Physicochemical Properties
| Molecular Formula | C11H16O2 |
| Molecular Weight | 180.2435 |
| Exact Mass | 180.115 |
| CAS # | 17092-92-1 |
| Related CAS # | (±)-Dihydroactinidiolide;15356-74-8 |
| PubChem CID | 6432173 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 296.1±9.0 °C at 760 mmHg |
| Melting Point | 70-71° |
| Flash Point | 120.2±16.1 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.504 |
| LogP | 2.26 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 13 |
| Complexity | 289 |
| Defined Atom Stereocenter Count | 1 |
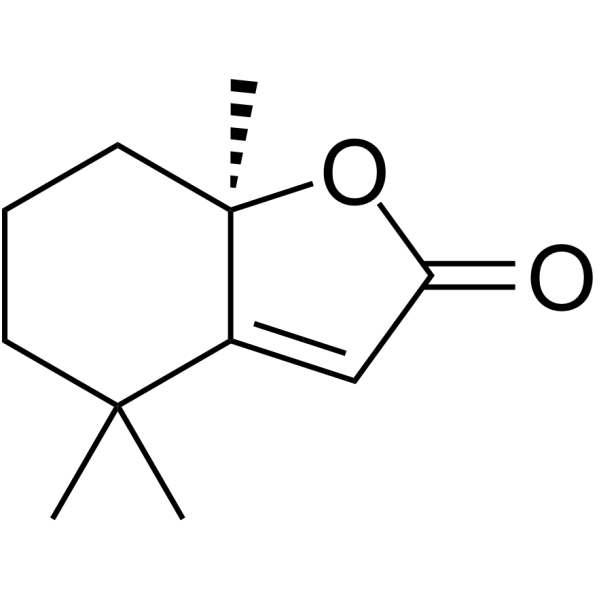
| SMILES | O1C(C([H])=C2C1(C([H])([H])[H])C([H])([H])C([H])([H])C([H])([H])C2(C([H])([H])[H])C([H])([H])[H])=O |
| InChi Key | IMKHDCBNRDRUEB-LLVKDONJSA-N |
| InChi Code | InChI=1S/C11H16O2/c1-10(2)5-4-6-11(3)8(10)7-9(12)13-11/h7H,4-6H2,1-3H3/t11-/m1/s1 |
| Chemical Name | (7aR)-4,4,7a-trimethyl-6,7-dihydro-5H-1-benzofuran-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Dihydroactinidiolide (DA) exhibits potent AChE inhibitory activity with an IC50 of 34.03 nM, which is better than some reported β-carboline and tacrine derivatives. [1] DA shows significant free radical scavenging activity: DPPH radical scavenging IC50 < 50 nM, nitric oxide (·NO) scavenging IC50 < 50 nM, and metal chelating activity IC50 > 270 nM. [1] DA at 270 nM significantly prevents the self-aggregation of Aβ25-35 peptide and promotes the disaggregation of pre-formed Aβ25-35 fibrils, as confirmed by Thioflavin T assay and confocal microscopy. [1] In Neuro2a (N2a) cells, DA (50 and 270 nM) significantly increases cell viability and reduces intracellular ROS generation induced by Aβ25-35 treatment. [1] DA does not show cytotoxic effects on N2a cells up to 270 nM for 24 hours. [1] |
| Enzyme Assay | Acetylcholinesterase (AChE) inhibitory activity was measured using a modified Ellman's method. Briefly, 10 μL of 0.1 U AChE enzyme was incubated with 5.5–55 nM of Dihydroactinidiolide (DA) in a 96-well plate for 45 minutes. The reaction was stopped by adding 50 mM Tris-HCl buffer (pH 8.0). Then, 125 μL of 3 mM DTNB and 50 μL of 15 mM acetylthiocholine iodide (substrate) were added to initiate the reaction. The absorbance was measured at 405 nm. The percentage inhibition was calculated, and the IC50 value was determined using GraphPad Prism software. [1] |
| Cell Assay |
Cell viability was assessed using the MTT assay. Neuro2a (N2a) cells were seeded and treated with different concentrations (50–270 nM) of Dihydroactinidiolide (DA) or the standard drug donepezil for 24 hours. After treatment, MTT reagent was added and incubated for 3 hours. The formed formazan crystals were dissolved in DMSO, and absorbance was measured at 570 nm. Cell viability was expressed as a percentage relative to the control. [1] For the neuroprotective assay, N2a cells were pre-treated with DA (50 and 270 nM) or donepezil (50 μM) for 2 hours, then exposed to 20 μM Aβ25-35 for 24 hours. Cell viability was assessed by MTT assay as described above. [1] Intracellular ROS levels were measured using the fluorescent probe DCFH-DA. N2a cells were pre-treated with DA (50 and 270 nM) or donepezil (50 μM) for 2 hours, then exposed to 20 μM Aβ25-35 for 24 hours. Cells were then incubated with 10 μM DCFH-DA for 30 minutes in the dark. After lysis, fluorescence intensity was measured at excitation/emission wavelengths of 485/535 nm. [1] |
| ADME/Pharmacokinetics |
The ADME properties of Dihydroactinidiolide (DA) were predicted in silico using QikProp and SwissADME tools. Predicted parameters were within normal ranges for drug-like molecules: polarizability 20.26 ų, predicted octanol/water partition coefficient (log Po/w) 1.57, predicted aqueous solubility (log S) -1.98, Caco-2 permeability 2174.55 nm/sec, predicted brain/blood partition coefficient (log BB) -1.72, predicted human oral absorption 95.88%, and zero violations of Lipinski's Rule of Five and Jorgensen's Rule of Three. [1] DA was predicted not to be a Cytochrome P450 inhibitor. [1] Molecular weight is 180.24 g/mol, with 0 hydrogen bond donors and 3 hydrogen bond acceptors. [1] |
| Toxicity/Toxicokinetics |
Cytotoxicity against human peripheral blood mononuclear cells (PBMCs) was evaluated. PBMCs were isolated from healthy human blood and treated with different concentrations of Dihydroactinidiolide (DA) for 24 hours. Cell viability was assessed by MTT assay. DA did not show significant cytotoxicity against PBMCs, whereas 1 mM H2O2 (positive control) decreased viability by 57%. [1] Hemolytic activity was tested using human erythrocytes. Erythrocytes were incubated with DA (50–270 nM) for 1 hour at 37°C. After centrifugation, released hemoglobin in the supernatant was measured at 540 nm. DA showed very low hemolytic activity, ranging from 2.35% to 5.61%, compared to 100% hemolysis by 1% SDS (positive control). [1] |
| References |
[1]. Dihydroactinidiolide, a natural product against Aβ25-35 induced toxicity in Neuro2a cells: Synthesis, in silico and in vitro studies. Bioorg Chem. 2018 Dec;81:340-349. |
| Additional Infomation |
Actinidiolide, dihydro- has been reported in Agastache rugosa, Artemisia annua, and other organisms with data available. See also: Calendula Officinalis Flower (part of). Dihydroactinidiolide (DA) is a natural product, a structural analog of loliolide and a degradation product of carotenoids, found in black tea and tobacco aroma. It is reported as a plant growth inhibitor and a regulator of gene expression. [1] This study is the first to report the neuroprotective potential of synthesized DA against Alzheimer's disease (AD) pathology, demonstrating its multi-targeted profile including AChE inhibition, antioxidant activity, anti-amyloid aggregation, and cytoprotective effects. [1] The compound was synthesized from β-ionone via a two-step oxidation method involving m-chloroperoxybenzoic acid followed by chromic anhydride oxidation. [1] The authors propose DA as a promising multi-target directed ligand (MTDL) candidate for further development in anti-AD therapy. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~554.82 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (13.87 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (13.87 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (13.87 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.5482 mL | 27.7408 mL | 55.4816 mL | |
| 5 mM | 1.1096 mL | 5.5482 mL | 11.0963 mL | |
| 10 mM | 0.5548 mL | 2.7741 mL | 5.5482 mL |