Data
| Solubility (In Vitro) | Typically soluble in DMSO (e.g. 10 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.4894 mL | 27.4469 mL | 54.8938 mL | |
| 5 mM | 1.0979 mL | 5.4894 mL | 10.9788 mL | |
| 10 mM | 0.5489 mL | 2.7447 mL | 5.4894 mL |