Physicochemical Properties
| Molecular Formula | C15H8CL2N2O3S |
| Molecular Weight | 367.20662021637 |
| Exact Mass | 365.963 |
| CAS # | 302826-61-5 |
| PubChem CID | 1102776 |
| Appearance | Orange to red solid powder |
| LogP | 3.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 23 |
| Complexity | 547 |
| Defined Atom Stereocenter Count | 0 |
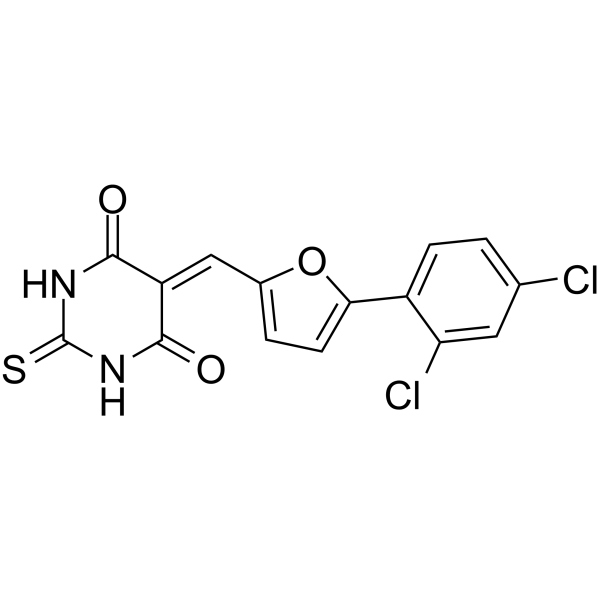
| SMILES | ClC1C=C(C=CC=1C1=CC=C(/C=C2/C(NC(NC/2=O)=S)=O)O1)Cl |
| InChi Key | QQAOFKJHOUFHKI-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C15H8Cl2N2O3S/c16-7-1-3-9(11(17)5-7)12-4-2-8(22-12)6-10-13(20)18-15(23)19-14(10)21/h1-6H,(H2,18,19,20,21,23) |
| Chemical Name | 5-[[5-(2,4-dichlorophenyl)furan-2-yl]methylidene]-2-sulfanylidene-1,3-diazinane-4,6-dione |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | EPAC1 (Ki = 1.8 μM); EPAC2 (Ki = 2.5 μM) [1] |
| ln Vitro |
One important modulator of cAMP signaling is Epac. A thiobarbituric acid variant of 5225554, EPAC 5376753 has a higher capacity to suppress Epac signaling in cells and lessen the conformational changes necessary for Epac activation. 5376753 inhibits fibroblast migration mediated by Epac in a selective manner[1]. Fluorescence Resonance Energy Transfer (FRET) assays showed that EPAC 5376753 exhibited concentration-dependent allosteric inhibition of cAMP-induced EPAC activation. At concentrations ranging from 0.1 to 10 μM, the compound significantly reduced the FRET signal change mediated by EPAC1 and EPAC2, with Ki values of 1.8 μM (EPAC1) and 2.5 μM (EPAC2), respectively. [1] Surface Plasmon Resonance (SPR) experiments demonstrated direct binding of EPAC 5376753 to the allosteric site of EPAC1, with a dissociation constant (KD) of approximately 2.1 μM, confirming the specific interaction between the compound and EPAC. [1] Molecular docking and molecular dynamics simulations revealed that EPAC 5376753 binds to a conserved allosteric pocket in EPAC, disrupting the conformational change required for cAMP-induced activation of the protein. [1] |
| Enzyme Assay |
Recombinant EPAC1 and EPAC2 catalytic domains were expressed and purified. FRET probes were constructed by fusing EPAC with cyan fluorescent protein (CFP) and yellow fluorescent protein (YFP) at specific sites. The assay mixture contained the EPAC-FRET probe, varying concentrations of EPAC 5376753, and a buffer system optimized for cAMP signaling. After incubation at 37°C for 30 minutes, cAMP was added to initiate EPAC activation, and FRET signals were measured using a fluorescence microplate reader. The inhibition rate was calculated based on the change in FRET ratio, and Ki values were determined by nonlinear regression analysis. [1] For SPR assays, purified EPAC1 protein was immobilized on a sensor chip. EPAC 5376753 was serially diluted in running buffer and injected over the chip surface at a constant flow rate. The binding response was recorded as resonance units (RU), and KD values were derived by fitting the sensorgrams to a 1:1 binding model using data analysis software. [1] |
| Cell Assay |
HeLa cells were transfected with the EPAC1-FRET reporter plasmid and cultured until reaching 70-80% confluence. The cells were treated with different concentrations of EPAC 5376753 (0.5, 2, 5, 10 μM) for 1 hour, followed by stimulation with 10 μM 8-pCPT-2'-O-Me-cAMP (a selective EPAC activator). FRET signals in live cells were monitored using confocal laser scanning microscopy, and the FRET ratio (YFP/CFP) was quantified to assess EPAC1 activation. The results showed that EPAC 5376753 inhibited cAMP-induced EPAC1 activation in cells in a concentration-dependent manner, with significant inhibition observed at concentrations ≥2 μM. [1] To evaluate the effect on downstream signaling, cells treated with EPAC 5376753 and 8-pCPT-2'-O-Me-cAMP were lysed, and the level of GTP-bound Rap1 (a downstream effector of EPAC) was detected by pull-down assay followed by immunoblotting. The assay confirmed that EPAC 5376753 reduced Rap1 activation in a concentration-dependent manner, consistent with its inhibitory effect on EPAC. [1] |
| References |
[1]. Allosteric inhibition of Epac: computational modeling and experimental validation to identify allosteric sites and inhibitors. J Biol Chem. 2014 Oct 17;289(42):29148-57. |
| Additional Infomation |
EPAC 5376753 is a small-molecule allosteric inhibitor of EPAC (Exchange Protein Directly Activated by cAMP), identified through a combination of computational modeling and experimental validation. [1] The compound binds to a unique allosteric site on EPAC, distinct from the cAMP-binding pocket, and exerts its inhibitory effect by stabilizing the inactive conformation of EPAC, thereby preventing cAMP-induced conformational rearrangement and downstream signaling activation. [1] Computational approaches including virtual screening of chemical libraries and molecular dynamics simulations were used to predict potential allosteric inhibitors, and EPAC 5376753 was selected based on its high binding affinity and predicted allosteric modulation capacity, which was subsequently validated by in vitro and cell-based assays. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7232 mL | 13.6162 mL | 27.2324 mL | |
| 5 mM | 0.5446 mL | 2.7232 mL | 5.4465 mL | |
| 10 mM | 0.2723 mL | 1.3616 mL | 2.7232 mL |