Physicochemical Properties
| Molecular Formula | C32H48O8 |
| Molecular Weight | 560.7187 |
| Exact Mass | 560.334 |
| CAS # | 13201-14-4 |
| PubChem CID | 267250 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 692.2±55.0 °C at 760 mmHg |
| Flash Point | 215.4±25.0 °C |
| Vapour Pressure | 0.0±4.9 mmHg at 25°C |
| Index of Refraction | 1.559 |
| LogP | 2.56 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 40 |
| Complexity | 1170 |
| Defined Atom Stereocenter Count | 9 |
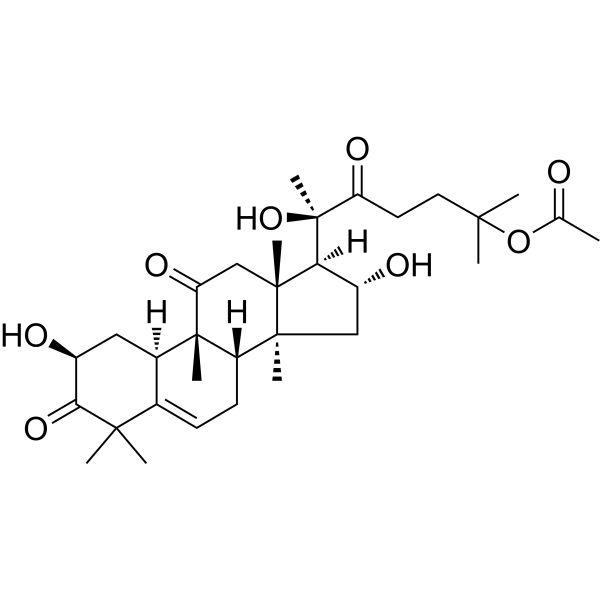
| SMILES | O([H])[C@]1([H])C([H])([H])[C@@]2(C([H])([H])[H])[C@]3([H])C([H])([H])C([H])=C4C(C([H])([H])[H])(C([H])([H])[H])C([C@]([H])(C([H])([H])[C@@]4([H])[C@]3(C([H])([H])[H])C(C([H])([H])[C@]2(C([H])([H])[H])[C@@]1([H])[C@@](C(C([H])([H])C([H])([H])C(C([H])([H])[H])(C([H])([H])[H])OC(C([H])([H])[H])=O)=O)(C([H])([H])[H])O[H])=O)O[H])=O |
| InChi Key | QZJJDOYZVRUEDY-NRNCYQGDSA-N |
| InChi Code | InChI=1S/C32H48O8/c1-17(33)40-27(2,3)13-12-23(36)32(9,39)25-21(35)15-29(6)22-11-10-18-19(14-20(34)26(38)28(18,4)5)31(22,8)24(37)16-30(25,29)7/h10,19-22,25,34-35,39H,11-16H2,1-9H3/t19-,20+,21-,22+,25+,29+,30-,31+,32+/m1/s1 |
| Chemical Name | [(6R)-6-[(2S,8S,9R,10R,13R,14S,16R,17R)-2,16-dihydroxy-4,4,9,13,14-pentamethyl-3,11-dioxo-2,7,8,10,12,15,16,17-octahydro-1H-cyclopenta[a]phenanthren-17-yl]-6-hydroxy-2-methyl-5-oxoheptan-2-yl] acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Nuclear factor of activated T cells (NFAT) (Inhibits activation) [1] |
| ln Vitro |
Dihydrocucurbitacin B inhibited the proliferation of phytohemagglutinin (PHA)-stimulated human T lymphocytes with an IC₅₀ of 1.48 µM (range 1.0-2.5 µM). At 2.5 µM, it completely inhibited cell proliferation. The compound was not cytotoxic at concentrations up to 10 µM. Dihydrocucurbitacin B (2.5 µM) arrested the cell cycle of PHA-stimulated T lymphocytes in the G₀/G₁ phase, with minimal progression to the S phase. This effect was similar to dexamethasone (5 µM), while aphidicolin (0.75 µM) arrested the cycle in the S phase. Dihydrocucurbitacin B (2.5-1.0 µM) significantly inhibited the production of interferon-γ (IFN-γ), interleukin-2 (IL-2), and interleukin-10 (IL-10) in PHA-stimulated human T lymphocytes. It also slightly reduced interleukin-4 (IL-4) production, but this was not statistically significant. Reverse transcription-polymerase chain reaction (RT-PCR) analysis showed that Dihydrocucurbitacin B (2.5 µM) attenuated the mRNA expression of IFN-γ, IL-2, IL-4, and IL-10 in stimulated T lymphocytes. Western blot analysis demonstrated that Dihydrocucurbitacin B (2.5 µM) inhibited the expression of key cell cycle cyclins (A₁, B₁, D₂, and E₁) in PHA-stimulated human T lymphocytes. An electrophoretic mobility shift assay (EMSA) showed that Dihydrocucurbitacin B (2.5 µM) inhibited the activation of the transcription factor NFAT in PHA-stimulated T lymphocytes, without affecting calcium influx into the cells. [1] |
| ln Vivo |
In a mouse model of oxazolone-induced delayed-type hypersensitivity (DTH), topical application of Dihydrocucurbitacin B (0.1, 0.3, and 0.5 mg/ear) significantly inhibited ear swelling in a dose-dependent manner. In a mouse model of dinitrofluorobenzene (DNFB)-induced DTH, topical application of Dihydrocucurbitacin B (0.1, 0.3, and 0.5 mg/ear) also significantly inhibited ear swelling. In a mouse model of sheep red blood cell (SRBC)-induced DTH, intraperitoneal administration of Dihydrocucurbitacin B (10 mg/kg) inhibited paw swelling. Histological analysis of ear tissue from oxazolone- and DNFB-treated mice revealed that Dihydrocucurbitacin B reduced edema, inflammatory cell infiltration (neutrophils, lymphocytes, macrophages), and epidermal changes (papillomatosis, spongiosis) compared to control groups. Ex vivo analysis of inflamed ear tissue from oxazolone- and DNFB-treated mice showed that Dihydrocucurbitacin B reduced the levels of key pro-inflammatory cytokines, including interleukin-1β (IL-1β), interleukin-4 (IL-4), and tumor necrosis factor-α (TNF-α). [1] |
| Cell Assay |
T Lymphocyte Proliferation Assay: Human peripheral blood lymphocytes were isolated and resuspended. Cells were seeded into 96-well plates and stimulated with phytohemagglutinin (PHA). Dihydrocucurbitacin B at various concentrations (1.0 to 2.5 µM) or dexamethasone (5 µM) were added. Plates were incubated for 4 days. Cell proliferation was then quantified using a modified colorimetric MTT assay. Controls included PHA-stimulated cells (100% activity) and unstimulated cells (0% activity). Cell Cycle Analysis: T lymphocytes were adjusted to a density of 1 × 10⁶ cells/mL and seeded into 24-well plates with or without PHA stimulation. Dihydrocucurbitacin B (2.5 µM), dexamethasone (5 µM), or aphidicolin (0.75 µM) were added. After incubation for specified times, cells were harvested, fixed in ethanol, stained with propidium iodide and ribonuclease A, and analyzed by flow cytometry to determine DNA content and cell cycle phase distribution. Cell Viability and Cytotoxicity Determination: T lymphocytes were cultured and stimulated with PHA for 4 days. Propidium iodide solution was then added, and cell viability was measured by flow cytometry after 30 minutes. Cytokine Production Determination: T lymphocytes were cultured with PHA alone or in combination with various concentrations of Dihydrocucurbitacin B for 4 days. Cell culture supernatants were collected and assayed for IL-2, IL-4, IL-10, and IFN-γ levels using enzyme-linked immunosorbent assay (ELISA) kits. RNA Extraction and RT-PCR for Cytokine mRNA: T cells were treated with or without PHA and cultured with Dihydrocucurbitacin B for 18 hours. Total RNA was extracted using spin columns. RNA concentration and purity were determined spectrophotometrically. Reverse transcription was performed to generate cDNA, which was then used as a template for PCR amplification with specific primers for IFN-γ, IL-2, IL-4, IL-10, and β-actin (as a housekeeping control). PCR products were analyzed by gel electrophoresis and densitometry. [1] |
| Animal Protocol |
Oxazolone- and Dinitrofluorobenzene (DNFB)-Induced DTH in Mice: Female Swiss mice were sensitized by topical application of the allergen (oxazolone or DNFB) on days 1 and 2. On day 6, a challenge dose was applied to the ears. Dihydrocucurbitacin B (0.1, 0.3, or 0.5 mg/ear) dissolved in acetone was topically applied to the ears at 2, 24, 48, and 72 hours after challenge. Ear thickness was measured with a micrometer before treatment and at 24, 48, and 72 hours post-challenge to calculate edema. Sheep Red Blood Cell (SRBC)-Induced DTH in Mice: Dihydrocucurbitacin B was administered intraperitoneally at a dose of 10 mg/kg. The specific timing of administration relative to SRBC immunization and challenge is not detailed in the provided text. Paw swelling was measured as an indicator of the inflammatory response. [1] |
| Toxicity/Toxicokinetics | Dihydrocucurbitacin B exhibited no cytotoxicity towards human T lymphocytes at concentrations up to 10 µM as determined by propidium iodide exclusion and flow cytometry. [1] |
| References |
[1]. Dihydrocucurbitacin B inhibits delayed type hypersensitivity reactions by suppressing lymphocyte proliferation. J Pharmacol Exp Ther. 2007 Sep;322(3):1261-8. |
| Additional Infomation |
23,24-dihydrocucurbitacin B is a 23,24-dihydrocucurbitacin in which a lanostane skeleton is multi-substituted with hydroxy, methyl and oxo substituents, with unsaturation at position 5; a hydroxy function at C-25 is acetylated. It is a 23,24-dihydrocucurbitacin, a secondary alpha-hydroxy ketone and a tertiary alpha-hydroxy ketone. It is functionally related to a cucurbitacin B. Dihydrocucurbitacin B has been reported in Begonia nantoensis, Trichosanthes cucumeroides, and other organisms with data available. Dihydrocucurbitacin B is a triterpene isolated from the roots of Cayaponia tayuya. It exhibits anti-inflammatory and potential immunomodulatory activities. Its primary mechanism of action in suppressing T cell-mediated delayed-type hypersensitivity (DTH) reactions involves the inhibition of the transcription factor NFAT. This inhibition leads to reduced expression of key cell cycle cyclins (A₁, B₁, D₂, E₁) and pro-inflammatory cytokines (e.g., IL-2, IFN-γ), resulting in cell cycle arrest at the G₀/G₁ phase and suppressed proliferation of activated T lymphocytes. The compound does not appear to act through glucocorticoid-like mechanisms or by directly inhibiting calcium influx. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7834 mL | 8.9171 mL | 17.8342 mL | |
| 5 mM | 0.3567 mL | 1.7834 mL | 3.5668 mL | |
| 10 mM | 0.1783 mL | 0.8917 mL | 1.7834 mL |