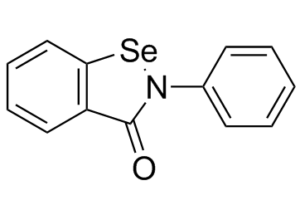
Ebselen (formerly SPI-1005; PZ-51; DR-3305; CCG-39161), is an organoselenium-based small molecule biochemical compound with potential anti-inflammatory, anti-oxidant, and cytoprotective activity. It is a capsid inhibitor of HIV-1 replication and is also a voltage-dependent calcium channel (VDCC) blocker. Ebselen inhibits Mpro and COVID-19 virus with an IC50 of 0.67 μM and an EC50 of 4.67 μM, respectively. After oral gavage, Ebselen acts by mimicking the activity of glutathione peroxidase (GPx) and can utilize glutathione to reduce other unstable molecules, thus preventing the formation of reactive oxygen species (ROS) and reducing oxidative stress on the cell. In the cochlea, this agent may prevent drug-induced injury to the auditory hair cells thereby preventing hearing loss. GPx is the main antioxidant enzyme in the cochlea and protects the inner ear from loud sounds and biochemical damage. In addition, ebselen is able to inhibit the activity of many enzymes involved in inflammation.
Physicochemical Properties
| Molecular Formula | C13H9NOSE | |
| Molecular Weight | 274.17666220665 | |
| Exact Mass | 274.984 | |
| Elemental Analysis | C, 56.95; H, 3.31; N, 5.11; O, 5.84; Se, 28.80 | |
| CAS # | 60940-34-3 | |
| Related CAS # |
|
|
| PubChem CID | 3194 | |
| Appearance | Light yellow to yellow solid powder | |
| Boiling Point | 402.8±28.0 °C at 760 mmHg | |
| Melting Point | 178-181 °C | |
| Flash Point | 197.4±24.0 °C | |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C | |
| LogP | 2.047 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 1 | |
| Rotatable Bond Count | 1 | |
| Heavy Atom Count | 16 | |
| Complexity | 275 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | [Se]1C2C=CC=CC=2C(N1C1C=CC=CC=1)=O |
|
| InChi Key | DYEFUKCXAQOFHX-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C13H9NOSe/c15-13-11-8-4-5-9-12(11)16-14(13)10-6-2-1-3-7-10/h1-9H | |
| Chemical Name | 2-phenyl-1,2-benzoselenazol-3-one | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
VDCC/voltage-dependent calcium channel; HIV-1; COVID-19
Glutathione Peroxidase (GPx) (acts as a GPx mimic) [2] COVID-19 Main Protease (Mpro) (IC50 = 0.67 μM, enzyme activity inhibition assay) [3] HIV-1 Capsid (IC50 = 0.2 μM, viral replication inhibition assay) [4] Quiescin Sulfhydryl Oxidase 1 (QSOX1) (IC50 = 1.2 μM, enzyme activity inhibition assay) [6] |
| ln Vitro |
In COVID-19 virus-infected Vero cells, Ebselen (SPI-1005; 0.4-100 μM; 20-24 hours) exhibits potent antiviral effects at a concentration of 10 μM treatment. Ebsele attaches itself covalently to COVID-19 virus Mpro's catalytic dyad at position C145[3]. Viral postentry events of the HIV-1 life cycle are inhibited by ebselen through a decrease in the incoming capsid uncoating process[4]. In the mouse brain, endogenous inositol monophosphatase is inhibited by ebselen, which also penetrates the blood-brain barrier. Inositol monophosphatase (IMPase) is inhibited by ebselen [5]. Ebselen suppresses invasion of pancreatic and renal cancer cell lines and inhibits the enzymatic activity of QSOX1[6]. 1. Protection against cochlear synapse degeneration: Ebselen (1, 5, 10 μM) dose-dependently attenuated electrical stimulation-induced cochlear synapse degeneration in neonatal rat cochlear explants. At 10 μM, it reduced the loss of presynaptic ribbons (CtBP2-positive) and postsynaptic receptors (GluA2-positive) by 65% compared to the control group. It also decreased reactive oxygen species (ROS) levels by 58% and increased the expression of antioxidant enzymes SOD1 (1.8-fold) and CAT (1.6-fold) (immunofluorescence staining and ROS assay) [1] 2. Glutathione peroxidase mimetic activity: Ebselen exhibited GPx-like activity, catalyzing the reduction of hydrogen peroxide (H2O2) and organic hydroperoxides using glutathione as a co-substrate. It inhibited lipid peroxidation in rat liver microsomes with an IC50 of ~5 μM, as measured by thiobarbituric acid-reactive substances (TBARS) assay [2] 3. Inhibition of COVID-19 Mpro activity: Ebselen dose-dependently inhibited the protease activity of recombinant COVID-19 Mpro, with an IC50 of 0.67 μM. It blocked Mpro-mediated cleavage of viral polyproteins, as demonstrated by a cell-based viral replication assay (Vero cells), reducing viral load by 70% at 1 μM [3] 4. Inhibition of HIV-1 replication: Ebselen suppressed HIV-1 replication in infected TZM-bl cells and primary human CD4+ T cells with IC50 values of 0.2 μM and 0.3 μM, respectively. It interfered with HIV-1 capsid assembly and stability, as shown by capsid protein (p24) aggregation assays and transmission electron microscopy [4] 5. Lithium-mimetic activity: Ebselen (1-10 μM) inhibited glycogen synthase kinase 3β (GSK3β) activity in HEK293 cells, increasing phosphorylation of GSK3β at Ser9 (1.9-fold at 5 μM) without affecting total GSK3β expression (Western blot). It also mimicked lithium's effect on Wnt/β-catenin signaling, increasing β-catenin nuclear translocation [5] 6. Inhibition of cancer cell invasion and QSOX1 activity: Ebselen (0.5-5 μM) dose-dependently inhibited the invasion of pancreatic cancer (PANC-1, Mia PaCa-2) and renal cancer (ACHN, 786-O) cells. At 3 μM, it reduced cell invasion by 55-68% (Matrigel invasion assay) and downregulated the expression of matrix metalloproteinases MMP-2 (0.4-fold) and MMP-9 (0.3-fold) (Western blot). It also directly inhibited QSOX1 enzyme activity with an IC50 of 1.2 μM [6] |
| ln Vivo |
In a dose-dependent manner, ebselen (5, 10 mg/kg; IP) reduces head twitches induced by 5-HT2 agonists[5]. Ebselen is pharmacologically active in the brain [5] To determine whether ebselen can cross the blood–brain barrier and thus be pharmacologically active in mouse brain, as reported for rat23, we exploited the irreversible inhibition property of ebselen in an ex vivo method based on IMPase activity in brain homogenate (Fig. 2a). As the initial experiments that identified ebselen as an inhibitor used recombinant human IMPase (Fig. 1b), we first needed to ensure that recombinant mouse IMPase was enzymatically active. Recombinant mouse IMPase was inhibited by lithium and L-690, 330 and ebselen (Fig. 2b). Having validated that ebselen inhibited the mouse form of IMPase, we demonstrated that in homogenates of mouse brain, IMPase activity was detectable and inhibited by lithium, L-690,330 and ebselen (Fig. 2c). In an ex vivo experiment, IMPase activity was measured in brain homogenates prepared at various times after intraperitoneal injection of ebselen (Fig. 2a)24. Over time, IMPase inhibition developed and then returned to control levels (Fig. 2d,e). Therefore, systemic administration of ebselen inhibits IMPase in mouse brain in whole animals. Ebselen alters the function of the central nervous system [5] Ebselen decreased 5-HT2 agonist-induced head twitches in a dose-dependent manner (Fig. 3a), and this was associated with decreased expression of Arc mRNA (a molecular marker of neural activity26) in the prefrontal cortex (Fig. 3b) and cingulate cortex (Fig. 3c). Thus, ebselen attenuates a cortically mediated 5-HT2 receptor response that is linked to phosphoinositide turnover, as would be predicted for an inhibitor of IMPase. Ebselen exhibits lithium-like effects on behaviour [5] In the open field test (Fig. 3d), rearing was decreased by ebselen over time and then returned to baseline (Fig. 3e), a time course that paralleled that for IMPase inhibition in the ex vivo assay (Fig. 2e), as well as plasma ebselen concentrations in humans after oral administration34. Rearing is an exploratory behaviour that correlates with impulsivity33, which in turn correlates with suicidal thoughts and actions35. Mania has also been modelled by amphetamine-induced hyperactivity (Fig. 3f)33,36. Similarly to lithium37, ebselen reduced amphetamine-induced hyperactivity in a manner that depended on both the dose of amphetamine and the dose of ebselen (Fig. 3g), as is the case for lithium37. Baseline mobility was not significantly reduced (one-tailed, paired t-tests: amphetamine 2 mg/kg and ebselen 5 mg/kg, p=0.24; amphetamine 4 mg/kg and ebselen 5 mg/kg, p=0.08). 1. Lithium-mimetic effect in bipolar disorder model: Male C57BL/6 mice were orally administered Ebselen (10 mg/kg/day) for 14 days. It reduced amphetamine-induced hyperlocomotion by 45% compared to the vehicle group. Western blot analysis of brain tissue showed increased phosphorylation of GSK3β (Ser9) in the prefrontal cortex and hippocampus (1.7-fold and 1.5-fold, respectively) [5] |
| Enzyme Assay |
IMPase Activity [5] Phosphate hydrolyzed from Ins1P was detected using the malachite green assay. For the in vitro assays, recombinant HsIMPase (10 ng/well) or MmIMPase (30 ng/well) was incubated (1 h, 37°C) with Ins1P (1mM) in 20 μL Tris buffer (50 mM Tris-HCl, 1 mM EGTA, 3 mM MgCl2, 150 mM KCl, 0.5 mg/mL BSA and 0.01% v/v Triton X pH 7.4). Absorbance was measured at 595 nm for samples and phosphate standards. For the ex vivo assays, brain homogenate (0.5 mg/mL) was incubated (37°C, 1 h) with Ins1P (0.1-2.4 mM) in the presence or absence of LiCl (30 mM) to determine IMPase-specific activity. Chemical Library and Screening [5] Compounds (100 μM) were screened at three concentrations of Ins1P. Initial hits were confirmed with concentration–inhibition curves spanning six orders of magnitude. Subsequent experiments used ebselen. For compound screening, compound at 100 μM (in 0.2% v/v DMSO) was incubated with IMPase (10 min, room temperature) in buffer, before addition of Ins1P (1 mM) to a final volume of 20 μL and further incubated (37°C, 1 h). Phosphatase concentration was determined by the malachite green assay. LiCl and L-690,330 (Tocris) were used as positive controls. rQSOX1 activity assay [6] The sulfhydryl oxidase activity of rQSOX1 was confirmed using DTT and RNAse A substrates and a fluorogenic hydrogen peroxide indicator, homovanilic acid (HVA) [8]. In this assay, 150 nM rQSOX1 was added to 600 μM thiols from reduced DTT or RNAse A, 1 mM HVA, 1.4 μM HRP, and 300 μM EDTA in PBS at 25°C, pH 7.5. Assays were performed in black 96-well plates with a final reaction volume of 150 μl. Fluorescence signal was measured over 10 minutes at λex 320 nm and λem 420 nm using a FlexStation spectrophotometer). Readings were taken in 20 second intervals after the addition of rQSOX1. Ebselen was added to reactions at least 10 minutes prior to the addition of rQSOX1 at concentrations ranging from 250 nM – 4 μM. 1. GPx-like activity assay: The reaction mixture contained glutathione, glutathione reductase, NADPH, and H2O2 as the substrate. Different concentrations of Ebselen (0.1-10 μM) were added, and the decrease in NADPH absorbance at 340 nm was monitored continuously for 5 minutes. The GPx-like activity was calculated based on the rate of NADPH oxidation [2] 2. COVID-19 Mpro protease activity assay: Recombinant Mpro was incubated with Ebselen (0.01-10 μM) for 30 minutes at 37℃. A fluorogenic substrate (MCA-AVLQSGFR-Lys(Dnp)-Lys-NH2) was added, and fluorescence intensity was measured at excitation 328 nm and emission 393 nm for 60 minutes. The IC50 value was derived from the dose-response curve of protease activity inhibition [3] 3. QSOX1 enzyme activity assay: Recombinant QSOX1 was mixed with Ebselen (0.1-10 μM) in assay buffer. The reaction was initiated by adding dithiothreitol (DTT) as the substrate, and the formation of disulfide bonds was detected using 5,5'-dithiobis-(2-nitrobenzoic acid) (DTNB). The absorbance at 412 nm was measured, and the inhibition rate of QSOX1 activity was calculated to determine the IC50 [6] |
| Cell Assay |
RT-PCR[3] Cell Types: COVID-19 virus infected Vero cells Tested Concentrations: 0.4, 1.2, 3.7, 11.1, 33.3, 100 μM Incubation Duration: 20-24 hrs (hours) Experimental Results: demonstrated strong antiviral effects at a concentration of 10 μM treatment. Growth kinetics of ebselen-treated tumor cells [6] 1 × 104 cells/well MIAPaCa-2, BXPC3, 786-O, and UOK1117 were plated in duplicate in 24-well plates. Cells were adhered overnight prior to the addition of fresh media (untreated), vehicle (0.15% DMSO), or 5 μM – 15 μM ebselen. Cells were counted using a hemacytometer and Trypan Blue exclusion to assess viability. Cells were counted on days 3 and 5, and “floaters” (disadhered and dead cells) were saved for determination of overall viability. Media was replaced on day 3 for the 5th day time point; floaters were saved and added back to each well for counting on day 5. Viability was determined as [1-(# dead cells / (# live cells + # dead cells))*100]. Error is represented as the standard error of the mean. Significance was determined using paired T-testing for each time point compared to vehicle-treated cells. Trans-well invasion assays [6] 2 × 104 MIAPaCa-2, BXPC3, 786-O, or UOK117 cells were seeded in rehydrated 24-well invasion assay inserts containing 8 μm pores overlaid with Matrigel in serum-free media; cells were adhered for 1 hour prior to addition of ebselen or vehicle. Inserts were incubated in wells containing complete media for 20 hours at 37°C. Non-invading cells were removed with cotton swabs and membranes were fixed with 100% methanol and mounted on slides with DAPI. The total number of invading cells was determined by manual counting of DAPI-stained nuclei. 1. Cochlear explant culture and synapse analysis: Neonatal rat cochleae were dissected and cultured in vitro for 3 days. Explants were pre-treated with Ebselen (1, 5, 10 μM) for 1 hour, then subjected to electrical stimulation (100 Hz, 2 hours). After 24 hours of incubation, explants were fixed, immunostained with anti-CtBP2 (presynaptic) and anti-GluA2 (postsynaptic) antibodies, and imaged using a confocal microscope. The number of synapses was counted to evaluate degeneration [1] 2. HIV-1 replication assay: TZM-bl cells were seeded in 96-well plates and infected with HIV-1 (NL4-3 strain) at a multiplicity of infection (MOI) of 0.01. Ebselen (0.01-1 μM) was added immediately after infection. After 48 hours, luciferase activity was measured to quantify viral replication, and IC50 values were calculated [4] 3. GSK3β phosphorylation assay: HEK293 cells were seeded in 6-well plates and treated with Ebselen (1-10 μM) for 24 hours. Cells were lysed in RIPA buffer with protease/phosphatase inhibitors, and total protein was analyzed by Western blot using antibodies against p-GSK3β (Ser9), total GSK3β, and GAPDH. Band intensities were quantified to assess phosphorylation levels [5] 4. Cancer cell invasion assay: PANC-1 and ACHN cells were resuspended in serum-free medium and seeded in Matrigel-coated transwell inserts (8 μm pore size) at 5×10^4 cells/well. Ebselen (0.5, 1, 3, 5 μM) was added to both upper and lower chambers, with the lower chamber containing medium with 10% FBS. After 24 hours (renal cancer) or 48 hours (pancreatic cancer), invading cells were fixed, stained with crystal violet, and counted under a microscope [6] 5. Western blot for MMPs: Cancer cells were treated with Ebselen (3 μM) for 24 hours, lysed, and total protein was separated by SDS-PAGE. Membranes were probed with antibodies against MMP-2, MMP-9, and GAPDH. Chemiluminescent signals were detected and quantified [6] |
| Animal Protocol |
Animal/Disease Models: 20-25 g 10-12 week old male C57Bl6 mice[5] Doses: 5, 10 mg/kg Route of Administration: IP Experimental Results: diminished 5-HT2 agonist-induced head twitches in a dose-dependent manner. Ex Vivo Mouse Brain Homogenate [5] Mice were injected with ebselen (10 mg/kg) or vehicle (4% w/v hydroxypropyl ß-cyclodextrin) and left for varying amounts of time before euthanization by cervical dislocation, or by CO2 followed by cervical dislocation. Brains were removed and frozen on dry ice immediately. One hemisphere was homogenized using a Precellys 24 bead mill homogenizer and diluted in Tris buffer (50 mM Tris HCl, 3 mM MgCl2, 150 mM KCl, 1 mM EGTA, 0.01% v/v Triton X pH 7.4) to a final concentration of 0.5 mg/mL. Ex vivo Inositol Measurement by Nuclear Magnetic Resonance [5] Mice were euthanized by cervical dislocation 1 h after administration of ebselen (10 mg/kg) or vehicle (4% w/v hydroxypropyl ß-cyclodextrin), then brains were extracted and frozen immediately on dry ice. Brains were weighed then homogenized using a Precellys 24 bead mill homogenizer. Acetonitrile was added to homogenate (1:1 v/v) to precipitate protein, the sample was centrifuged (13,000×g, 10 min), and the supernatant was prepared for NMR by lyophilization and reconstitution in D2O with 0.008% w/v 3- (trimethylsilyl)propionic 2233d acid sodium salt (600 mg/mL). Amphetamine-induced Hyperactivity [5] Mice were treated with ebselen or vehicle and immediately placed in Linton AM1053 X, Y, Z IR Activity Monitor for 1 h to habituate. Mice were then injected with d-amphetamine/saline and returned to the cage, and activity was monitored for an additional 1 h. Rearing behavior [5] Mice were injected with ebselen (10 mg/kg) or vehicle (4% w/v hydroxypropyl ß-cyclodextrin) and left for varying amounts of time before being placed in the Linton AM1053 X, Y, Z IR Activity Monitor for 30 mins while their activity was monitored. Rearing was measured by counting the number of beam breaks in upper grid. DOI-induced Head Twitches [5] Mice were placed in an arena and left to acclimatize to the novel environment. After 1 h, they were injected with vehicle or ebselen (5 or 10 mg/kg) followed 1 h later by the non-selective 5HT2A agonist 1- (2,5-dimethoxy-4 iodophenyl)-2-aminopropane (DOI, 2 mg/kg). Head twitches were recorded 5 min after agonist injection for 15 min. Mice were constantly monitored by a video camera, and behavioural recordings were analysed offline independently by two observers who were blind to the treatment. Nude mouse-human tumor xenograft model [6] For ebselen treatment of nude mice, three groups were tested: 1) 20% DMSO (vehicle), 2) 160 μg/day ebselen, and 3) 640 μg/day ebselen. 160 and 640 μg ebselen represent an equivalent dose of 150mg and 600mg for a 70 kg human, respectively. 1 × 106 MIAPaCa-2 cells were injected subcutaneously into each mouse as before, and tumors were allowed to grow for 3 days. Ebselen was then administered once daily through oral gavage for 28 days. Real-time tumor volume was determined through caliper measurement of tumors over the course of the study. 1. Bipolar disorder mouse model: Male C57BL/6 mice (8-10 weeks old, 20-25 g) were randomly divided into 2 groups (n=8/group): vehicle control (0.5% carboxymethylcellulose sodium) and Ebselen 10 mg/kg/day group. Ebselen was suspended in 0.5% CMC-Na and administered orally by gavage once daily for 14 days. On day 14, mice were injected with amphetamine (2 mg/kg, i.p.) 30 minutes before locomotor activity testing. Locomotor activity was monitored for 60 minutes in an open field arena. After behavioral testing, mice were sacrificed, and brain tissues (prefrontal cortex, hippocampus) were collected for Western blot analysis [5] |
| ADME/Pharmacokinetics |
1. Oral bioavailability: In rats, oral administration of Ebselen (10 mg/kg) resulted in an absolute bioavailability of 35% [2] 2. Plasma pharmacokinetics: After oral administration (10 mg/kg) in rats, the peak plasma concentration (Cmax) was 1.8 μM (achieved at 1 hour), area under the curve (AUC0-24h) was 12.6 μM·h, and elimination half-life (t1/2) was 5.2 hours [2] 3. Tissue distribution: In mice, 2 hours after oral administration of Ebselen (10 mg/kg), the highest drug concentrations were detected in the liver (6.3 μM) and kidney (4.8 μM), followed by the brain (1.2 μM) and lung (1.0 μM) [5] |
| Toxicity/Toxicokinetics |
rat LD oral >4600 mg/kg European Patent Application., #0044971 mouse LDLo oral 5 gm/kg SENSE ORGANS AND SPECIAL SENSES: PTOSIS: EYE; BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD; BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) Yakuri to Chiryo. Pharmacology and Therapeutics., 25(Suppl pig LD oral >2 gm/kg Yakuri to Chiryo. Pharmacology and Therapeutics., 25(Suppl 1. Acute toxicity: In rats, single oral administration of Ebselen at doses up to 500 mg/kg did not cause significant mortality or obvious toxic symptoms (e.g., lethargy, weight loss, gastrointestinal distress) within 14 days of observation [2] 2. Chronic toxicity: Mice treated with Ebselen (10 mg/kg/day, oral) for 28 days showed no significant changes in liver function (ALT, AST) or kidney function (BUN, creatinine) compared to the vehicle group. Histopathological analysis of major organs (liver, kidney, heart, brain) revealed no abnormal lesions [5] 3. Cell toxicity: Ebselen at concentrations up to 10 μM did not affect the viability of neonatal cochlear explant cells or normal human fibroblasts (WI-38) (MTT assay) [1][6] |
| References |
[1]. Electrical Stimulation Degenerated Cochlear Synapses Through Oxidative Stress in NeonatalCochlear Explants. Front Neurosci. 2019 Oct 14;13:1073. [2]. Ebselen, a Selenoorganic Compound as Glutathione Peroxidase Mimic. Free Radic Biol Med. 1993 Mar;14(3):313-23. [3]. Structure of Mpro from COVID-19 virus and discovery of its inhibitors. Nature. 2020 Apr 9. [4]. Ebselen, a Small-Molecule Capsid Inhibitor of HIV-1 Replication. Antimicrob Agents Chemother. 2016 Mar 25;60(4):2195-208. [5]. A safe lithium mimetic for bipolar disorder. Nat Commun. 2013;4:1332. [6]. Ebselen inhibits QSOX1 enzymatic activity and suppresses invasion of pancreatic and renal cancer cell lines. Oncotarget. 2015 Jul 30;6(21):18418-28. |
| Additional Infomation |
Ebselen is a benzoselenazole that is 1,2-benzoselenazol-3-one carrying an additional phenyl substituent at position 2. Acts as a mimic of glutathione peroxidase. It has a role as a neuroprotective agent, an apoptosis inducer, an anti-inflammatory drug, an antioxidant, a hepatoprotective agent, a genotoxin, a radical scavenger, an enzyme mimic, an EC 1.3.1.8 [acyl-CoA dehydrogenase (NADP(+))] inhibitor, an EC 1.8.1.12 (trypanothione-disulfide reductase) inhibitor, an EC 1.13.11.33 (arachidonate 15-lipoxygenase) inhibitor, an EC 1.13.11.34 (arachidonate 5-lipoxygenase) inhibitor, an EC 2.5.1.7 (UDP-N-acetylglucosamine 1-carboxyvinyltransferase) inhibitor, an EC 2.7.10.1 (receptor protein-tyrosine kinase) inhibitor, an EC 3.5.4.1 (cytosine deaminase) inhibitor, an EC 5.1.3.2 (UDP-glucose 4-epimerase) inhibitor, a ferroptosis inhibitor, an antifungal agent, an EC 3.4.22.69 (SARS coronavirus main proteinase) inhibitor, an anticoronaviral agent, an antibacterial agent, an antineoplastic agent and an EC 3.1.3.25 (inositol-phosphate phosphatase) inhibitor. Ebselen has been investigated for the treatment and basic science of Meniere's Disease, Type 2 Diabetes Mellitus, and Type 1 Diabetes Mellitus. Ebselen is a organoselenium compound with anti-inflammatory, anti-oxidant and cytoprotective activity. Ebselen acts as a glutathione peroxidase mimetic and is thereby able to prevent cellular damage induced by reactive oxygen species (ROS). In addition, this agent inhibits the activity of a variety of enzymes including nitric oxide synthase (NOS), 5-lipoxygenase, cyclooxygenase, protein kinase C (PKC), NADPH oxidase and gastric H+/K+-ATPase. Furthermore, ebselen may be neuroprotective due to its ability to neutralize free radicals upon NMDA receptor activation thus, reducing lipoperoxidation mediated by glutamate-induced excitotoxicity. \n\nThe selenoorganic compound ebselen, 2-phenyl-1,2-benzisoselenazol-3(2H)-one, exhibits activity as an enzyme mimic. The reaction catalyzed is that of a glutathione (GSH) peroxidase (i.e., the reduction of a hydroperoxide at the expense of thiol). The specificity for substrates ranges from hydrogen peroxide and smaller organic hydroperoxides to membrane-bound phospholipid and cholesterol hydroperoxides. In addition to glutathione, the thiol reductant cosubstrate can be dithioerythritol, N-acetylcysteine or dihydrolipoate, or other suitable thiol compounds. Ebselen also has properties such as free radical and singlet oxygen quenching. Model experiments in vitro with liposomes, microsomes, isolated cells, and organs show that the protection against oxidative challenge afforded by ebselen can be explained largely by the activity as GSH peroxidase mimic. Whether this also explains the known preliminary results in clinical settings is yet open. The metabolism and disposition of ebselen is presented in this review. The main point is that the selenium is not bioavailable, explaining the extremely low toxicity observed in animal studies. The occurrence of natural GPx mimics, ovothiol and related compounds, is briefly mentioned. [2] \nA new coronavirus, known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is the aetiological agent responsible for the 2019-2020 viral pneumonia outbreak of coronavirus disease 2019 (COVID-19)1-4. Currently, there are no targeted therapeutic agents for the treatment of this disease, and effective treatment options remain very limited. Here we describe the results of a programme that aimed to rapidly discover lead compounds for clinical use, by combining structure-assisted drug design, virtual drug screening and high-throughput screening. This programme focused on identifying drug leads that target main protease (Mpro) of SARS-CoV-2: Mpro is a key enzyme of coronaviruses and has a pivotal role in mediating viral replication and transcription, making it an attractive drug target for SARS-CoV-25,6. We identified a mechanism-based inhibitor (N3) by computer-aided drug design, and then determined the crystal structure of Mpro of SARS-CoV-2 in complex with this compound. Through a combination of structure-based virtual and high-throughput screening, we assayed more than 10,000 compounds-including approved drugs, drug candidates in clinical trials and other pharmacologically active compounds-as inhibitors of Mpro. Six of these compounds inhibited Mpro, showing half-maximal inhibitory concentration values that ranged from 0.67 to 21.4 μM. One of these compounds (ebselen) also exhibited promising antiviral activity in cell-based assays. Our results demonstrate the efficacy of our screening strategy, which can lead to the rapid discovery of drug leads with clinical potential in response to new infectious diseases for which no specific drugs or vaccines are available.\n[3] \nThe human immunodeficiency virus type 1 (HIV-1) capsid plays crucial roles in HIV-1 replication and thus represents an excellent drug target. We developed a high-throughput screening method based on a time-resolved fluorescence resonance energy transfer (HTS-TR-FRET) assay, using the C-terminal domain (CTD) of HIV-1 capsid to identify inhibitors of capsid dimerization. This assay was used to screen a library of pharmacologically active compounds, composed of 1,280in vivo-active drugs, and identified ebselen [2-phenyl-1,2-benzisoselenazol-3(2H)-one], an organoselenium compound, as an inhibitor of HIV-1 capsid CTD dimerization. Nuclear magnetic resonance (NMR) spectroscopic analysis confirmed the direct interaction of ebselen with the HIV-1 capsid CTD and dimer dissociation when ebselen is in 2-fold molar excess. Electrospray ionization mass spectrometry revealed that ebselen covalently binds the HIV-1 capsid CTD, likely via a selenylsulfide linkage with Cys198 and Cys218. This compound presents anti-HIV activity in single and multiple rounds of infection in permissive cell lines as well as in primary peripheral blood mononuclear cells. Ebselen inhibits early viral postentry events of the HIV-1 life cycle by impairing the incoming capsid uncoating process. This compound also blocks infection of other retroviruses, such as Moloney murine leukemia virus and simian immunodeficiency virus, but displays no inhibitory activity against hepatitis C and influenza viruses. This study reports the use of TR-FRET screening to successfully identify a novel capsid inhibitor, ebselen, validating HIV-1 capsid as a promising target for drug development.\n[4] \nLithium is the most effective mood stabilizer for the treatment of bipolar disorder, but it is toxic at only twice the therapeutic dosage and has many undesirable side effects. It is likely that a small molecule could be found with lithium-like efficacy but without toxicity through target-based drug discovery; however, therapeutic target of lithium remains equivocal. Inositol monophosphatase is a possible target but no bioavailable inhibitors exist. Here we report that the antioxidant ebselen inhibits inositol monophosphatase and induces lithium-like effects on mouse behaviour, which are reversed with inositol, consistent with a mechanism involving inhibition of inositol recycling. Ebselen is part of the National Institutes of Health Clinical Collection, a chemical library of bioavailable drugs considered clinically safe but without proven use. Therefore, ebselen represents a lithium mimetic with the potential both to validate inositol monophosphatase inhibition as a treatment for bipolar disorder and to serve as a treatment itself.\n[5] \nQuiescin sulfhydryl oxidase 1 (QSOX1) is a highly conserved disulfide bond-generating enzyme that is overexpressed in diverse tumor types. Its enzymatic activity promotes the growth and invasion of tumor cells and alters extracellular matrix composition. In a nude mouse-human tumor xenograft model, tumors containing shRNA for QSOX1 grew significantly more slowly than controls, suggesting that QSOX1 supports a proliferative phenotype in vivo. High throughput screening experiments identified ebselen as an in vitro inhibitor of QSOX1 enzymatic activity. Ebselen treatment of pancreatic and renal cancer cell lines stalled tumor growth and inhibited invasion through Matrigel in vitro. Daily oral treatment with ebselen resulted in a 58% reduction in tumor growth in mice bearing human pancreatic tumor xenografts compared to controls. Mass spectrometric analysis of ebselen-treated QSOX1 mechanistically revealed that C165 and C237 of QSOX1 covalently bound to ebselen. This report details the anti-neoplastic properties of ebselen in pancreatic and renal cancer cell lines. The results here offer a \"proof-of-principle\" that enzymatic inhibition of QSOX1 may have clinical relevancy.\n[6] 1. Ebselen is a synthetic selenoorganic compound with multiple biological activities, primarily acting as a glutathione peroxidase (GPx) mimic to scavenge reactive oxygen species (ROS) and inhibit lipid peroxidation, thereby exerting antioxidant effects [2] 2. It exhibits broad-spectrum pharmacological activities, including inhibition of viral proteases (COVID-19 Mpro) and HIV-1 capsid assembly, making it a potential antiviral agent. Its lithium-mimetic activity (inhibiting GSK3β) suggests utility in treating bipolar disorder [3][4][5] 3. In cancer, Ebselen inhibits cell invasion by targeting QSOX1 and downregulating MMP-2/9 expression, highlighting its potential as an anti-metastatic agent. Its low toxicity and favorable tissue distribution (including brain penetration) support clinical development for multiple indications [6] 4. The mechanism of action of Ebselen involves covalent binding to cysteine residues of target proteins (e.g., Mpro, QSOX1, GSK3β), modulating their activity. Its multifunctional nature is attributed to its ability to interact with multiple cellular and viral targets [3][5][6] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (9.12 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6472 mL | 18.2362 mL | 36.4724 mL | |
| 5 mM | 0.7294 mL | 3.6472 mL | 7.2945 mL | |
| 10 mM | 0.3647 mL | 1.8236 mL | 3.6472 mL |