Physicochemical Properties
| Molecular Formula | C44H43N3O9 |
| Molecular Weight | 757.83 |
| Exact Mass | 757.2999 |
| CAS # | 114784-59-7 |
| PubChem CID | 6610268 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.3±0.1 g/cm3 |
| Index of Refraction | 1.613 |
| LogP | 7.6 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 56 |
| Complexity | 1320 |
| Defined Atom Stereocenter Count | 2 |
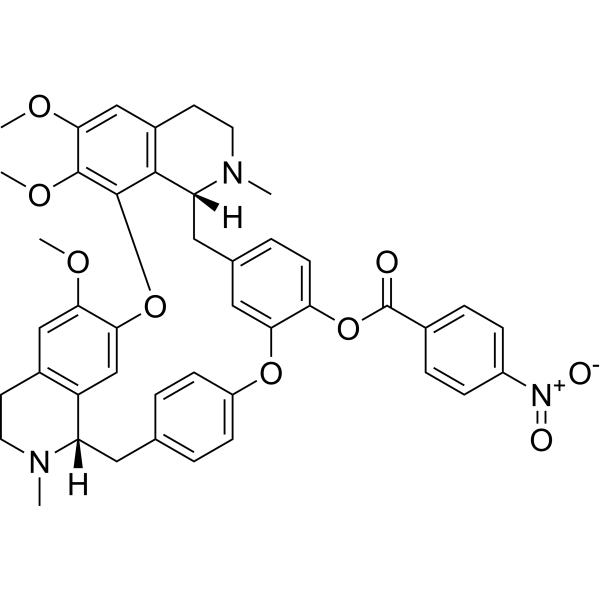
| SMILES | O1C2C(=CC3CCN(C)[C@@H](CC4C=CC(=CC=4)OC4C(=CC=C(C=4)C[C@@H]4C5C1=C(C(=CC=5CCN4C)OC)OC)OC(C1C=CC(=CC=1)[N+](=O)[O-])=O)C=3C=2)OC |
| InChi Key | PUAVTDKYTXNVBU-OIDHKYIRSA-N |
| InChi Code | InChI=1S/C44H43N3O9/c1-45-18-16-29-23-37(51-3)39-25-33(29)34(45)20-26-6-13-32(14-7-26)54-38-22-27(8-15-36(38)56-44(48)28-9-11-31(12-10-28)47(49)50)21-35-41-30(17-19-46(35)2)24-40(52-4)42(53-5)43(41)55-39/h6-15,22-25,34-35H,16-21H2,1-5H3/t34-,35+/m0/s1 |
| Chemical Name | [(1S,14R)-20,21,25-trimethoxy-15,30-dimethyl-7,23-dioxa-15,30-diazaheptacyclo[22.6.2.23,6.18,12.114,18.027,31.022,33]hexatriaconta-3(36),4,6(35),8,10,12(34),18,20,22(33),24,26,31-dodecaen-9-yl] 4-nitrobenzoate |
| Synonyms | E6 berbamine; 114784-59-7; CHEMBL405686; [(1S,14R)-20,21,25-trimethoxy-15,30-dimethyl-7,23-dioxa-15,30-diazaheptacyclo[22.6.2.23,6.18,12.114,18.027,31.022,33]hexatriaconta-3(36),4,6(35),8,10,12(34),18,20,22(33),24,26,31-dodecaen-9-yl] 4-nitrobenzoate; 3,4,4aS,5,16aR,17,18,19-octahydro-21,22,26-trimethoxy-4,17-dimethyl-16H-1,24:6,9-dietheno-11,15-metheno-2H-pyrido[2',3':17,18][1,11]dioxacycloeicosino[2,3,4-ij]isoquinolin-12-ol-4-nitrobenzoate; Bio1_000778; Calmodulin inhibitor; CBiol_002003; |
Biological Activity
| Targets | CaM (calmodulin) |
| ln Vitro | The interaction of the berbamine compound E6 and calmodulin (CaM)-dependent myosin light chain kinase (MLCK) has been studied. The experimental results showed that the inhibition of MLCK activity was increased with increasing amounts of E6 and was overcome completely by the addition of excessive CaM. The stimulatory activity of MLCK induced by CaM was gradually inhibited by the increasing concentrations of compound E6, showing that the inhibition of MLCK activity by compound E6 was concentration dependent; and the Ki was 0.95 microM. Compound E6 diminished the fluorescence intensity of dansyl-labeled CaM and the intensity was increased gradually by the addition of different amounts of CaM. Compound E6 had no effect on the activity of MLCK fragments produced by limited trypsinization, and it is a novel and considerably potent calmodulin antagonist.[1] |
| References |
[1]. Interaction of berbamine compound E6 and calmodulin-dependent myosin light chain kinase. Biochem Pharmacol. 1992 Oct 20;44(8):1543-7. [2]. Berbamine derivatives: a novel class of compounds for anti-leukemia activity. Eur J Med Chem. 2009 Aug;44(8):3293-8. |
| Additional Infomation | Our previous studies showed that the natural compound berbamine, from Chinese herb Berberis amurensis, selectively induces apoptosis of imatinib (IM)-resistant-Bcr/Abl-expressing leukemia cells from the K562 cell line and CML patients. Here, a series of new berbamine derivatives were obtained by synthesis. In this series, high to very high activity in vitro has been found. Compounds 2e, 2g, 3f, 3k, 3q and 3u exhibited consistent high anti-tumor activity for imatinib-resistant K562 leukemia cells. Their IC(50) values at 48h were 0.36-0.55 microM, whereas berbamine IC(50) value was 8.9 microM. Cell cycle analysis results showed that compound 3h could reduce G0/G1 cells. In particular, these compounds displayed potent inhibition of the cytoplasm-to-nucleus translocation of NF-kappaB p65 which plays a critical role in the survival of leukemia stem cells. These results suggest that berbamine could be a good starting point for the development of novel lead compounds in the fight against leukemia.[2] |
Solubility Data
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3196 mL | 6.5978 mL | 13.1956 mL | |
| 5 mM | 0.2639 mL | 1.3196 mL | 2.6391 mL | |
| 10 mM | 0.1320 mL | 0.6598 mL | 1.3196 mL |