Physicochemical Properties
| Molecular Formula | C68H109N17O22S2 |
| Molecular Weight | 1580.82376 |
| Exact Mass | 1579.74 |
| CAS # | 272105-42-7 |
| Related CAS # | Disitertide diammonium;Disitertide TFA |
| PubChem CID | 9833838 |
| Appearance | White to off-white solid powder |
| Density | 1.327g/cm3 |
| Boiling Point | 1968.718ºC at 760 mmHg |
| LogP | 1.272 |
| Hydrogen Bond Donor Count | 22 |
| Hydrogen Bond Acceptor Count | 25 |
| Rotatable Bond Count | 51 |
| Heavy Atom Count | 109 |
| Complexity | 3140 |
| Defined Atom Stereocenter Count | 17 |
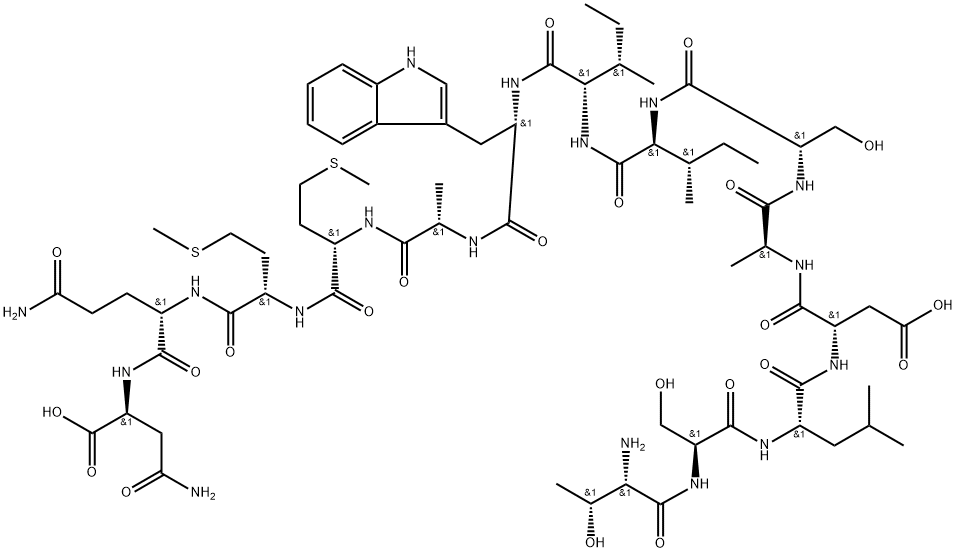
| SMILES | CC[C@H](C)[C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)N[C@@H](C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H](C)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H]([C@@H](C)O)N |
| InChi Key | IUYPEUHIWDMJLM-SWHDLQTQSA-N |
| InChi Code | InChI=1S/C68H109N17O22S2/c1-12-32(5)53(85-67(105)54(33(6)13-2)84-64(102)48(30-87)82-56(94)35(8)74-61(99)45(27-51(91)92)79-62(100)43(24-31(3)4)78-63(101)47(29-86)83-65(103)52(71)36(9)88)66(104)80-44(25-37-28-72-39-17-15-14-16-38(37)39)60(98)73-34(7)55(93)75-41(20-22-108-10)58(96)77-42(21-23-109-11)59(97)76-40(18-19-49(69)89)57(95)81-46(68(106)107)26-50(70)90/h14-17,28,31-36,40-48,52-54,72,86-88H,12-13,18-27,29-30,71H2,1-11H3,(H2,69,89)(H2,70,90)(H,73,98)(H,74,99)(H,75,93)(H,76,97)(H,77,96)(H,78,101)(H,79,100)(H,80,104)(H,81,95)(H,82,94)(H,83,103)(H,84,102)(H,85,105)(H,91,92)(H,106,107)/t32-,33-,34-,35-,36+,40-,41-,42-,43-,44-,45-,46-,47-,48-,52-,53-,54-/m0/s1 |
| Chemical Name | (2S)-4-amino-2-[[(2S)-5-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S,3S)-2-[[(2S,3S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S,3R)-2-amino-3-hydroxybutanoyl]amino]-3-hydroxypropanoyl]amino]-4-methylpentanoyl]amino]-3-carboxypropanoyl]amino]propanoyl]amino]-3-hydroxypropanoyl]amino]-3-methylpentanoyl]amino]-3-methylpentanoyl]amino]-3-(1H-indol-3-yl)propanoyl]amino]propanoyl]amino]-4-methylsulfanylbutanoyl]amino]-4-methylsulfanylbutanoyl]amino]-5-oxopentanoyl]amino]-4-oxobutanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Transforming Growth Factor β (TGF-β) [4][5] |
| ln Vitro |
Disitertide (P144, 100 μg/mL) lowers the protein expression levels of PI3K and p-Akt in MC3T3-E1 cells and promotes the protein expression of Bax [2]. Disitertide (TGF-β1 inhibitor) reduces the expression of MACC1-AS1 in GC cells, indicating that targeting the TGFβ signaling pathway may be a feasible technique to suppress MSC-induced stemness and chemotherapy resistance [3]. Disitertide (10 μg/mL to 200 μg/mL) influences the proliferation, causes apoptosis and anoikis of A172 and U-87 MG GBM cell lines [5]. - Disitertide (P144®) is a TGF-β inhibitor peptide with antitumoral activity against human glioblastoma cell lines. At concentrations of 10, 50, 100 μM, it dose-dependently inhibited the proliferation of U87 and U251 glioblastoma cells by 22±3%, 45±4%, 68±5% and 18±2%, 41±3%, 63±4%, respectively [5] - It modified the expression of TGF-β signaling-related proteins: 100 μM Disitertide increased SMAD7 protein levels by 2.3±0.2-fold and decreased SKI protein levels by 58±4% in U87 cells [5] - It induced apoptosis of glioblastoma cells: 100 μM increased the apoptotic rate of U251 cells by 35±3% compared to the control group [5] |
| ln Vivo |
After two weeks of treatment, 300 μg/mL topical application of Disitertide enhanced the morphological features of hypertrophic scars and encouraged scar development in a "in vivo" nude mouse model [4]. - In nude mouse human hypertrophic scar model: Local intralesional injection of Disitertide (P144®) (100 μg/scar, 3 times weekly for 4 weeks) significantly reduced scar thickness by 42±3% and collagen deposition by 51±4% [4] - It inhibited TGF-β-mediated fibrosis: The treatment decreased the expression of TGF-β1, collagen I, and α-smooth muscle actin (α-SMA) in scar tissues by 48±4%, 55±5%, and 49±3%, respectively [4] |
| Enzyme Assay |
- TGF-β signaling pathway activity assay: Human glioblastoma cells (U87, U251) were treated with Disitertide (10–100 μM) for 48 hours. Cell lysates were prepared for Western blot analysis to detect SMAD7 and SKI protein levels, reflecting the inhibition of TGF-β signaling [5] - TGF-β-induced fibrosis-related protein assay: Fibroblasts isolated from human hypertrophic scars were treated with Disitertide (50–100 μM) and TGF-β1. The expression of collagen I and α-SMA was detected by Western blot to evaluate anti-fibrotic activity [4] |
| Cell Assay |
Western Blot analysis [2] Cell Types: Mouse embryonic osteoblast precursor MC3T3-E1 cell Tested Concentrations: 100 μg/mL Incubation Duration: 4 h Experimental Results: Dramatically inhibited PI3K and p-Akt protein expression levels, and induced Bax protein Comparison of cells expressing MC3T3-E1 with the miR-590 group - Glioblastoma cell proliferation assay: U87 and U251 cells were seeded in 96-well plates (5×10³ cells/well) and incubated with Disitertide (10–100 μM) for 72 hours. Cell viability was measured by MTT assay to calculate proliferation inhibition rates [5] - Apoptosis assay: U251 cells were treated with Disitertide (50, 100 μM) for 48 hours, stained with Annexin V-FITC/PI, and analyzed by flow cytometry to determine apoptotic rates [5] - TGF-β signaling protein expression assay: Glioblastoma cells were treated with the peptide (10–100 μM) for 48 hours. Total protein was extracted for Western blot detection of SMAD7 and SKI [5] |
| Animal Protocol |
Animal/Disease Models: Human hypertrophic scars were implanted into 60 nude mice [4]. Doses: Add 300 μg/mL to Lipogel. Route of Administration: Apply topically daily. Experimental Results: 83.3% of xenografts were successfully expelled. - Nude mouse human hypertrophic scar model: Human hypertrophic scar tissue fragments were implanted subcutaneously into nude mice. After 2 weeks of implantation, Disitertide (P144®) was dissolved in physiological saline and administered via intralesional injection at 100 μg/scar, 3 times weekly for 4 weeks. Mice were sacrificed, and scar tissues were collected for histological analysis (hematoxylin-eosin and Masson staining) and Western blot detection of TGF-β1, collagen I, and α-SMA [4] |
| References |
[1]. Targeting the TGFβ pathway for cancer therapy. Pharmacol Ther. 2015 Mar;147:22-31. [2]. Upregulation of microRNA‑590 in rheumatoid arthritis promotes apoptosis of bone cells through transforming growth factor‑β1/phosphoinositide 3‑kinase/Akt signaling. Int J Mol Med. 2019 May;43(5):2212-2220. [3]. MSC-regulated lncRNA MACC1-AS1 promotes stemness and chemoresistance through fatty acid oxidation in gastric cancer. Oncogene. 2019 Jun;38(23):4637-4654. [4]. Effect of P144® (Anti-TGF-β) in an "In Vivo" Human Hypertrophic Scar Model in Nude Mice. PLoS One. 2015 Dec 31;10(12):e0144489. [5]. P144, a Transforming Growth Factor beta inhibitor peptide, generates antitumoral effects and modifies SMAD7 and SKI levels in human glioblastoma cell lines. Cancer Lett. 2016 Oct 10;381(1):67-75. |
| Additional Infomation |
Disitertide has been used in trials studying the treatment of Skin Fibrosis. - Disitertide, also known as P144®, is a synthetic peptide that specifically inhibits the TGF-β signaling pathway [4][5] - Its mechanism of action involves blocking TGF-β-mediated signal transduction, thereby suppressing cell proliferation, fibrosis, and promoting apoptosis in target cells [4][5] - It shows potential therapeutic applications in the treatment of TGF-β-related diseases, including human hypertrophic scars and glioblastoma [4][5] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.6326 mL | 3.1629 mL | 6.3258 mL | |
| 5 mM | 0.1265 mL | 0.6326 mL | 1.2652 mL | |
| 10 mM | 0.0633 mL | 0.3163 mL | 0.6326 mL |