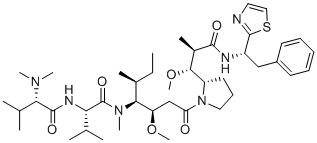
Dolastatin 10 (also known as DLS 10), a peptide-based natural product isolated from the marine mollusk Dolabela auricularia, is a highly potent antimitotic peptide that inhibits tubulin polymerization with an IC50 value of 1.2μM. It is an analog of Monomethyl auristatin E (MMAE) and can induce DC (dendritic cells) homing and activate cellular antitumor immune responses in patients.
Physicochemical Properties
| Molecular Formula | C42H68N6O6S |
| Molecular Weight | 785.09092 |
| Exact Mass | 784.492 |
| CAS # | 110417-88-4 |
| Related CAS # | 160800-57-7 (Auristatin E, a synthetic analog of dolastatin 10); 123884-00-4 (Dolastatin 15); 163768-50-1 (Auristatin F, a synthetic analog of dolastatin 10) |
| PubChem CID | 9810929 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 903.6±65.0 °C at 760 mmHg |
| Flash Point | 500.3±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.537 |
| LogP | 6.44 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 21 |
| Heavy Atom Count | 55 |
| Complexity | 1220 |
| Defined Atom Stereocenter Count | 9 |
| SMILES | CC[C@H](C)[C@@H]([C@@H](CC(=O)N1CCC[C@H]1[C@@H]([C@@H](C)C(=O)N[C@@H](CC2=CC=CC=C2)C3=NC=CS3)OC)OC)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](C(C)C)N(C)C |
| InChi Key | OFDNQWIFNXBECV-VFSYNPLYSA-N |
| InChi Code | InChI=1S/C42H68N6O6S/c1-13-28(6)37(47(10)42(52)35(26(2)3)45-40(51)36(27(4)5)46(8)9)33(53-11)25-34(49)48-22-17-20-32(48)38(54-12)29(7)39(50)44-31(41-43-21-23-55-41)24-30-18-15-14-16-19-30/h14-16,18-19,21,23,26-29,31-33,35-38H,13,17,20,22,24-25H2,1-12H3,(H,44,50)(H,45,51)/t28-,29+,31-,32-,33+,35-,36-,37-,38+/m0/s1 |
| Chemical Name | (S)-2-((S)-2-(dimethylamino)-3-methylbutanamido)-N-((3R,4S,5S)-3-methoxy-1-((S)-2-((1R,2R)-1-methoxy-2-methyl-3-oxo-3-(((S)-2-phenyl-1-(thiazol-2-yl)ethyl)amino)propyl)pyrrolidin-1-yl)-5-methyl-1-oxoheptan-4-yl)-N,3-dimethylbutanamide |
| Synonyms | NSC-376128; DLS-10; NSC376128; DLS10; NSC 376128; DLS 10 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product is not stable in solution, please use freshly prepared working solution for optimal results.(2). Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The molecular target of dolastatin 10 is not explicitly stated in this study. The paper focuses on structural modifications (auristatins) and their anticancer activity, referencing the parent compound’s properties without detailing its specific target or providing IC50/Ki/EC50 values for dolastatin 10 itself in this context. |
| ln Vitro |
Dolastatin 10 is a distinct pentapeptide that was found in Dolabella aurillaryia, a type of sea hare. These in vitro results are similar to those obtained from similar human cell mini-panels with GI50 values of 10-5-10-6 μg/mL (10-2-10-3) for both dolastatin 10 and auristatin PE [2]. The anti-CD30 monoclonal antibody cAC10 is combined with the cytotoxic compound monomethyl auristatin E (MMAE), a synthetic analogue of the tubulin polymerization inhibitor dolastatin 10, to form the antibody-drug combination (ADC) [3]. Dolastatin 10 and auristatin PE (a close derivative) are referenced as having GI50 values of 10^-5 to 10^-6 µg/mL (10^-2 to 10^-3 nM) against a minipanel of human cancer cell lines. This is mentioned as a benchmark for comparing the newly synthesized auristatin analogues (TP, 2-AQ, 6-AQ). [2] |
| ln Vivo |
In severe combined immunodeficiency (SCID) mouse xenograft models of ALCL or Hodgkin lymphoma, treatment with the ADC brentuximab vedotin (containing the dolastatin 10 analog MMAE) at 30 mg/kg showed no signs of toxicity. [3] - In preclinical models of ALCL, the ADC brentuximab vedotin (containing the dolastatin 10 analog MMAE) exhibited dose-dependent antitumor activity, with complete regressions achieved using doses ≥0.5 mg/kg (with repeat dosing) and ≥1 mg/kg (with single dosing). [3] |
| Cell Assay |
The in vitro cytotoxic activity is primarily attributed to the antibody-drug conjugate brentuximab vedotin and its released payload MMAE (a dolastatin 10 analog). [3] - The conjugate showed potent cytotoxic activity against CD30-expressing cells (IC50 < 10 ng/mL) but was 300-fold less potent against antigen-negative cells. [3] |
| Toxicity/Toxicokinetics |
The maximum tolerated dose (MTD) of the antibody-drug conjugate brentuximab vedotin (containing the dolastatin 10 analog MMAE) in a phase I trial was defined as 1.8 mg/kg (administered every 3 weeks). Dose-limiting toxicities (hyperglycemia, prostatitis, neutropenic fever) were observed at 2.7 mg/kg. [3] - One patient treated at 3.6 mg/kg experienced fever, neutropenia, sepsis, and died 14 days after the first dose. [3] - Common adverse events associated with the ADC (and thus indirectly related to the payload mechanism) included peripheral sensory neuropathy, fatigue, nausea, neutropenia, and thrombocytopenia. [3] |
| References |
[1]. Phase I trial of dolastatin-10 (NSC 376128) in patients with advanced solid tumors. Clin Cancer Res. 1999 Mar;5(3):525-31. [2]. Antineoplastic agents. 592. Highly effective cancer cell growth inhibitory structural modifications ofdolastatin 10. J Nat Prod. 2011 May 27;74(5):962-8. [3]. Brentuximab vedotin. Drugs R D. 2011;11(1):85-95. |
| Additional Infomation |
Dolastatin 10 is a tetrapeptide that is isolated from the sea hare Dolabella auricularia. It is a potent anticancer agent which inhibits tubulin polymerization. It has a role as an animal metabolite, a marine metabolite, an antineoplastic agent, an apoptosis inducer and a microtubule-destabilising agent. It is a member of 1,3-thiazoles and a tetrapeptide. It is functionally related to a L-valine. Dolastatin 10 has been used in trials studying the treatment of Sarcoma, Leukemia, Lymphoma, Liver Cancer, and Kidney Cancer, among others. Dolastatin 10 has been reported in Symploca, Dolabella auricularia, and other organisms with data available. Dolastatin 10 is a pentapeptide originally isolated from the marine mollusk Dolabella auricularia with potential antineoplastic activity. Binding to tubulin, Dolastatin 10 inhibits microtubule assembly, resulting in the formation of tubulin aggregates and inhibition of mitosis. This agent also induces tumor cell apoptosis through a mechanism involving bcl-2, an oncoprotein that is overexpressed in some cancers. (NCI04) Dolastatin 10 is a unique pentapeptide originally isolated from the sea hare Dolabella auricularia. [2] - It possesses remarkable anticancer properties and has generated intense interest in closely related derivatives called auristatins, which are suitable for clinical trials. [2] - Dolastatin 10 itself, along with three auristatin analogues, is mentioned as being in human cancer clinical trials ranging from phase I to phase III at the time of publication. [2] - The study explores structural modifications (phosphate and aminoquinoline conjugates) of the dolastatin 10 scaffold to create water-soluble prodrugs (e.g., auristatin TP salts) aimed at improving bioavailability and efficacy. The rationale is that serum phosphatases can dephosphorylate these salts to release the active drug intracellularly. [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 100 mg/mL (~127.37 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2737 mL | 6.3687 mL | 12.7374 mL | |
| 5 mM | 0.2547 mL | 1.2737 mL | 2.5475 mL | |
| 10 mM | 0.1274 mL | 0.6369 mL | 1.2737 mL |