Physicochemical Properties
| Molecular Formula | C24H29CL2N9O6 |
| Molecular Weight | 610.449762105942 |
| Exact Mass | 609.161 |
| Elemental Analysis | C, 47.22; H, 4.79; Cl, 11.61; N, 20.65; O, 15.73 |
| CAS # | 1190332-25-2 |
| Related CAS # | EB-47;366454-36-6 |
| PubChem CID | 9986189 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 41 |
| Complexity | 951 |
| Defined Atom Stereocenter Count | 4 |
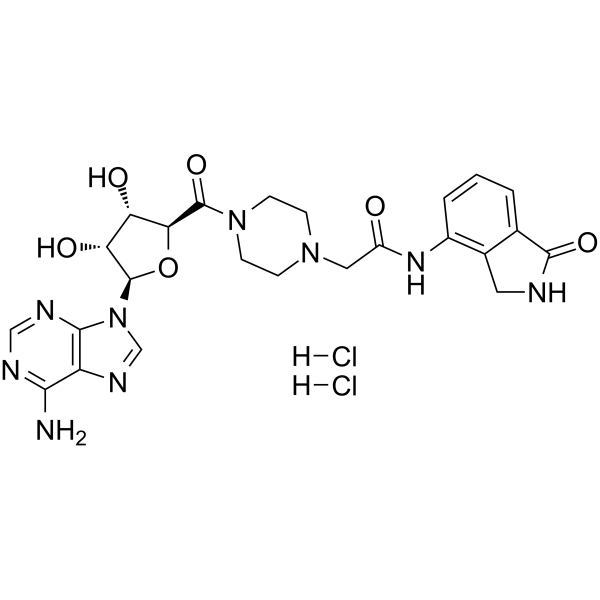
| SMILES | Cl.Cl.O1[C@@H]([C@H]([C@H]([C@H]1C(N1CCN(CC(NC2C=CC=C3C(NCC=23)=O)=O)CC1)=O)O)O)N1C=NC2C(N)=NC=NC1=2 |
| InChi Key | VVMQSDIMNDTMII-LLGQWWOSSA-N |
| InChi Code | InChI=1S/C24H27N9O6.2ClH/c25-20-16-21(28-10-27-20)33(11-29-16)24-18(36)17(35)19(39-24)23(38)32-6-4-31(5-7-32)9-15(34)30-14-3-1-2-12-13(14)8-26-22(12)37;;/h1-3,10-11,17-19,24,35-36H,4-9H2,(H,26,37)(H,30,34)(H2,25,27,28);2*1H/t17-,18?,19+,24-;;/m1../s1 |
| Chemical Name | 2-[4-[(2S,3R,5R)-5-(6-aminopurin-9-yl)-3,4-dihydroxyoxolane-2-carbonyl]piperazin-1-yl]-N-(1-oxo-2,3-dihydroisoindol-4-yl)acetamide;dihydrochloride |
| Synonyms | EB-47 2HCl; EB-47 dihydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | EB-47 dihydrochloride has an IC50 value of 0.86 for CdPARP and 1.0 μM for HsPARP, respectively, and can inhibit both CdPARP and HsPARP by more than 50% [1]. |
| ln Vivo | EB-47 diHClide (2 μM; 5 days) lowers the amount of cells implanting and growing embryos on day 5. PARP1 is involved in the cell implantation process [3]. |
| References |
[1]. Evaluation and Structural Basis for the Inhibition of Tankyrases by PARP Inhibitors.ACS Med Chem Lett. 2013 Nov 20;5(1):18-22. [2]. Comparative inhibitory profile and distribution of bacterial PARPs, using Clostridioides difficile CD160 PARP as a model. Sci Rep. 2018 May 23;8(1):8056. [3]. The discovery and synthesis of novel adenosine substituted 2,3-dihydro-1H-isoindol-1-ones: potent inhibitors of poly(ADP-ribose) polymerase-1 (PARP-1).Bioorg Med Chem Lett. 2004 Jan 5;14(1):81-5. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~62.5 mg/mL (~102.38 mM) H2O : ~33.33 mg/mL (~54.60 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.10 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.10 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.10 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 2.94 mg/mL (4.82 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6381 mL | 8.1907 mL | 16.3814 mL | |
| 5 mM | 0.3276 mL | 1.6381 mL | 3.2763 mL | |
| 10 mM | 0.1638 mL | 0.8191 mL | 1.6381 mL |