Physicochemical Properties
| Molecular Formula | C36H54CLN7O6 |
| Molecular Weight | 716.31 |
| Exact Mass | 787.335 |
| Elemental Analysis | C, 54.79; H, 7.15; Cl, 13.48; N, 12.42; O, 12.16 |
| CAS # | 2711717-77-8 |
| Related CAS # | 1024829-44-4 (acetate);1024828-77-0;2742623-88-5 (TFA);2711717-77-8 (3HCl); 413256-25-2 (1HCl); |
| PubChem CID | 163284813 |
| Appearance | Solid powder |
| Hydrogen Bond Donor Count | 10 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 52 |
| Complexity | 1080 |
| Defined Atom Stereocenter Count | 4 |
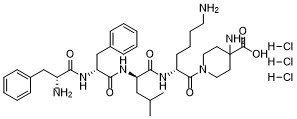
| SMILES | CC(C)C[C@H](C(=O)N[C@H](CCCCN)C(=O)N1CCC(CC1)(C(=O)O)N)NC(=O)[C@@H](CC2=CC=CC=C2)NC(=O)[C@@H](CC3=CC=CC=C3)N.Cl.Cl.Cl |
| InChi Key | AKXASUQJXPQETP-KKUHLPMVSA-N |
| InChi Code | InChI=1S/C36H53N7O6.3ClH/c1-24(2)21-29(32(45)40-28(15-9-10-18-37)34(47)43-19-16-36(39,17-20-43)35(48)49)42-33(46)30(23-26-13-7-4-8-14-26)41-31(44)27(38)22-25-11-5-3-6-12-25;;;/h3-8,11-14,24,27-30H,9-10,15-23,37-39H2,1-2H3,(H,40,45)(H,41,44)(H,42,46)(H,48,49);3*1H/t27-,28-,29-,30-;;;/m1.../s1 |
| Chemical Name | 1-(D-phenylalanyl-D-phenylalanyl-D-leucyl-D-lysyl)-4-aminopiperidine-4-carboxylic acid trihydrochloride |
| Synonyms | Difelikefalin trihydrochloride; Difelikefalin hydrochloride; Difelikefalin HCl; CR845; CR 845; CR-845; FE 202845; FE202845; FE-202845; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References | 1: Difelikefalin Acetate. Am J Health Syst Pharm. 2022 Jan 1;79(1):e1-e2. doi: 10.1093/ajhp/zxab377. PMID: 34849553. 2: Shram MJ, Spencer RH, Qian J, Munera CL, Lewis ME, Henningfield JE, Webster L, Menzaghi F. Evaluation of the abuse potential of difelikefalin, a selective kappa-opioid receptor agonist, in recreational polydrug users. Clin Transl Sci. 2021 Oct 28. doi: 10.1111/cts.13173. Epub ahead of print. PMID: 34708917. 3: Deeks ED. Difelikefalin: First Approval. Drugs. 2021 Nov;81(16):1937-1944. doi: 10.1007/s40265-021-01619-6. PMID: 34674115. 4: Inan S, Dun NJ, Cowan A. Antipruritic Effect of Nalbuphine, a Kappa Opioid Receptor Agonist, in Mice: A Pan Antipruritic. Molecules. 2021 Sep 11;26(18):5517. doi: 10.3390/molecules26185517. PMID: 34576988; PMCID: PMC8466557. 5: Pilla JE, Devulapally P. Difelikefalin. 2021 Aug 31. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan–. PMID: 34662080. 6: Viscusi ER, Torjman MC, Munera CL, Stauffer JW, Setnik BS, Bagal SN. Effect of difelikefalin, a selective kappa opioid receptor agonist, on respiratory depression: A randomized, double-blind, placebo-controlled trial. Clin Transl Sci. 2021 Sep;14(5):1886-1893. doi: 10.1111/cts.13042. Epub 2021 Jul 21. PMID: 33982405; PMCID: PMC8504812. 7: Santos-Alonso C, Maldonado Martín M, Sánchez Villanueva R, Álvarez García L, Vaca Gallardo MA, Bajo Rubio MA, Del Peso Gilsanz G, Ossorio González M, Selgas Gutiérrez R. Pruritus in dialysis patients. Review and new perspectives. Nefrologia (Engl Ed). 2021 Mar 8:S0211-6995(21)00032-1. English, Spanish. doi: 10.1016/j.nefro.2020.12.010. Epub ahead of print. PMID: 33707097. 8: Inan S, Cowan A. Antipruritic Effects of Kappa Opioid Receptor Agonists: Evidence from Rodents to Humans. Handb Exp Pharmacol. 2022;271:275-292. doi: 10.1007/164_2020_420. PMID: 33296031. 9: Lipman ZM, Yosipovitch G. An evaluation of difelikefalin as a treatment option for moderate-to-severe pruritus in end stage renal disease. Expert Opin Pharmacother. 2021 Apr;22(5):549-555. doi: 10.1080/14656566.2020.1849142. Epub 2020 Dec 14. PMID: 33190563. 10: Trachtenberg AJ, Collister D, Rigatto C. Recent advances in the treatment of uremic pruritus. Curr Opin Nephrol Hypertens. 2020 Sep;29(5):465-470. doi: 10.1097/MNH.0000000000000625. PMID: 32740217. 11: Vernon M, Ständer S, Munera C, Spencer RH, Menzaghi F. Clinically meaningful change in itch intensity scores: An evaluation in patients with chronic kidney disease-associated pruritus. J Am Acad Dermatol. 2021 Apr;84(4):1132-1134. doi: 10.1016/j.jaad.2020.06.991. Epub 2020 Jun 27. PMID: 32603719. 12: Fishbane S. Difelikefalin in Hemodialysis Patients with Pruritus. Reply. N Engl J Med. 2020 May 21;382(21):2065-2066. doi: 10.1056/NEJMc2002485. PMID: 32433848. 13: Lin CH. Difelikefalin in Hemodialysis Patients with Pruritus. N Engl J Med. 2020 May 21;382(21):2064-2065. doi: 10.1056/NEJMc2002485. PMID: 32433847. 14: Manenti L, Rossi GM, Fiaccadori E. Difelikefalin in Hemodialysis Patients with Pruritus. N Engl J Med. 2020 May 21;382(21):2064. doi: 10.1056/NEJMc2002485. PMID: 32433846. 15: Brennan F, Davison S, Brown M. Difelikefalin in Hemodialysis Patients with Pruritus. N Engl J Med. 2020 May 21;382(21):2064. doi: 10.1056/NEJMc2002485. PMID: 32433845. 16: Lagacé F, Jfri A, Litvinov IV, Netchiporouk E. Newer and Safer Kappa-Opioid Agonist for Your Patients With Uremic Pruritus. J Cutan Med Surg. 2020 Sep/Oct;24(5):525-526. doi: 10.1177/1203475420926989. Epub 2020 May 19. PMID: 32426990. 17: Fishbane S, Mathur V, Germain MJ, Shirazian S, Bhaduri S, Munera C, Spencer RH, Menzaghi F; Trial Investigators. Randomized Controlled Trial of Difelikefalin for Chronic Pruritus in Hemodialysis Patients. Kidney Int Rep. 2020 Jan 28;5(5):600-610. doi: 10.1016/j.ekir.2020.01.006. PMID: 32405581; PMCID: PMC7210745. 18: Steele DJR. Difelikefalin for the Treatment of Uremic Pruritus. N Engl J Med. 2020 Jan 16;382(3):289-290. doi: 10.1056/NEJMe1916598. PMID: 31940704. 19: Fishbane S, Jamal A, Munera C, Wen W, Menzaghi F; KALM-1 Trial Investigators. A Phase 3 Trial of Difelikefalin in Hemodialysis Patients with Pruritus. N Engl J Med. 2020 Jan 16;382(3):222-232. doi: 10.1056/NEJMoa1912770. Epub 2019 Nov 8. PMID: 31702883. 20: Walker G. The opioid crisis: a 21st century pain. Drugs Today (Barc). 2018 Apr;54(4):283-286. doi: 10.1358/dot.2018.54.4.2812620. PMID: 29869649. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3960 mL | 6.9802 mL | 13.9604 mL | |
| 5 mM | 0.2792 mL | 1.3960 mL | 2.7921 mL | |
| 10 mM | 0.1396 mL | 0.6980 mL | 1.3960 mL |