EIPA is a novel and potent blocker of Na+/H+ antiport and an inhibitor of TRPP3 channel with an IC50 of 10.5 μM.
Physicochemical Properties
| Molecular Formula | C11H18CLN7O |
| Molecular Weight | 299.76 |
| Exact Mass | 299.126 |
| Elemental Analysis | C, 44.08; H, 6.05; Cl, 11.83; N, 32.71; O, 5.34 |
| CAS # | 1154-25-2 |
| Related CAS # | EIPA hydrochloride;1345839-28-2 |
| PubChem CID | 1795 |
| Appearance | Typically exists as White to yellow solids at room temperature |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 543.9±60.0 °C at 760 mmHg |
| Melting Point | 202-205ºC(lit.) |
| Flash Point | 282.8±32.9 °C |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.665 |
| LogP | 4.86 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 20 |
| Complexity | 372 |
| Defined Atom Stereocenter Count | 0 |
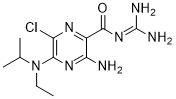
| SMILES | O=C(C1=NC(Cl)=C(N(CC)C(C)C)N=C1N)NC(N)=N |
| InChi Key | QDERNBXNXJCIQK-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H18ClN7O/c1-4-19(5(2)3)9-7(12)16-6(8(13)17-9)10(20)18-11(14)15/h5H,4H2,1-3H3,(H2,13,17)(H4,14,15,18,20) |
| Chemical Name | 3-amino-6-chloro-N-(diaminomethylidene)-5-[ethyl(propan-2-yl)amino]pyrazine-2-carboxamide |
| Synonyms | Ethyl isopropylamiloride; EIPA; 1154-25-2; 5-(N-Ethyl-N-isopropyl)amiloride; Ethylisopropylamiloride; 5-(Ethylisopropyl)amiloride; Ethyl isopropyl amiloride; 5-(N-Ethyl-N-isopropyl) Amiloride; 5-(N-ethyl-N-isopropyl)-Amiloride; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
|
|
| ln Vitro | In X, EIPA (100 μM, 30 min) inhibits the Ca2+ uptake mediated by TRPP3. Ovum laevis [1]. The basal Na+ current is reversibly inhibited by EIPA hydrochloride (10-100 μM) (IC50: 19.5 μM)[1]. In IEC-18 cells, EIPA (300 μM, 6 h) increases autophagy via NHE3 (Na+/H+-exchanger 3)[2]. EIPA (20 μM, 2 h) inhibits the large absorption of CA-PZ via macropinocytosis in both MIA PaCa-2 and HT-29 cells[3]. Zinc/kainate toxicity is attenuated by EIPA (30 μM, 3h) by reducing Zn2+ entrance in cerebellar granule neurons[4]. EIPA (5-100 μM, 48h) inhibits MKN28 cell growth by upregulating p21 expression[5]. The increase in COX-2 protein level generated by LPS is inhibited by EIPA (3 μM, 6 h)[7]. | |
| ln Vivo | EIPA (intravenous injection, 1 mg/kg) dose-dependently reduces I/R (ischemia/reperfusion)-induced renal dysfunction in ddY strain mice [6]. EIPA (orally, 10 mg/kg) inhibits LPS-induced inflammation in a balloon-type LPS-induced inflammation model [7]. | |
| Enzyme Assay | Methods: Effects of EIPA on proliferation, pH(c), [Cl(-)](c) and expression of proteins regulating cell cycle and MAPKs were studied in MKN28 expressing NHE exposed to EIPA for 48 h. Results: EIPA suppressed proliferation of MKN28 cells by causing G(0)/G(1) arrest without any significant effects on pH(c), but associated with reduction of [Cl(-)](c). Although EIPA alone had no effects on pH(c), EIPA co-applied with DIDS (an inhibitor of Cl(-)/HCO(3)(-) exchangers; i.e., anion exchanger (AE) and Na+-driven Cl(-)/HCO(3)(-) exchanger (NDCBE)) reduced pH(c), suggesting that DIDS-sensitive Cl(-)/HCO(3)(-) transporters such as AE and/or NDCBE keep pH(c) normal by stimulating HCO(3)(-) uptake coupled with Cl(-) release under an NHE-inhibited condition. EIPA-induced lowered [Cl(-)](c) up-regulated expression of p21associated with phosphorylation of MAPKs, suppressing proliferation associated with G(0)/G(1) arrest. Conclusions: EIPA suppressed proliferation of MKN28 cells through up-regulation of p21 expression via reduction of [Cl(-)](c) as a result from DIDS-sensitive Cl(-)/HCO(3)(-) exchanger-mediated compensation for keeping pH(c) normal under an NHE-inhibited condition. This is the first study revealing that an NHE inhibitor suppressed the proliferation of cancer cells by reducing [Cl(-)](c) but not pH(c)[5]. | |
| Cell Assay |
Cell Proliferation Assay[5] Cell Types: MKN28 cells Tested Concentrations: 5, 10, 25, 50, and 100 μM Incubation Duration: 48 h Experimental Results: Inhibited cell proliferation in a dose- and time-dependent manner. Western Blot Analysis[2] Cell Types: IEC-18 cells Tested Concentrations: 300 μM Incubation Duration: 6 h Experimental Results: Increased total LC3-II protein levels and P62 flux. Increased ATG5, 7, 12 and P62 expression. |
|
| Animal Protocol |
Animal/Disease Models: Male ddY strain mouse [6] Doses: 1 mg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results: Reduce histological renal damage and improve the increase in renal ET-1 content caused by I/R. Animal/Disease Models: air bag type LPS-induced inflammation model [7] Doses: 10 mg/kg Route of Administration: Oral Experimental Results:Inhibited LPS-induced leukocyte infiltration into air bags. Suppresses the amount of PGE2 in ostomy bag fluid. |
|
| References |
[1]. Inhibition of TRPP3 channel by MK-870 and analogs. Mol Pharmacol. 2007 Dec;72(6):1576-85. [2]. Na+/H+ Exchanger Regulates Amino Acid-Mediated Autophagy in Intestinal Epithelial Cells. Cell Physiol Biochem. 2017;42(6):2418-2429. [3]. A new HDAC inhibitor cinnamoylphenazine shows antitumor activity in association with intensive macropinocytosis. [4]. Acidosis and 5-(N-ethyl-N-isopropyl)amiloride (EIPA) Attenuate Zinc/Kainate Toxicity in Cultured Cerebellar Granule Neurons. Biochemistry (Mosc). 2015 Aug;80(8):1065-72. [5]. An inhibitor of Na(+)/H(+) exchanger (NHE), ethyl-isopropyl amiloride (EIPA), diminishes proliferation of MKN28 human gastric cancer cells by decreasing the cytosolic Cl(-) concentration via DIDS-sensitive pathways. Cell Physiol Biochem. 2012;30(5):1241-53. [6]. Role of Na+/H+ exchanger in the pathogenesis of ischemic acute renal failure in mice. J Cardiovasc Pharmacol. 2007 Mar;49(3):154-60. [7]. Inhibition of lipopolysaccharide-induced prostaglandin E2 production and inflammation by the Na+/H+ exchanger inhibitors. J Pharmacol Exp Ther. 2007 Apr;321(1):345-52. |
|
| Additional Infomation | Ethylisopropylamiloride is a member of the class of pyrazines that is amiloride in which the amino substitutent of the pyrazine ring that is adjacent to the chloro substituent has been substituted by an ethyl group and by an isopropyl group. It has a role as an anti-arrhythmia drug, a neuroprotective agent and a sodium channel blocker. It is a member of guanidines, an aromatic amine, an organochlorine compound, a tertiary amino compound, a member of pyrazines and a monocarboxylic acid amide. It is functionally related to an amiloride. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~50 mg/mL (~166.80 mM) H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.34 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3360 mL | 16.6800 mL | 33.3600 mL | |
| 5 mM | 0.6672 mL | 3.3360 mL | 6.6720 mL | |
| 10 mM | 0.3336 mL | 1.6680 mL | 3.3360 mL |