Physicochemical Properties
| Molecular Formula | C10H8N2O2S2 |
| Molecular Weight | 252.31272 |
| Exact Mass | 252.003 |
| CAS # | 3696-28-4 |
| PubChem CID | 3109 |
| Appearance | White to off-white solid powder |
| Density | 1.38 g/cm3 |
| Boiling Point | 582.8ºC at 760 mmHg |
| Melting Point | 205ºC |
| Flash Point | 306.2ºC |
| Index of Refraction | 1.681 |
| LogP | 3.343 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 16 |
| Complexity | 199 |
| Defined Atom Stereocenter Count | 0 |
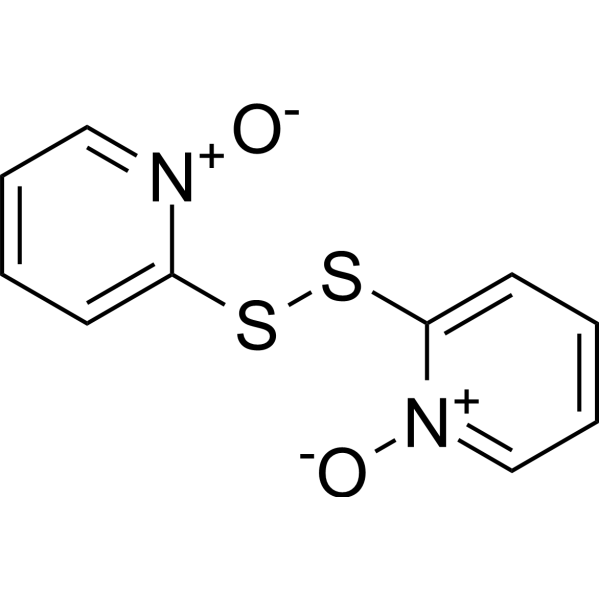
| SMILES | C1=CC=[N+](C(=C1)SSC2=CC=CC=[N+]2[O-])[O-] |
| InChi Key | ZHDBTKPXEJDTTQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C10H8N2O2S2/c13-11-7-3-1-5-9(11)15-16-10-6-2-4-8-12(10)14/h1-8H |
| Chemical Name | 1-oxido-2-[(1-oxidopyridin-1-ium-2-yl)disulfanyl]pyridin-1-ium |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Trichophyton rubrum is susceptible to the antifungal effects of dipyrithione (20 μg/mL), as evidenced by its MIC value of 6.03 µM[1]. With an IC50 value of 0.22 µM, dipyrithione (72 h) exhibits cytotoxic action against 293 T cells[1]. In RAW264.7 cells, dipyrithione (1–5 µM; 8.5 h) dose-dependently suppresses the upregulation of iNOS and COX-2 caused by LPS (100 ng/ml) [2]. Dipyrithione (1 µM; 8.5 h) suppresses the rise in iNOS that is brought on by LPS, but it has no effect on COX-2 mRNA levels or the creation of more NO as a result of LPS [2]. In RAW 246.7 cells, dipyrithione (3 µM; 2, 5 hours) inhibits LPS-induced STAT1 phosphorylation but has no effect on LPS-induced MAPK and NF-κB activation [2]. Dipyrithione (0–5 μg/mL; 48 hours) has dose-dependent antiproliferative action on KB, 231, U937, and K562 cells [3]. Dipyrithione causes G1 cycle arrest and apoptosis at 2.5 μg/ml [3]. |
| ln Vivo | In guinea pigs, dipyrithione (0.2 mg/cm2; topically applied once daily for 10 days) exhibited good antidermatophyte activity [1]. In mice, dipyrithione (1, 2.5, and 5 mg/kg; i.p.) exhibits anti-inflammatory properties [2]. Mice treated once daily for 10 days with dipyrithione (5 mg/kg; i.p.) exhibit antitumor activity [3]. |
| Cell Assay |
Western Blot Analysis[2] Cell Types: RAW264.7 Cell Tested Concentrations: 1-5 µM Incubation Duration: 8.5 hrs (hours) Experimental Results: Inhibited LPS (100 ng/ml)-induced upregulation of iNOS and COX-2 expression in a dose-dependent manner. Cell proliferation assay [2] Cell Types: KB, 231, U937, K562 Cell Tested Concentrations: 2.5 μg/ml Incubation Duration: 24 hrs (hours) Experimental Results: Induced cell cycle arrest in G1 phase, induced p21 accumulation, and downregulated the expression of CyclinD1 and CyclinE1. Apoptosis analysis [3] Cell Types: KB, 231, U937, K562 Cell Tested Concentrations: 2.5 μg/ml Incubation Duration: 36 hrs (hours) Experimental Results: Induced apoptosis by inducing caspase-9, caspase-3 and PARP cleavage. Western Blot Analysis[3] Cell Types: RAW264.7 Cell Tested Concentrations: 1-5 µM Incubation Duration: 8.5 hrs (hours) Experimental Results: Inhibited LPS (100 ng/ml)-induced upregulation of iNOS and COX-2 expression in a dose-dependent manner. |
| Animal Protocol |
Animal/Disease Models: Guinea pig (infected with Trichophyton rubrum) [1] Doses: 0.2 mg/cm2 Route of Administration: External use, one time/day for 10 days. Experimental Results: Hair growth is normal, and the skin is free of scales. Animal/Disease Models: 18-22g male ICR mice 2 Doses: 1, 2.5, 5 mg/kg Route of Administration: intraperitoneal (ip) injection Experimental Results: The survival rate was increased from 10% to 30%, 60% and 90% respectively. Animal/Disease Models: 6 weeks, 18-20 g male ICR mice (liver cancer 22 (H22) cells) [3] Doses: 2.5 mg/kg Route of Administration: intraperitoneal (ip) injection; one time/day for 10 days Experimental Results: Inhibition of tumor growth . |
| References |
[1]. Song X, et al. In vivo antifungal activity of dipyrithione against Trichophyton rubrum on guinea pig dermatophytosis models. Biomed Pharmacother. 2018 Dec;108:558-564. [2]. Liu Z, et al. Dipyrithione inhibits lipopolysaccharide-induced iNOS and COX-2 up-regulation in macrophages and protects against endotoxic shock in mice. FEBS Lett. 2008 May 28;582(12):1643-50. [3]. Fan Y, et al. Dipyrithione induces cell-cycle arrest and apoptosis in four cancer cell lines in vitro and inhibits tumor growth in a mouse model. BMC Pharmacol Toxicol. 2013 Oct 21;14:54. |
| Additional Infomation |
Dipyrithione is a pyridinium ion. Dipyrithione, a bactericidal and fungicidal pyrithione derivate, was formulated as Crimanex anti-dandruff shampoo, but is no longer available. It is currently used as a pesticide, and not used in any FDA approved drug products. Interestingly, dipyrithione has been studied and shown to have cytotoxic and potent broad-spectrum antitumor activity, which suggests a potential basis for an anticancer drug development. Pyrithione derivatives, such as [DB06815] and sodium pyrithione, are widely used as cosmetic preservatives and as anti-dandruff agents in shampoos. It may be combined with other ingredients, such as triclosan to serve as antifungal and antibacterial skin treatments. Dandruff is a common scalp disease affecting >40% of the world's adult population, and may be caused by fungi such as Malassezia globosa and M. restricta. Dipyrithione has been reported in Marsypopetalum modestum and Allium stipitatum with data available. Drug Indication Scalp dandruff Mechanism of Action This drug is a fungicidal agent. Fungicide, also called antimycotic, any toxic substance used to kill or inhibit the growth of fungi. Pharmacodynamics This drug decreases or eliminates dandruff from the scalp, which is caused by various types of fungi., |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.9634 mL | 19.8169 mL | 39.6338 mL | |
| 5 mM | 0.7927 mL | 3.9634 mL | 7.9268 mL | |
| 10 mM | 0.3963 mL | 1.9817 mL | 3.9634 mL |