Physicochemical Properties
| Molecular Formula | C22H22FN7O2 |
| Molecular Weight | 435.454186916351 |
| Exact Mass | 435.181 |
| Elemental Analysis | C, 60.68; H, 5.09; F, 4.36; N, 22.52; O, 7.35 |
| CAS # | 2408839-73-4 |
| PubChem CID | 147817927 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 1.8 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 32 |
| Complexity | 775 |
| Defined Atom Stereocenter Count | 2 |
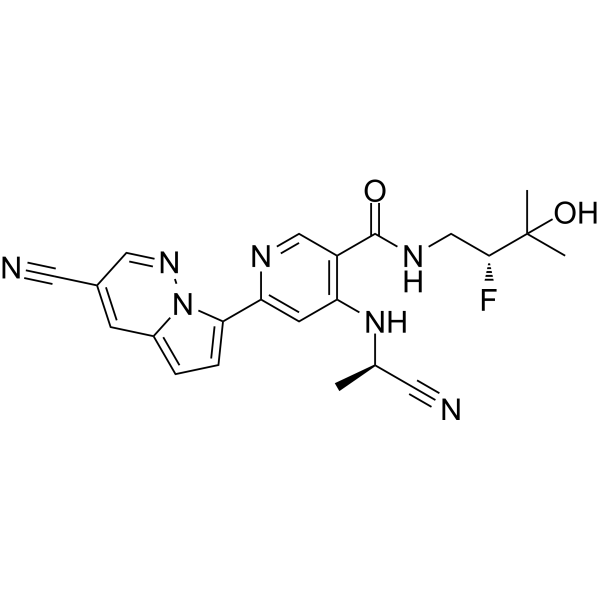
| SMILES | C1=NC(C2N3C(=CC=2)C=C(C#N)C=N3)=CC(N[C@@H](C#N)C)=C1C(NC[C@@H](F)C(O)(C)C)=O |
| InChi Key | HORBHQPSWJRDSV-ZUOKHONESA-N |
| InChi Code | InChI=1S/C22H22FN7O2/c1-13(8-24)29-17-7-18(19-5-4-15-6-14(9-25)10-28-30(15)19)26-11-16(17)21(31)27-12-20(23)22(2,3)32/h4-7,10-11,13,20,32H,12H2,1-3H3,(H,26,29)(H,27,31)/t13-,20-/m1/s1 |
| Chemical Name | 4-[[(1R)-1-cyanoethyl]amino]-6-(3-cyanopyrrolo[1,2-b]pyridazin-7-yl)-N-[(2R)-2-fluoro-3-hydroxy-3-methylbutyl]pyridine-3-carboxamide |
| Synonyms | Edecesertib; 3NUJ7N1277; 4-[[(1R)-1-cyanoethyl]amino]-6-(3-cyanopyrrolo[1,2-b]pyridazin-7-yl)-N-[(2R)-2-fluoro-3-hydroxy-3-methylbutyl]pyridine-3-carboxamide; 4-(((1R)-1-cyanoethyl)amino)-6-(3-cyanopyrrolo(1,2-b)pyridazin-7-yl)-N-((2R)-2-fluoro-3-hydroxy-3-methylbutyl)pyridine-3-carboxamide; 2408839-73-4; Edecesertib [INN]; UNII-3NUJ7N1277; 3-Pyridinecarboxamide, 4-(((1R)-1-cyanoethyl)amino)-6-(3-cyanopyrrolo(1,2-b)pyridazin-7-yl)-N-((2R)-2-fluoro-3-hydroxy-3-methylbutyl)-; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Interleukin-1 receptor-associated kinase 4 (IRAK4): Selective inhibitor with an IC50 of 0.1 μM in kinase assays [1]. IRAK4 (Interleukin-1 receptor-associated kinase 4) (IC₅₀ = 0.6 nM in biochemical assay; EC₅₀ = 15 nM in cell-based assay) [1,2] |
| ln Vitro |
- Potent IRAK4 Inhibition: Edecesertib (GS-5718) demonstrated IC₅₀ = 0.6 nM in IRAK4 kinase activity assay and EC₅₀ = 15 nM in human whole blood cell assay, potently blocking TNF-α production [1,2] - Selectivity Profile: At 1000 nM, Edecesertib showed >500-fold selectivity against 72 kinases except IRAK1 (176-fold selectivity), with IRAK1 IC₅₀ = 70 nM [1] - Anti-Inflammatory Activity: In LPS-stimulated THP-1 macrophages, Edecesertib (10 μM) reduced IL-6 and IL-1β secretion by 82% and 75%, respectively, via suppression of NF-κB pathway activation [1] |
| ln Vivo |
- Efficacy in Lupus Model: In NZB/W F1 mice (a lupus model), Edecesertib (30 mg/kg, p.o.) administered daily for 28 days reduced anti-dsDNA antibody levels by 45% and improved renal histopathology [1] - Neuroinflammation Reduction: In a mouse model of experimental autoimmune encephalomyelitis (EAE), Edecesertib (20 mg/kg, BID) significantly decreased spinal cord inflammation and demyelination, with efficacy comparable to fingolimod (2.5 mg/kg) [1] - Safety in Non-Human Primates: In cynomolgus monkeys, Edecesertib (200 mg/kg, p.o.) for 7 days showed no drug-related adverse effects, and no QT interval prolongation was observed [1] |
| Enzyme Assay |
- IRAK4 Kinase Activity Assay: Recombinant IRAK4 kinase domain was incubated with ATP and a biotinylated peptide substrate in the presence of Edecesertib (0.01–1000 nM). Phosphorylation was detected via streptavidin-HRP, and IC₅₀ = 0.6 nM was determined [1] - Kinase Selectivity Panel: Edecesertib (1000 nM) was tested against 72 kinases using a fluorescence polarization-based assay. Selectivity ratios were calculated by comparing IRAK4 IC₅₀ with other kinases’ IC₅₀ values [1] |
| Cell Assay |
- Human Whole Blood Assay: Heparinized human blood was treated with Edecesertib (0.1–1000 nM) and stimulated with LPS (1 μg/mL). TNF-α levels in plasma were measured by ELISA, yielding EC₅₀ = 124 nM [1] - THP-1 Macrophage Assay: Cells (1×10⁶ cells/mL) were pretreated with Edecesertib (1–10 μM) for 1 hour, followed by LPS stimulation. Supernatants were analyzed for IL-6/IL-1β by ELISA, showing dose-dependent inhibition [1] |
| Animal Protocol |
- NZB/W F1 Mouse Model: Mice (8-week-old) received Edecesertib (30 mg/kg) dissolved in 0.5% CMC-Na via oral gavage daily for 28 days. Serum anti-dsDNA antibodies and renal histology were evaluated [1] - EAE Rat Model: Rats were immunized with MOG35-55 peptide and treated with Edecesertib (20 mg/kg, BID, p.o.) starting on day 0. Clinical scores and spinal cord inflammation were assessed at day 28 [1] - Cynomolgus Monkey Toxicity Study: Animals received Edecesertib (50–200 mg/kg) in capsules daily for 7 days. Safety parameters included ECG monitoring, hematology, and clinical chemistry [1] |
| ADME/Pharmacokinetics |
- Oral Bioavailability: In rats, Edecesertib showed F = 34% after oral administration (10 mg/kg), with Cmax = 1.2 μg/mL at 1.5 hours [1] - Half-Life: Plasma terminal half-life (t₁/₂) was 3.2 hours in rats and 4.8 hours in cynomolgus monkeys [1] - Tissue Distribution: Highest concentrations were observed in kidney (5.8 μg/g) and spleen (4.1 μg/g) 2 hours post-dose in rats [1] - Plasma Protein Binding: Moderate binding (free fraction = 4.4%) in human plasma [1] |
| Toxicity/Toxicokinetics |
- Preclinical Safety: In rat (14-day, 300 mg/kg) and cynomolgus monkey (7-day, 200 mg/kg) studies, no drug-related mortality or organ toxicity was observed. No QT interval prolongation was detected in telemetry-monitored dogs [1] - hERG Safety: Edecesertib showed IC₅₀ > 30 μM in hERG channel assay, indicating low cardiac arrhythmia risk [1] - Human Phase 1 Tolerability: In healthy subjects, single ascending doses up to 400 mg and multiple doses up to 200 mg QD were well-tolerated, with no serious adverse events [4] |
| References |
[1]. Emerging interleukin-1 receptor-associated kinase 4 (IRAK4) inhibitors or degraders as therapeutic agents for autoimmune diseases and cancer. Acta Pharmaceutica Sinica B, 2024 Dec;14(12):5091-5105. . [2]. POS0673 TOLEROGENIC DENDRITIC CELLS IN RHEUMATOID ARTHRITIS PATIENTS: NEWS AND PROMISES. 2021. [3]. Targeting TLR Signaling Cascades in Systemic Lupus Erythematosus and Rheumatoid Arthritis: An Update. Biomedicines. 2024 Jan 9;12(1):138. [4]. Phase 1 Study Results of GS-5718, an Oral IRAK4-Inhibitor: Pharmacodynamics of Single and Multiple Doses of GS-5718 in Healthy Subjects. 2021 Nov 6. [5]. WHO Drug Information. International Nonproprietary Names for Pharmaceutical. |
| Additional Infomation |
- Mechanism of Action: Edecesertib binds to IRAK4’s ATP-binding site, blocking Myddosome complex formation and downstream NF-κB/p38 MAPK activation, thereby inhibiting pro-inflammatory cytokine production [1,2] - Structural Optimization: The compound was developed by modifying pyridine core and R1/R3 substituents to eliminate hERG toxicity while maintaining IRAK4 potency. Key modifications included cyanomethyl substitution and pyrrolopyridazine ring [1,2] - Clinical Development: Currently in Phase II trials for cutaneous lupus erythematosus (CLE) and rheumatoid arthritis (RA) [1,2] - FDA Designation: Granted Fast Track designation for systemic lupus erythematosus (SLE) in 2023 [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~229.65 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.74 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.74 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2965 mL | 11.4824 mL | 22.9647 mL | |
| 5 mM | 0.4593 mL | 2.2965 mL | 4.5929 mL | |
| 10 mM | 0.2296 mL | 1.1482 mL | 2.2965 mL |