Dihydrocapsaicin is a naturally occuring capsaicin isolated from Capsicum fruit, it acts as a potent and selective TRPV1 agonist, and also increases p-Akt levels. Dihydrocapsaicin enhances the hypothermia-induced neuroprotection. Capsaicin is the primary active component of the heat and pain-eliciting lipid soluble fraction of the Capsicum pepper. Like capsaicin, dihydrocapsaicin is an irritant. Capsaicin is found in natural hot pepper extracts along with a number of impurities, including dihydrocapsaicin and several lesser impurities. Separation by HPLC is required in order to obtain pure dihydrocapsaicin. Dihydrocapsaicin represents about 10% of the compound present in commercial preparations purporting to be pure capsaicin, but it has about the same pungency as capsaicin. VR1 (vanilloid receptor 1) is a heat activated calcium ion channel which functions as a part of the normal nociceptive pain pathway. Capsaicin elicits a sensation of burning pain by activation of VR1 on small, non-myelinated polymodal C-type nociceptive nerve fibers. The potency of dihydrocapsaicin at VR1 appears equivalent to capsaicin. Antioxidant. Reduces oxidation of serum lipids. Mutagenic. Dihydrocapsaicin is an activator of VR1.
Physicochemical Properties
| Molecular Formula | C18H29NO3 |
| Molecular Weight | 307.4278 |
| Exact Mass | 307.214 |
| CAS # | 19408-84-5 |
| Related CAS # | Dihydrocapsaicin-d3;1330261-21-6 |
| PubChem CID | 107982 |
| Appearance | White to off-white solid powder |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 457.3±55.0 °C at 760 mmHg |
| Melting Point | 62-65 °C(lit.) |
| Flash Point | 230.4±31.5 °C |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.508 |
| LogP | 4.72 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 22 |
| Complexity | 307 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | XJQPQKLURWNAAH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H29NO3/c1-14(2)8-6-4-5-7-9-18(21)19-13-15-10-11-16(20)17(12-15)22-3/h10-12,14,20H,4-9,13H2,1-3H3,(H,19,21) |
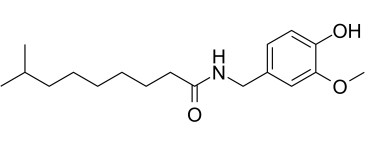
| Chemical Name | N-[(4-hydroxy-3-methoxyphenyl)methyl]-8-methylnonanamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
1. PI3K/Akt signaling pathway (regulates neuroprotective effects in ischemic stroke models) [2] 2. Platelet aggregation-related targets (inhibits in vitro platelet aggregation) [3] |
| ln Vitro |
Dihydrocapsaicin (0-100 μM) suppresses the activity of inhibitors IX (25-100 μM) and VIII:C (6.26-100 μM) as well as aggregation [3]. 1. Blood coagulation and platelet aggregation inhibition: Dihydrocapsaicin (10-100 μM) dose-dependently inhibited in vitro blood coagulation and platelet aggregation; at 100 μM, it prolonged activated partial thromboplastin time (APTT) by 32% and prothrombin time (PT) by 28% relative to control, and reduced adenosine diphosphate (ADP)-induced platelet aggregation by 45% and collagen-induced aggregation by 52% [3] 2. Neuroprotective-related signaling modulation: In oxygen-glucose deprivation (OGD)-induced neuronal cell models, Dihydrocapsaicin (1-10 μM) upregulated the phosphorylation level of Akt (p-Akt) by 68% at 10 μM, while the total Akt expression remained unchanged; the effect was reversed by PI3K inhibitors, confirming the regulation of PI3K/Akt pathway [2] |
| ln Vivo |
In the MCAO model, dihydrocapsaicin (0.5 mg/kg, IP, once) shows reduced effectiveness and neuroprotective effects [2]. 1. Hypothermia-enhanced neuroprotection in ischemic stroke: In middle cerebral artery occlusion (MCAO) rat models of ischemic stroke, combined treatment with Dihydrocapsaicin (5 mg/kg) and mild hypothermia (32℃ for 4 h post-ischemia) reduced cerebral infarct volume by 62% (vs 35% for hypothermia alone and 22% for Dihydrocapsaicin alone), improved neurological deficit scores (from 3.5 in control to 1.2 in combination group), and increased survival rate from 58% (control) to 85% (combination group) at 7 days post-stroke. Western blot of brain tissues showed that the combination group had 75% higher p-Akt levels and 58% lower cleaved caspase-3 levels than the MCAO control group [2] |
| Enzyme Assay |
1. Blood coagulation factor activity assay: Citrated human plasma was incubated with serial concentrations of Dihydrocapsaicin (10-100 μM) for 30 min at 37℃, then mixed with APTT or PT detection reagents. The time required for fibrin clot formation was recorded using a coagulation analyzer, and the change in clotting time was used to evaluate the compound’s effect on coagulation factor activity [3] 2. PI3K/Akt pathway activity detection: Neuronal cell lysates from OGD models treated with Dihydrocapsaicin were prepared, and equal amounts of protein were separated by SDS-PAGE and transferred to membranes. The membranes were incubated with primary antibodies against p-Akt, total Akt, and internal reference proteins overnight at 4℃, followed by secondary antibody incubation for 1 h at room temperature. The band intensity of p-Akt was quantified to assess PI3K/Akt pathway activation [2] |
| Cell Assay |
1. Platelet aggregation assay: Washed human platelets were suspended in buffer and incubated with Dihydrocapsaicin (10-100 μM) for 15 min at 37℃, then stimulated with ADP (10 μM) or collagen (5 μg/mL). The change in light transmittance was monitored continuously for 5 min using an aggregometer, and the maximum aggregation rate was calculated relative to the control group (without Dihydrocapsaicin) [3] 2. OGD-induced neuronal cell protection assay: Primary cortical neurons were seeded in 96-well plates and cultured for 7 days. The cells were subjected to OGD (0.5% O₂, glucose-free medium) for 2 h, then treated with Dihydrocapsaicin (1-10 μM) for 24 h under normoxic conditions. A cell viability detection reagent was added and incubated for 2 h at 37℃, and absorbance was measured to calculate cell viability (10 μM Dihydrocapsaicin increased viability from 52% in OGD control to 81%) [2] |
| Animal Protocol |
Animal/Disease Models: SD (SD (Sprague-Dawley)) rat (adult, male, 300-340 g, right mid-abdominal cerebral artery occlusion (MCAO) [2] Doses: 0.5 mg/kg Route of Administration: intraperitoneal (ip) injection, one time Experimental Results:demonstrated cooling effect, The rectal temperature dropped to approximately 35.0°C within 30 minutes, remained at or below 35.0°C for approximately 20 minutes, and then gradually returned to approximately 36.5°C over 120 minutes. Dramatically diminished ischemia-reperfusion-induced infarct volume (36.2 %±2.5%). Reduces ROS levels at 24 hrs (hrs (hours)) and reduces ischemia-reperfusion-induced high-level cell death. 1. MCAO ischemic stroke rat model and administration protocol: Male Sprague-Dawley rats (250-300 g) were anesthetized and subjected to MCAO for 2 h to induce focal cerebral ischemia, followed by reperfusion. Rats were randomly divided into 4 groups (sham, MCAO control, hypothermia alone, Dihydrocapsaicin + hypothermia), with 10 rats per group. Dihydrocapsaicin was dissolved in DMSO and diluted with normal saline (final DMSO < 0.1%) to prepare the administration solution, which was administered via intraperitoneal injection at 5 mg/kg immediately after reperfusion, and once daily for 3 consecutive days. The hypothermia group was subjected to 32℃ body temperature for 4 h starting at reperfusion, while the combination group received both treatments. The vehicle group received equal volume of DMSO-saline mixture [2] 2. Neurological deficit and infarct volume detection: Neurological deficit scores were evaluated using a 5-point scale at 24 h, 3 days, and 7 days post-reperfusion. At 7 days post-stroke, rats were euthanized, and brain tissues were sliced and stained with triphenyltetrazolium chloride (TTC). The infarct area was calculated using image analysis software to quantify infarct volume [2] |
| References |
[1]. Impairment in function and expression of transient receptor potential vanilloid type 4 in Dahl salt-sensitive rats: significance and mechanism. Hypertension. 2010 Apr;55(4):1018-25. [2]. Dihydrocapsaicin (DHC) enhances the hypothermia-induced neuroprotection following ischemic stroke via PI3K/Akt regulation in rat. Brain Res. 2017 Sep 15;1671:18-25. [3]. Effect of capsaicin and dihydrocapsaicin on in vitro blood coagulation and platelet aggregation. Thromb Res. 2009 Dec;124(6):721-3. |
| Additional Infomation |
Dihydrocapsaicin is a capsaicinoid. Dihydrocapsaicin has been reported in Capsicum pubescens, Capsicum annuum, and Ganoderma lucidum with data available. See also: Capsicum (part of); Paprika (part of); Habanero (part of) ... View More ... 1. Dihydrocapsaicin is a major capsaicinoid analog isolated from chili peppers (Capsicum annuum), with lower pungency but similar or stronger bioactivities compared to capsaicin [2][3] 2. Mechanism of neuroprotective effect: Dihydrocapsaicin enhances hypothermia-induced neuroprotection in ischemic stroke by activating the PI3K/Akt signaling pathway, which inhibits neuronal apoptosis (reduced cleaved caspase-3) and promotes cell survival in the ischemic penumbra [2] 3. Anti-thrombotic mechanism: Dihydrocapsaicin exerts in vitro anti-thrombotic effects by prolonging coagulation time and inhibiting platelet aggregation, potentially by interfering with platelet activation pathways and coagulation factor function, with no significant cytotoxicity to blood cells at experimental concentrations [3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~325.28 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.13 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.13 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.13 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2528 mL | 16.2639 mL | 32.5277 mL | |
| 5 mM | 0.6506 mL | 3.2528 mL | 6.5055 mL | |
| 10 mM | 0.3253 mL | 1.6264 mL | 3.2528 mL |