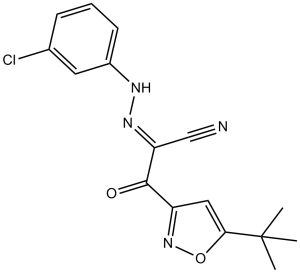
ESI-09 (ESI09) is a non-noncyclic nucleotide and specific inhibitor of exchange protein directly activated by cAMP (EPAC) with IC50 of 3.2 μM and 1.4 μM for EPAC1 and EPAC2, respectively. As an EPAC antagonist, it displays >100-fold selectivity over PKA. ESI-09 (25 µM) reduced EPAC1 and EPAC2 GEF activity to basal levels in the presence of 25 µM cAMP. In the presence of 25 µM cAMP, ESI-09 inhibited cAMP-mediated EPAC2 and EPAC1 GEF activity with IC50 values of 1.4 and 3.2 µM respectively and exhibited 100 times selectivity than PKA.
Physicochemical Properties
| Molecular Formula | C16H15CLN4O2 | |
| Molecular Weight | 330.77 | |
| Exact Mass | 330.088 | |
| Elemental Analysis | C, 58.10; H, 4.57; Cl, 10.72; N, 16.94; O, 9.67 | |
| CAS # | 263707-16-0 | |
| Related CAS # |
|
|
| PubChem CID | 6077765 | |
| Appearance | Yellow to orange solid powder | |
| LogP | 3.872 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 23 | |
| Complexity | 520 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | ClC1=C([H])C([H])=C([H])C(=C1[H])N([H])/N=C(\C#N)/C(C1C([H])=C(C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H])ON=1)=O |
|
| InChi Key | DXEATJQGQHDURZ-DEDYPNTBSA-N | |
| InChi Code | InChI=1S/C16H15ClN4O2/c1-16(2,3)14-8-12(21-23-14)15(22)13(9-18)20-19-11-6-4-5-10(17)7-11/h4-8,19H,1-3H3/b20-13+ | |
| Chemical Name | (E)-2-(5-(tert-butyl)isoxazol-3-yl)-N-(3-chlorophenyl)-2-oxoacetohydrazonoyl cyanide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | EPAC1(IC50=3.2 μM);EPAC2(IC50=1.4 μM) |
| ln Vitro | ESI-09 exhibits greater potency with an apparent IC50 of 10 μM, whereas cAMP competes with 8-NBD-cAMP binding with an IC50 of 39 μM. ESI-09 has an IC50 of 1.4μM for EPAC2 and 3.2μM for EPAC1 GEF activity, which is cAMP-mediated. With ESI-09's potential fit into EPAC1's functional cAMP-binding pocket, the protein's active-site residues may engage in advantageous hydrophobic and hydrogen bonding interactions. In a dose-dependent manner, ESI-09 prevents 007-AM-stimulated Akt phosphorylation at T308 and S473. AsPC-1 and PANC-1 migration of pancreatic cancer cells is inhibited by ESI-09. PDA cells' EPAC1-mediated adhesion to collagen I is inhibited by ESI-09[1]. When compared to similarly infected controls, exposure to ESI-09 significantly lowers the intracellular and total bacterial counts in HUVECs 30 minutes post-infection with 10 multiplicities of infection (MOI) of R. australis[2]. |
| ln Vivo | WT mice treated with ESI-09 have significantly improved survival rates and are significantly protected against R. australis infection with much milder disease manifestations[2]. |
| Cell Assay | Polylysine is coated in advance on 96-well plates before INS-1 cells are plated. Following an overnight incubation period, Krebs-Ringer bicarbonate (KRB) containing 2.9 mM glucose is added to the medium. Following a further two hours of incubation, the cells are stimulated for thirty minutes with 10 µM 007-AM, or they are treated with ESI-09 or DMSO vehicle as a control in fresh KRB containing 11.8 mM glucose for ten minutes. An Ultra Sensitive Rat Insulin ELISA kit from Crystal Chem Inc. is used to quantify insulin after the supernatant is collected[1]. |
| Animal Protocol | Mice;In buffered saline containing 10% (vol/vol) ethanol and 10% (vol/vol) Tween-80, ESI-09 is dissolved. There are four groups of thirty-three WT C57BL/6 mice: group A consists of eleven mice, group B of ten mice, and groups C and D of six mice each. Groups B and D receive vehicle treatment, followed by either mock inoculation for groups C and D or i.v. inoculation of R. australis for groups A and B. Groups A and C receive treatment with the Epac-specific inhibitor ESI-09 [10 mg/kg] via i.p. injection for five days prior to infection. After seven more days of ESI-09 or vehicle treatment, the mice are killed on day eight. Animals are observed daily throughout the trials for indications of disease and death[2]. |
| References |
[1]. A novel EPAC-specific inhibitor suppresses pancreatic cancer cell migration and invasion. Mol Pharmacol. 2013 Jan;83(1):122-8. [2]. Exchange protein directly activated by cAMP plays a critical role in bacterial invasion during fatal rickettsioses. Proc Natl Acad Sci U S A. 2013 Nov 26;110(48):19615-20. |
Solubility Data
| Solubility (In Vitro) |
DMSO : 66 mg/mL ( 199.53 mM ) Ethanol : 20 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.56 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.56 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: 10% Ethanol + 10% Tween 80 + 80% ddH2O: 1mg/ml (3.02mM) (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0232 mL | 15.1162 mL | 30.2325 mL | |
| 5 mM | 0.6046 mL | 3.0232 mL | 6.0465 mL | |
| 10 mM | 0.3023 mL | 1.5116 mL | 3.0232 mL |