Physicochemical Properties
| Molecular Formula | C11H13NO4 |
| Molecular Weight | 223.225223302841 |
| Exact Mass | 223.084 |
| Elemental Analysis | C, 59.19; H, 5.87; N, 6.27; O, 28.67 |
| CAS # | 170846-89-6 |
| PubChem CID | 3937355 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 431.3±40.0 °C at 760 mmHg |
| Flash Point | 214.7±27.3 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.594 |
| LogP | 1.5 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 16 |
| Complexity | 284 |
| Defined Atom Stereocenter Count | 0 |
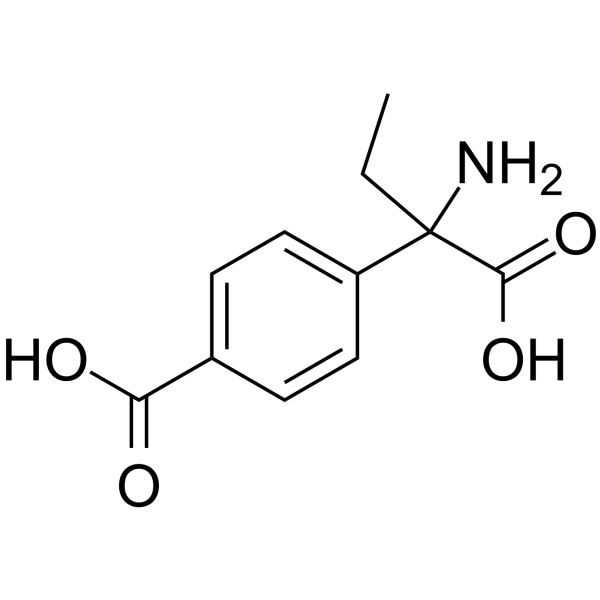
| SMILES | NC(C(O)=O)(CC)C1=CC=C(C(O)=O)C=C1 |
| InChi Key | AIEFWRHRHFRLFT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H13NO4/c1-2-11(12,10(15)16)8-5-3-7(4-6-8)9(13)14/h3-6H,2,12H2,1H3,(H,13,14)(H,15,16) |
| Chemical Name | 4-(1-amino-1-carboxypropyl)benzoic acid |
| Synonyms | E4CPG; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Rat cortical mGluR is affected by E4CPG, which has a KB value of 0.367 mM[3]. |
| ln Vitro |
Rat cortical mGluR is affected by E4CPG, which has a KB value of 0.367 mM[3]. (RS)-α-ethyl-4-carboxyphenylglycine (ECPG) (tested at 1 mM) showed antagonist activity against L-glutamate (10 µM)-induced inositol phosphate (IP) formation in Chinese hamster ovary (CHO) cells expressing mGluR1, partially inhibiting the response. [1] At the same concentration (1 mM), ECPG also partially antagonized the L-glutamate (20 µM)-mediated inhibition of forskolin-stimulated cyclic AMP formation in CHO cells expressing mGluR2. [1] ECPG (1 mM) showed no agonist or antagonist activity on mGluR6 expressed in CHO cells. [1] |
| ln Vivo | Significant inhibition of glutamate injection (i.pl.) induced nociception was observed with E4CPG (3-30 nmol/site (it), 1-10 μmol/paw (i.pl.), and 1-10 nmol/site (icv)). The maximum inhibitory values of E4CPG's antinociception in glutamate-induced nociception were 48% (i.pl.), 49% (it), and 40% (icv) [4]. In male Sprague-Dawley rats, long-term depression (LTD) induced by the group I mGluR agonist dihydroxyphenylglycine (DHPG, 100 nM/5 μL, icv) is completely blocked by E4CPG (35 nM/3.5 μL, icv) [ 5]. |
| Cell Assay |
For agonist/antagonist activity determination on mGluR1, CHO cells stably expressing mGluR1 were used. Cells were incubated with test compounds (e.g., 1 mM ECPG) for 20 minutes, followed by co-incubation with L-glutamate (10 µM) in the continued presence of the test compound for another 20 minutes. Total inositol phosphate formation was then measured as an indicator of receptor activity. [1] For agonist/antagonist activity determination on mGluR2 and mGluR6, CHO cells stably expressing these receptors were used. Cells were preincubated with test compounds (e.g., 1 mM ECPG) for 20 minutes, followed by co-incubation with L-glutamate (20 µM for mGluR2, 30 or 100 µM for mGluR6) and forskolin (10 µM) in the continued presence of the test compound for 10 minutes. Intracellular cyclic AMP levels were then measured. [1] Control experiments using endothelin receptor ETA-expressing cells (for IP assay) and vector-transfected cells (for cyclic AMP assay) confirmed that the effects of active compounds (including ECPG) were mediated via mGluRs and not through direct interference with intracellular signaling machinery. [1] |
| Animal Protocol |
Animal/Disease Models: Male Swiss mouse (25-35 g) [4] Doses: 3-30 nmol/site (it), 1-10 μmol/paw (i.pl.) and 1-10 nmol/site ( icv): Single injection Experimental Results: Maximum inhibition of the antinociceptive effect of E4CPG in glutamate-induced nociception was 48% (i.pl.), 49% (it) and 40% (icv). Animal/Disease Models: Male SD (SD (Sprague-Dawley)) rat [5] Doses: 35 nM/3.5 μL Route of Administration: single injection, intravenous (iv) (iv)injection Experimental Results:Complete blockade of EGFR induced by the group I mGluR agonist dihydroxyphenylglycine (DHPG) LTD. |
| References |
[1]. Structure-activity relationships of new agonists and antagonists of different metabotropic glutamate receptorsubtypes. Br J Pharmacol, 1996 Apr, 117(7):1493-503. [2]. GABAB receptor- and metabotropic glutamate receptor-dependent cooperative long-term potentiation of rat hippocampal GABAA synaptic transmission. J Physiol. 2003 Nov 15;553(Pt 1):155-67. [3]. Structure-activity relationships for a series of phenylglycine derivatives acting at metabotropic glutamate receptors (mGluRs). Br J Pharmacol. 1995 Dec;116(8):3323-9. [4]. Mechanisms underlying the nociception and paw oedema caused by injection of glutamate into the mouse paw. Brain Res. 2002 Jan 11;924(2):219-28. [5]. Acute cannabinoids impair working memory through astroglial CB1 receptor modulation of hippocampal LTD. Cell. 2012 Mar 2;148(5):1039-50. |
| Additional Infomation |
(RS)-α-ethyl-4-carboxyphenylglycine (ECPG) is a phenylglycine derivative. Its parent compound, (S)-4-carboxyphenylglycine (4CPG), is an mGluR2 agonist and mGluR1 antagonist. α-Ethylation of 4CPG (resulting in ECPG) converts its agonist activity at mGluR2 to antagonist activity, making it an antagonist at both mGluR1 and mGluR2. [1] The study demonstrates that substitution at the α-carbon (methylation or ethylation) of carboxyphenylglycine derivatives plays a critical role in determining agonist vs. antagonist properties at mGluR2. [1] The antagonist activity of ECPG against (1S,3R)-ACPD-stimulated IP formation in rat cerebral cortical slices has been reported elsewhere and is consistent with its mGluR1 antagonist profile identified in this recombinant cell system. [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~10 mg/mL (~44.80 mM) H2O : ~1 mg/mL (~4.48 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.4797 mL | 22.3984 mL | 44.7968 mL | |
| 5 mM | 0.8959 mL | 4.4797 mL | 8.9594 mL | |
| 10 mM | 0.4480 mL | 2.2398 mL | 4.4797 mL |