Physicochemical Properties
| Molecular Formula | C14H4N2O2S2 |
| Molecular Weight | 296.32376 |
| Exact Mass | 295.971 |
| CAS # | 3347-22-6 |
| PubChem CID | 18771 |
| Appearance |
Dark brown crystals, with a coppery luster BROWN CRYSTALS |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 402.1±45.0 °C at 760 mmHg |
| Melting Point | 220ºC |
| Flash Point | 197.0±28.7 °C |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.776 |
| LogP | 1.86 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 20 |
| Complexity | 629 |
| Defined Atom Stereocenter Count | 0 |
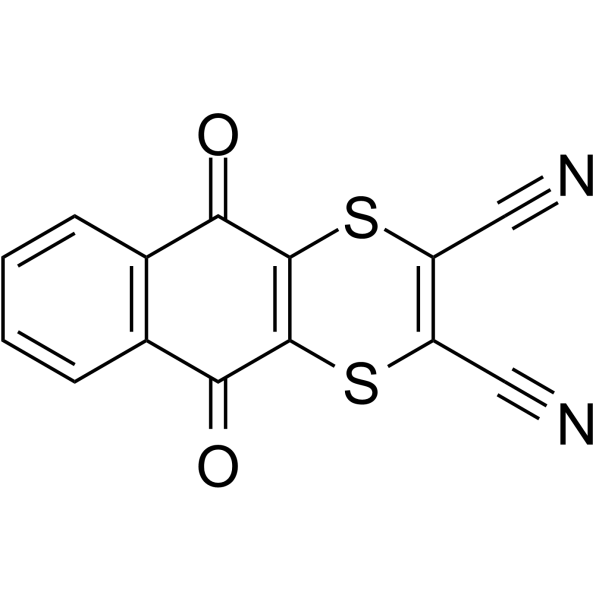
| SMILES | C1=CC2=C(C=C1)C(=O)C3=C(C2=O)SC(=C(C#N)S3)C#N |
| InChi Key | PYZSVQVRHDXQSL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H4N2O2S2/c15-5-9-10(6-16)20-14-12(18)8-4-2-1-3-7(8)11(17)13(14)19-9/h1-4H |
| Chemical Name | 5,10-dioxobenzo[g][1,4]benzodithiine-2,3-dicarbonitrile |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Reduction of protein and non-protein thiol groups is compatible with the behavior of exponentially aerobically developing Saccharomyces cerevisiae cells treated with acute dithianon, which causes the cells to lose cell wall and membrane integrity and die via necrosis. connected to consumption. Dithiacon-treated cells also showed a substantial increase in cellular reactive oxygen species (ROS) associated with change of the mitochondrial membrane potential [1]. Dithianon prevents conidial germination and hyphal development in filamentous fungus. Dithion suppresses respiration and fermentation, which affects various thiolase enzymes in the glycolytic pathway, including hexokinase, phosphofructokinase, and glyceraldehyde 3-phosphate dehydration, according to studies on Ehrlich ascites carcinoma and yeast cells. The enzyme hydrogenase[1]. In vitro, dithianon is cytotoxic and interferes with BLAB/c 3 T3 cells' ability to undergo cell transformation [1]. |
| ln Vivo | Male mice's liver microsomes show enhanced testosterone hydroxylase activity when given with acute dosages of dithianon, but female mice show deactivation [1]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Groups of 5 laying hens were given capsules containing 0, 0.36 or 3.6 mg 14C-dithianon/day (purity 99.3%), orally for 5 consecutive days. These doses were equivalent to 3 or 30 ppm in the feed, respectively. Excreta and eggs were collected twice daily. Radioactivity was excreted very rapidly and at a constant rate. Irrespective of the dose, 4 days after the last administration, total recovery of 14C was about 94% of the dose with about 90% recovered in the excreta and 3-5% in the GIT contents. Detectable tissue residues were found in kidney and liver (0.03% and 0.02%, respectively). The 14C recovered in eggs comprised less than 0.01% of the total radioactive dose. The radioactivity in egg yolk was higher than in egg white and was maximal at sacrifice (0.005 mg/kg and 0.075 mg/kg at the low- and the high-dose, respectively) |
| Toxicity/Toxicokinetics |
Toxicity Data LC50 (rat) = 3,000 mg/m3/4hr Non-Human Toxicity Values LD50 Rat oral 638 mg/kg LD50 Guinea pig oral 110 mg/kg LD50 Rat percutaneous >2000 mg/kg LD50 Mouse oral 1140 mg/kg For more Non-Human Toxicity Values (Complete) data for DITHIANON (6 total), please visit the HSDB record page. |
| References |
[1]. Necrotic cell death induced by dithianon on Saccharomyces cerevisiae. Pestic Biochem Physiol. 2018 Jul;149:137-142. [2]. Structures of four polymorphs of the pesticide dithianon solved from X-ray powder diffraction data. Acta Crystallogr B. 2012 Dec;68(Pt 6):661-6. |
| Additional Infomation | Dithianon is a naphthodithiin that is 5,10-dioxo-5,10-dihydronaphtho[2,3-b][1,4]dithiin which is substituted by nitrile groups at positions 2 and 3. It is a broad spectrum fungicide used to control scab, downy mildew, rust, and leaf spot in the commercial growing of grapes and other fruit, citrus, coffee, and vegetables. It has a role as an antifungal agrochemical. It is a naphthodithiin, a dinitrile and a member of p-quinones. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3747 mL | 16.8737 mL | 33.7473 mL | |
| 5 mM | 0.6749 mL | 3.3747 mL | 6.7495 mL | |
| 10 mM | 0.3375 mL | 1.6874 mL | 3.3747 mL |