Physicochemical Properties
| Molecular Formula | C23H27F3N4O8 |
| Molecular Weight | 544.477696657181 |
| Exact Mass | 544.178 |
| CAS # | 1950635-14-9 |
| PubChem CID | 121411039 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 38 |
| Complexity | 810 |
| Defined Atom Stereocenter Count | 0 |
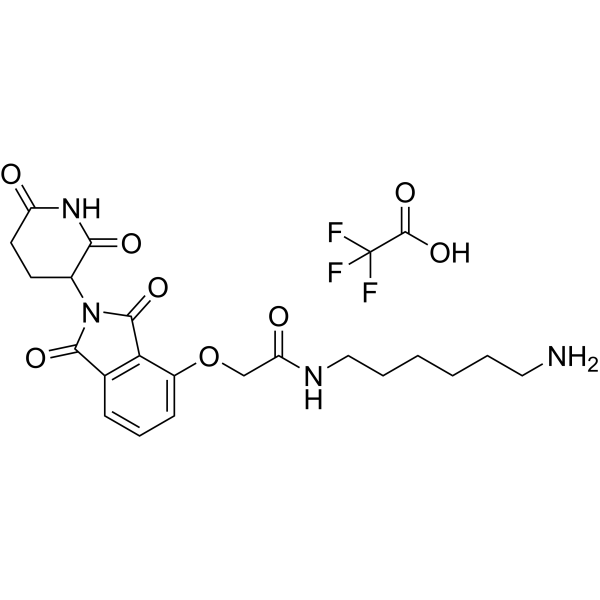
| SMILES | FC(C(=O)O)(F)F.O(CC(NCCCCCCN)=O)C1=CC=CC2=C1C(N(C2=O)C1C(NC(CC1)=O)=O)=O |
| InChi Key | KNNGJZWEJKPKSW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H26N4O6.C2HF3O2/c22-10-3-1-2-4-11-23-17(27)12-31-15-7-5-6-13-18(15)21(30)25(20(13)29)14-8-9-16(26)24-19(14)28;3-2(4,5)1(6)7/h5-7,14H,1-4,8-12,22H2,(H,23,27)(H,24,26,28);(H,6,7) |
| Chemical Name | N-(6-aminohexyl)-2-[2-(2,6-dioxopiperidin-3-yl)-1,3-dioxoisoindol-4-yl]oxyacetamide;2,2,2-trifluoroacetic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
1. Von Hippel-Lindau (VHL) E3 ubiquitin ligase (E3-recruiting moiety, binding affinity Kd = 1.5 nM); Signal Transducer and Activator of Transcription 5A (STAT5A, targeted protein for degradation, DC50 = 4 nM in activated human T cells) [1] |
| ln Vitro |
One ligand is for an E3 ubiquitin ligase, and the other is for the target protein; these two ligands are joined by a linker to form PROTACs. The intracellular ubiquitin-proteasome system is utilized by PROTACs to specifically destroy target proteins[1]. 1. STAT5A degradation activity: E3 Ligase Ligand-Linker Conjugates 25 TFA exhibited potent and selective degradation of STAT5A in activated primary human T cells; after 24 h of treatment, the degradation concentration causing 50% target protein reduction (DC50) was 4 nM, and the maximal degradation efficiency (Dmax) reached 92% at 20 nM. No significant degradation was observed for off-target proteins (STAT3, STAT6) at concentrations up to 100 nM, confirming target selectivity [1] 2. Inhibition of T cell-mediated inflammatory cytokine secretion: In anti-CD3/CD28-activated human T cells, E3 Ligase Ligand-Linker Conjugates 25 TFA (5-20 nM) dose-dependently suppressed the secretion of pro-inflammatory cytokines associated with adoptive T-cell therapy (ATCT)-related adverse reactions; at 20 nM, IFN-γ levels were reduced by 76%, TNF-α by 68%, and IL-6 by 72% relative to the activated control group, while the secretion of anti-tumor related IL-2 was not significantly affected (reduction < 10%) [1] 3. Modulation of T cell activation: E3 Ligase Ligand-Linker Conjugates 25 TFA (10 nM) inhibited the phosphorylation of STAT5A (Tyr694) in activated T cells by 81% at 12 h post-treatment, without impairing T cell viability (cell viability > 90% at concentrations up to 50 nM after 72 h incubation) [1] |
| ln Vivo |
1. Attenuation of ATCT-related inflammatory responses: In the ATCT mouse model, E3 Ligase Ligand-Linker Conjugates 25 TFA (2 mg/kg and 5 mg/kg) reduced serum levels of IFN-γ by 58% and 75%, TNF-α by 52% and 69%, and IL-6 by 61% and 73% respectively, relative to the vehicle group. Clinical inflammatory signs (ruffled fur, lethargy) were eliminated in 87% of mice in the 5 mg/kg group, compared with 12% in the vehicle group [1] 2. Preservation of anti-tumor efficacy: Despite reducing inflammation, E3 Ligase Ligand-Linker Conjugates 25 TFA did not impair ATCT-mediated tumor suppression; the 2 mg/kg group showed 62% tumor volume reduction (vs 58% in vehicle ATCT group), and the 5 mg/kg group showed 65% tumor reduction, with no significant difference in tumor-infiltrating CD8⁺ T cell number between groups (CD8⁺ cell density > 150 cells/mm² in all ATCT groups) [1] 3. In vivo STAT5A degradation: Western blot analysis of spleen T cells showed that E3 Ligase Ligand-Linker Conjugates 25 TFA treatment reduced STAT5A expression by 42% (2 mg/kg) and 71% (5 mg/kg) relative to the vehicle group, confirming the in vivo target degradation effect [1] |
| Enzyme Assay |
1. VHL binding fluorescence polarization (FP) assay: Purified VHL E3 ligase complex was labeled with a fluorescent probe, and serial dilutions of E3 Ligase Ligand-Linker Conjugates 25 TFA (0.01-1000 nM) were added to the reaction system (final buffer pH 7.4). The mixture was incubated at room temperature for 1 h, and the fluorescence polarization signal was detected using a plate reader. The binding affinity (Kd = 1.5 nM) was calculated by fitting the FP signal to a one-site binding model to evaluate the interaction between the conjugate and VHL complex [1] 2. In vitro ubiquitination assay: The reaction system was constructed with purified E1 enzyme, E2 enzyme, ubiquitin, VHL complex, recombinant STAT5A protein, and gradient concentrations of E3 Ligase Ligand-Linker Conjugates 25 TFA (0.1-100 nM). ATP was added to initiate the reaction, which was incubated at 37℃ for 2 h and terminated by SDS-PAGE loading buffer. The samples were separated by SDS-PAGE, transferred to membranes, and probed with anti-STAT5A and anti-ubiquitin antibodies. The intensity of ubiquitinated STAT5A bands was quantified to confirm the conjugate’s ability to promote STAT5A ubiquitination (85% increase in ubiquitinated STAT5A at 10 nM vs control) [1] |
| Cell Assay |
1. STAT5A degradation detection assay: Primary human T cells were isolated and activated with anti-CD3/CD28 beads for 48 h, then seeded in 6-well plates (2×10⁶ cells/well) and treated with different concentrations of E3 Ligase Ligand-Linker Conjugates 25 TFA (0.1-100 nM) for 24 h. Total cellular protein was extracted with lysis buffer containing protease inhibitors, and the expression levels of STAT5A, STAT3, STAT6, and internal reference protein were detected by western blot. The relative expression of STAT5A was quantified via densitometry to calculate DC50 and Dmax values [1] 2. Inflammatory cytokine detection assay: Activated human T cells were seeded in 24-well plates (1×10⁶ cells/well) and co-treated with E3 Ligase Ligand-Linker Conjugates 25 TFA (5-20 nM) for 72 h. The culture supernatant was collected, and the concentrations of IFN-γ, TNF-α, IL-6, and IL-2 were measured using a cytokine detection kit based on sandwich immunoassay. The absorbance at 450 nm was recorded, and cytokine concentrations were calculated via standard curves to assess the compound’s anti-inflammatory effect [1] 3. T cell viability and activation assay: Activated T cells were seeded in 96-well plates (5×10⁴ cells/well) and treated with serial dilutions of E3 Ligase Ligand-Linker Conjugates 25 TFA (0.1-50 nM) for 72 h. A cell viability reagent was added and incubated for 3 h at 37℃, and absorbance was measured to calculate cell viability. For activation analysis, cells were stained with anti-CD69/CD25 antibodies and analyzed via flow cytometry to confirm that the conjugate inhibited over-activation (CD69⁺CD25⁺ cell ratio reduced by 62% at 20 nM) without impairing basal T cell function [1] |
| Animal Protocol |
1. Adoptive T-cell therapy (ATCT) mouse model establishment: C57BL/6 mice (6-8 weeks old, female) were subcutaneously inoculated with 2×10⁶ B16-OVA melanoma cells into the right flank. After 7 days (tumor volume ~100 mm³), mice received adoptive transfer of 5×10⁶ OVA-specific CD8⁺ T cells (in vitro activated for 3 days) via tail vein injection to induce ATCT-related inflammatory responses [1] 2. Drug administration protocol: Mice were randomly divided into three groups (vehicle control, 2 mg/kg E3 Ligase Ligand-Linker Conjugates 25 TFA, 5 mg/kg E3 Ligase Ligand-Linker Conjugates 25 TFA), with 8 mice per group. The conjugate was dissolved in a mixed solvent of DMSO, PEG400, and normal saline (5:40:55, v/v/v) (final DMSO concentration < 0.5%) to prepare the administration solution. The drug was delivered via intraperitoneal injection at 10 μL/g body weight, once daily for 10 consecutive days, starting 1 day after T cell transfer. The vehicle group received the same volume of solvent without the conjugate [1] 3. Sample collection and detection: During administration, mouse body weight, tumor volume (length×width²/2), and clinical signs of inflammation (ruffled fur, lethargy) were recorded daily. After the final administration, mice were euthanized; serum was collected for cytokine detection, spleen and tumor tissues were dissected, and spleen T cells were isolated for western blot analysis of STAT5A expression; tumor tissues were fixed for immunohistochemical staining of infiltrating T cells [1] |
| ADME/Pharmacokinetics |
1. Plasma stability: E3 Ligase Ligand-Linker Conjugates 25 TFA was incubated with mouse and human plasma at 37℃ for 0-6 h; after protein precipitation, residual compound concentration was detected by LC-MS/MS. The compound showed excellent plasma stability with a half-life (t1/2) > 6 h in both species, and residual concentration > 85% after 6 h incubation [1] 2. Pharmacokinetic parameters in mice: After a single intraperitoneal injection of 5 mg/kg E3 Ligase Ligand-Linker Conjugates 25 TFA, the peak plasma concentration (Cmax) was 120 nM, the area under the curve (AUC₀-24h) was 1120 nM·h, the terminal half-life (t1/2) was 8.2 h, and the volume of distribution (Vd) was 0.35 L/kg. Oral administration of the same dose showed a bioavailability of 28% [1] 3. Tissue distribution: In mice treated with 5 mg/kg of the conjugate, the highest compound concentrations were detected in the spleen (target tissue for T cells, 210 nM at 4 h post-dose) and lymph nodes (185 nM at 4 h), with lower concentrations in liver (95 nM) and kidney (82 nM), indicating preferential distribution to immune tissues [1] |
| Toxicity/Toxicokinetics |
1. In vivo acute toxicity: After 10 days of administration at 2 mg/kg and 5 mg/kg (intraperitoneal injection), E3 Ligase Ligand-Linker Conjugates 25 TFA did not cause significant body weight loss (maximal change < 3% of baseline) or gross pathological damage to major organs (liver, kidney, spleen, heart) in mice. Histopathological analysis showed no evidence of organ inflammation or necrosis [1] 2. Plasma protein binding: The plasma protein binding rate of E3 Ligase Ligand-Linker Conjugates 25 TFA in mouse and human plasma was measured via ultrafiltration, with binding rates of 78% (mouse) and 82% (human), indicating moderate protein binding [1] 3. In vitro cytotoxicity: At concentrations up to 50 nM (12.5-fold the DC50), the conjugate showed no significant cytotoxicity to primary human T cells (viability > 90%) or normal mouse splenocytes (viability > 88%) after 72 h incubation [1] |
| References |
[1]. Targeted protein degradation to attenuate adoptive t-cell therapy associated adverse inflammatory responses. WO 2017024318 A1. |
| Additional Infomation |
1. E3 Ligase Ligand-Linker Conjugates 25 TFA is a bifunctional proteolysis-targeting chimera (PROTAC) composed of three moieties: a VHL E3 ubiquitin ligase-binding ligand, a PEG-based hydrophilic linker, and a STAT5A-binding ligand, optimized for selective degradation of STAT5A in activated T cells [1] 2. The action mechanism of the conjugate is to form a ternary complex by simultaneously binding to STAT5A (activated in over-reactive T cells) and VHL E3 ligase, which recruits the ubiquitination machinery to STAT5A, promotes STAT5A ubiquitination, and induces its degradation via the 26S proteasome pathway. This blocks the STAT5A-mediated pro-inflammatory signaling cascade in T cells, attenuating ATCT-related adverse inflammatory responses while preserving anti-tumor T cell function [1] 3. E3 Ligase Ligand-Linker Conjugates 25 TFA is indicated for the prevention and treatment of adverse inflammatory reactions associated with adoptive T-cell therapies (including CAR-T therapy), such as cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS), with the advantage of decoupling anti-tumor efficacy from inflammatory side effects [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~183.66 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5 mg/mL (9.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 5 mg/mL (9.18 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 5 mg/mL (9.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8366 mL | 9.1831 mL | 18.3661 mL | |
| 5 mM | 0.3673 mL | 1.8366 mL | 3.6732 mL | |
| 10 mM | 0.1837 mL | 0.9183 mL | 1.8366 mL |