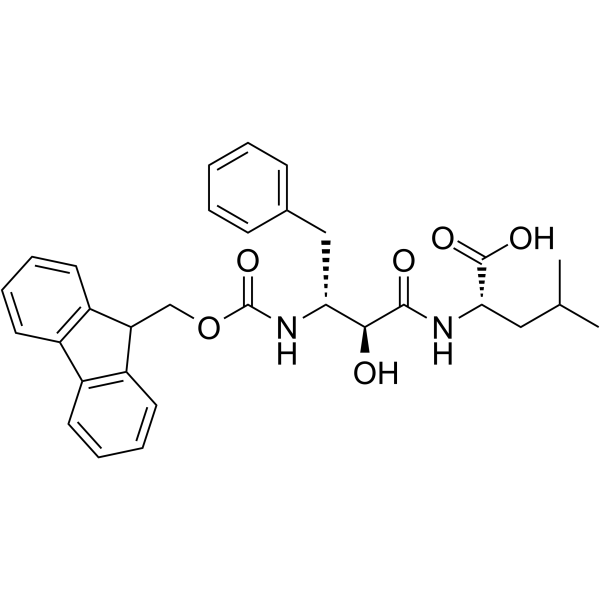
Physicochemical Properties
| Molecular Formula | C31H34N2O6 |
| Molecular Weight | 530.6115 |
| Exact Mass | 530.241 |
| CAS # | 1225383-33-4 |
| PubChem CID | 138911308 |
| Appearance | White to off-white solid powder |
| LogP | 5.1 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 39 |
| Complexity | 806 |
| Defined Atom Stereocenter Count | 3 |
| SMILES | O(C(N([H])[C@]([H])(C([H])([H])C1C([H])=C([H])C([H])=C([H])C=1[H])[C@@]([H])(C(N([H])[C@]([H])(C(=O)O[H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])[H])=O)O[H])=O)C([H])([H])C1([H])C2=C([H])C([H])=C([H])C([H])=C2C2=C([H])C([H])=C([H])C([H])=C12 |
| InChi Key | DYKHBFJZCIEBJE-PKTNWEFCSA-N |
| InChi Code | InChI=1S/C31H34N2O6/c1-19(2)16-27(30(36)37)32-29(35)28(34)26(17-20-10-4-3-5-11-20)33-31(38)39-18-25-23-14-8-6-12-21(23)22-13-7-9-15-24(22)25/h3-15,19,25-28,34H,16-18H2,1-2H3,(H,32,35)(H,33,38)(H,36,37)/t26-,27+,28+/m1/s1 |
| Chemical Name | (2S)-2-[[(2S,3R)-3-(9H-fluoren-9-ylmethoxycarbonylamino)-2-hydroxy-4-phenylbutanoyl]amino]-4-methylpentanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
cellular inhibitor of apoptosis protein 1 (cIAP1) [1] |
| ln Vitro |
Compound 24 (an LCL-161 derivative), when conjugated as the E3 ligase ligand in SNIPER(AR)-31 (compound 23), showed superior activity in inducing androgen receptor (AR) protein degradation compared to SNIPERs incorporating bestatin (79) or MV-1. In 22Rv1 prostate cancer cells, SNIPER(AR)-31 (containing compound 24) effectively reduced AR protein levels at concentrations of 3 μM and above, whereas SNIPERs with bestatin or MV-1 required 30 μM and 20 μM, respectively, for similar effects. [1] The degradation of AR protein by SNIPER(AR)s (which recruit cIAP1 via ligands like compound 24) is proteasome-dependent, as co-treatment with the proteasome inhibitor MG-132 (77) abolished AR reduction in 22Rv1 and VCaP cells. [1] SNIPER(AR)-51 (compound 42a), which also contains compound 24 as the E3 ligase ligand, induced caspase-3 activation and PARP cleavage in VCaP cells at 3 μM, indicating induction of apoptosis. This effect was blocked by the pan-caspase inhibitor zVAD-FMK (78). [1] SNIPER(AR)-51 (42a) at 3 μM suppressed the expression of AR-regulated genes (PSA, TMPRSS2, KLK2, NKX3.1) in VCaP cells stimulated with 1 nM dihydrotestosterone (DHT). [1] SNIPER(AR)-51 (42a) at 3 μM inhibited the proliferation of androgen-dependent prostate cancer cell lines VCaP and LNCaP but did not inhibit the growth of androgen-independent cell lines PC-3, A549, or HT1080 under the same conditions. At 100 μM, it reduced viability across all cell lines, indicating potential toxicity at high concentrations. [1] |
| Cell Assay |
Western Blotting for AR Protein Degradation: Human prostate cancer cells (22Rv1, VCaP) were treated with graded concentrations of SNIPER(AR) compounds (e.g., 23, 42a) containing compound 24 for specified times (e.g., 6 h). Cells were lysed with SDS lysis buffer. Protein concentration was measured, and equal amounts of lysate were separated by SDS-PAGE, transferred to PVDF membranes, and probed with anti-AR antibody. GAPDH was used as a loading control. Signals were detected and quantified. [1] Cell Viability Assay (WST-8): Cells were seeded in 96-well plates. After 24 h, they were treated with indicated concentrations of SNIPER(AR) compounds (e.g., 42a) for 48 h. The WST-8 reagent was added, and cells were incubated for 0.5 h. Absorbance at 450 nm was measured. [1] Apoptosis Analysis by Flow Cytometry: After treatment with compounds, cells were harvested, resuspended in binding buffer, and stained with Annexin V-FITC and propidium iodide (PI) for 5 min in the dark. Samples were analyzed by flow cytometry to distinguish live, apoptotic, and necrotic cells. [1] Quantitative PCR for Gene Expression: Total RNA was extracted from treated cells. cDNA was synthesized using an oligo-dT primer. Quantitative real-time PCR was performed using SYBR GreenER and gene-specific primers for AR-regulated genes (PSA, TMPRSS2, KLK2, NKX3.1). Human 36B4 mRNA served as an invariant control. Relative mRNA levels were calculated using the comparative Ct method. [1] |
| Toxicity/Toxicokinetics |
In cell-based assays, SNIPER(AR)-51 (42a, containing compound 24) at a high concentration (100 μM for 6 h) seriously reduced the viability of all tested cell lines (VCaP, LNCaP, PC-3, A549, HT1080), suggesting compound toxicity at high doses. [1] |
| References |
[1]. Development of Protein Degradation Inducers of Androgen Receptor by Conjugation of Androgen Receptor Ligands and Inhibitor of Apoptosis Protein Ligands. J Med Chem. 2018 Jan 25;61(2):543-575. |
| Additional Infomation |
Compound 24 is a derivative of the IAP antagonist LCL-161 and functions as a ligand for cellular inhibitor of apoptosis protein 1 (cIAP1), an E3 ubiquitin ligase. [1] It is used as a component in the design of Specific and Nongenetic IAP-dependent Protein Erasers (SNIPERs), which are heterobifunctional molecules that link a target protein ligand (e.g., an AR antagonist) to an E3 ligase ligand (compound 24) via a linker. These molecules promote ubiquitination and subsequent proteasomal degradation of the target protein. [1] Compared to other IAP ligands like bestatin (79) or MV-1 tested in the study, incorporation of compound 24 into SNIPER(AR) hybrids conferred the greatest AR protein degradation activity. [1] SNIPERs, by recruiting cIAP1, can induce the degradation of both the target protein (POI) and cIAP1 itself. As cIAP1 is an anti-apoptotic protein often overexpressed in tumors, its simultaneous degradation with the POI may be beneficial for killing cancer cells. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~188.46 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.71 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8846 mL | 9.4231 mL | 18.8462 mL | |
| 5 mM | 0.3769 mL | 1.8846 mL | 3.7692 mL | |
| 10 mM | 0.1885 mL | 0.9423 mL | 1.8846 mL |