Physicochemical Properties
| Molecular Formula | C18H15CL3N2O |
| Molecular Weight | 381.6835 |
| Exact Mass | 380.024 |
| CAS # | 27220-47-9 |
| Related CAS # | Econazole nitrate;24169-02-6 |
| PubChem CID | 3198 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 533.8±50.0 °C at 760 mmHg |
| Melting Point | 86.8ºC |
| Flash Point | 276.6±30.1 °C |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.615 |
| LogP | 5.32 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 24 |
| Complexity | 379 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | LEZWWPYKPKIXLL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H15Cl3N2O/c19-14-3-1-13(2-4-14)11-24-18(10-23-8-7-22-12-23)16-6-5-15(20)9-17(16)21/h1-9,12,18H,10-11H2 |
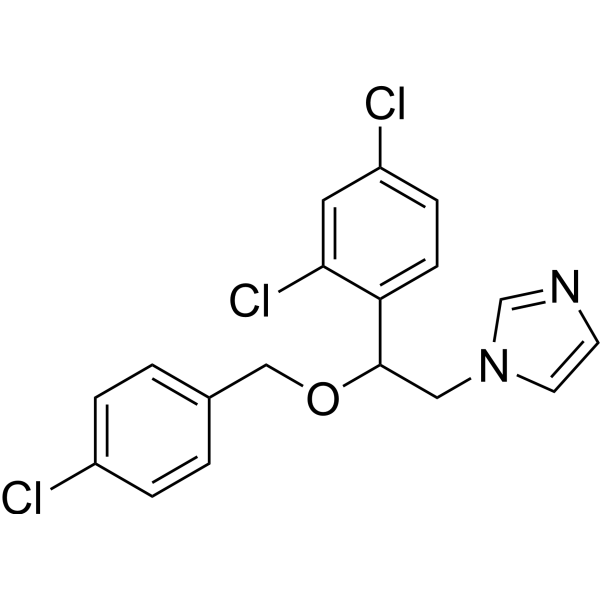
| Chemical Name | 1-[2-[(4-chlorophenyl)methoxy]-2-(2,4-dichlorophenyl)ethyl]imidazole |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Fungal lanosterol 14α-demethylase (key enzyme in ergosterol biosynthesis) [1] - Fungal cell membrane integrity [1] |
| ln Vitro |
- Econazole is a broad-spectrum antimycotic with potent inhibitory activity against various fungi. It inhibited the growth of Candida albicans with a minimum inhibitory concentration (MIC) of 0.125 μg/mL [1] - It showed activity against dermatophytes: MIC values were 0.0625 μg/mL for Trichophyton rubrum, 0.125 μg/mL for Microsporum canis, and 0.25 μg/mL for Epidermophyton floccosum [1] - It suppressed the growth of Aspergillus fumigatus and Cryptococcus neoformans with MICs of 1 μg/mL and 0.5 μg/mL, respectively [1] - The compound disrupted fungal cell membrane structure by inhibiting ergosterol synthesis, leading to increased membrane permeability and cell lysis [1] |
| ln Vivo |
- In guinea pig dermatophytosis model (induced by Trichophyton mentagrophytes): Topical application of Econazole (1% cream) twice daily for 7 days resulted in 90±3% clearance of skin lesions, with complete fungal eradication in 85% of animals [1] - In mouse systemic candidiasis model (intraperitoneal inoculation of Candida albicans): Intraperitoneal injection of Econazole (20 mg/kg daily for 5 days) reduced fungal load in the kidneys by 78±4% and prolonged survival rate by 60% compared to the control group [1] |
| Enzyme Assay |
- Lanosterol 14α-demethylase inhibition assay: Fungal microsomal fractions (from Candida albicans) were incubated with Econazole (0.01–2 μg/mL) and lanosterol (substrate) at 30°C for 2 hours. The reaction product (4,4-dimethylcholesta-8,14,24-trien-3β-ol) was quantified by thin-layer chromatography (TLC) to evaluate enzyme inhibition efficiency [1] |
| Cell Assay |
- Fungal growth inhibition assay: Fungal strains (Candida albicans, Trichophyton rubrum, etc.) were inoculated into Sabouraud dextrose broth containing Econazole (0.03125–4 μg/mL) and incubated at 28°C for 48–72 hours. Fungal growth was assessed by measuring absorbance at 600 nm, and MIC values were determined as the lowest concentration inhibiting visible growth [1] - Fungal cell membrane permeability assay: Candida albicans cells were treated with Econazole (0.25 μg/mL) for 24 hours, then stained with a membrane-impermeable fluorescent dye. Fluorescence intensity was measured by fluorometry to assess membrane disruption [1] |
| Animal Protocol |
- Guinea pig dermatophytosis model: Guinea pigs were infected with Trichophyton mentagrophytes by topical application on shaved dorsal skin. Econazole was formulated as a 1% cream and applied topically twice daily for 7 days. Skin lesions were scored visually, and fungal cultures were performed to confirm eradication [1] - Mouse systemic candidiasis model: Mice were intraperitoneally inoculated with Candida albicans (1×10⁶ CFU/mouse). Econazole was dissolved in polyethylene glycol and administered intraperitoneally at 20 mg/kg daily for 5 days. Kidneys were collected for fungal load quantification (CFU/g tissue) and survival was monitored for 14 days [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion After topical application to the skin of normal subjects, systemic absorption of econazole nitrate is extremely low. Although most of the applied drug remains on the skin surface, drug concentrations were found in the stratum corneum which, by far, exceeded the minimum inhibitory concentration for dermatophytes. Metabolism / Metabolites Hepatic. Hepatic. - Absorption: Minimal percutaneous absorption (≤5%) after topical application; oral absorption is poor (bioavailability <10%) [1] - Distribution: When administered systemically, it distributes to various tissues, with highest concentrations in the liver and kidneys [1] - Excretion: Primarily excreted via feces (65%) and urine (25%) as unchanged drug and metabolites [1] |
| Toxicity/Toxicokinetics |
Toxicity Summary Econazole interacts with 14-α demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol. As ergosterol is an essential component of the fungal cell membrane, inhibition of its synthesis results in increased cellular permeability causing leakage of cellular contents. Econazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and/or phospholipid biosynthesis. Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Topical econazole has not been studied during breastfeeding. Because less than 1% is absorbed after topical application, it is considered a low risk to the nursing infant.[1] Avoid application to the nipple area and ensure that the infant's skin does not come into direct contact with the areas of skin that have been treated. Only water-miscible cream or gel products should be applied to the breast because ointments may expose the infant to high levels of mineral paraffins via licking.[2] ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Toxicity Data LD50: 462 mg/kg (Oral, Mouse) (A308) LD50: 462 mg/kg (Oral, Rat) (A308) LD50: 668 mg/kg (Oral, Guinea pig) (A308) LD50: >160 mg/kg (Oral, Dog) (A308) - Acute toxicity: Oral LD50 in rats was >2000 mg/kg; intraperitoneal LD50 in mice was 1200 mg/kg [1] - Skin toxicity: No irritant or sensitizing effects were observed in guinea pig skin irritation tests with 1% Econazole cream [1] - Systemic toxicity: Subchronic administration (20 mg/kg daily for 28 days) in rats showed no significant changes in liver/kidney function or hematological parameters [1] |
| References |
[1]. Bilogical and toxicological properties of econazole, a broad-spectrum antimycotic. Arzneimittelforschung. 1975 Feb;25(2):224-30. |
| Additional Infomation |
1-{2-(4-chlorobenzyloxy)-2-(2,4-dichlorophenyl)ethyl}imidazole is a member of the class of imidazoles that is 1-(2,4-dichlorophenyl)-2-(imidazol-1-yl)ethanol in which the hydroxyl hydrogen is replaced by a 4-chlorobenzyl group. It is an ether, a member of imidazoles, a dichlorobenzene and a member of monochlorobenzenes. A broad spectrum antimycotic with some action against Gram positive bacteria. It is used topically in dermatomycoses also orally and parenterally. Econazole is an Azole Antifungal. Econazole is an imidazole with antifungal property. Econazole compromises the integrity of fungal cell wall through inhibiting 14-alpha demethylase, which catalyzes conversion of lanosterol to ergosterol, an essential component of the fungal cell wall. As a result, this agent increases cellular permeability thereby resulting in leakage of cellular contents. Furthermore, econazole has also been implicated to inhibit endogenous respiration, interact with membrane phospholipids, inhibit purine uptake, and impair triglyceride and/or phospholipid biosynthesis. Econazole is only found in individuals that have used or taken this drug. It is a broad spectrum antimycotic with some action against Gram positive bacteria. It is used topically in dermatomycoses also orally and parenterally. [PubChem] Econazole interacts with 14-alpha demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol. As ergosterol is an essential component of the fungal cell membrane, inhibition of its synthesis results in increased cellular permeability causing leakage of cellular contents. Econazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and/or phospholipid biosynthesis. An imidazole derivative that is commonly used as a topical antifungal agent. See also: Econazole Nitrate (has salt form); Econazole sulfosalicylate (is active moiety of). Drug Indication For topical application in the treatment of tinea pedis, tinea cruris, and tinea corporis caused by Trichophyton rubrum, Trichophyton mentagrophytes, Trichophyton tonsurans, Microsporum canis, Microsporum audouini, Microsporum gypseum, and Epidermophyton floccosum, in the treatment of cutaneous candidiasis, and in the treatment of tinea versicolor. FDA Label Mechanism of Action Econazole interacts with 14-α demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol. As ergosterol is an essential component of the fungal cell membrane, inhibition of its synthesis results in increased cellular permeability causing leakage of cellular contents. Econazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and/or phospholipid biosynthesis. Pharmacodynamics Econazole is an antifungal medication related to fluconazole (Diflucan), ketoconazole (Nizoral), itraconazole (Sporanox), and clotrimazole (Lotrimin, Mycelex). Econazole prevents fungal organisms from producing vital substances required for growth and function. This medication is effective only for infections caused by fungal organisms. It will not work for bacterial or viral infections. - Econazole is an imidazole-class broad-spectrum antimycotic agent [1] - Its mechanism of action involves inhibiting fungal lanosterol 14α-demethylase, a key enzyme in ergosterol biosynthesis, leading to depletion of ergosterol and accumulation of toxic sterol intermediates, which disrupts fungal cell membrane integrity [1] - It is clinically indicated for the treatment of superficial fungal infections (e.g., tinea corporis, tinea pedis, candidiasis) and has potential for systemic use in severe fungal infections [1] - It shows no significant cross-resistance with other antimycotics and has low toxicity to mammalian cells [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~262.00 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.55 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6200 mL | 13.1000 mL | 26.2000 mL | |
| 5 mM | 0.5240 mL | 2.6200 mL | 5.2400 mL | |
| 10 mM | 0.2620 mL | 1.3100 mL | 2.6200 mL |