Physicochemical Properties
| Molecular Formula | C24H31NO4 |
| Molecular Weight | 397.50724 |
| Exact Mass | 397.225 |
| CAS # | 302776-68-7 |
| PubChem CID | 10111431 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 524.8±40.0 °C at 760 mmHg |
| Melting Point | 54, Decomposes at 314 |
| Flash Point | 271.2±27.3 °C |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.565 |
| LogP | 6.93 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 29 |
| Complexity | 505 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | FDATWRLUYRHCJE-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H31NO4/c1-4-7-8-11-16-29-24(28)20-13-10-9-12-19(20)23(27)21-15-14-18(17-22(21)26)25(5-2)6-3/h9-10,12-15,17,26H,4-8,11,16H2,1-3H3 |
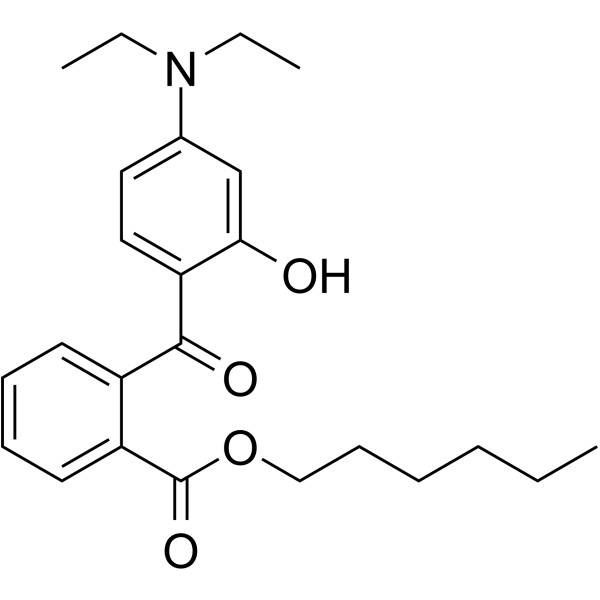
| Chemical Name | hexyl 2-[4-(diethylamino)-2-hydroxybenzoyl]benzoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
UV-A radiation is absorbed by diethylaminohydroxybenzoylhexylbenzoate (DHHB), which peaks at 354 nm. Diethylaminohydroxybenzoyl hexyl benzoate's molar absorption coefficient in ethanol at 25°C was determined to be 39 000 mol/dm3/cm at 354 nm. The wavelength of excitation determines the emission spectra of diethylaminohydroxybenzoylhexyl benzoate in ethanol [1]. Diethylamino hydroxybenzoyl hexyl benzoate exhibits strong absorption in the UV-A region (320–400 nm) with a maximum absorption wavelength (λmax) ranging from 354 to 358 nm in different solvents (methanol, ethanol, isopropanol, and octanol). [1] The molar extinction coefficient (ε) of the compound at λmax is approximately 42,000 M⁻¹cm⁻¹, indicating high UV-A absorption capacity. [1] Fluorescence measurements show that the compound has weak fluorescence emission, with a quantum yield (Φf) less than 0.01 in all tested solvents, suggesting efficient non-radiative deactivation of excited states. [1] Photostability assays demonstrate that the compound retains over 95% of its initial absorption intensity after 2 hours of UV-A irradiation (365 nm, 10 mW cm⁻²) in both solution and thin film forms, indicating good photostability. [1] The compound shows high solubility (≥100 mg mL⁻¹) in common organic solvents used in cosmetic formulations, including ethanol, isopropanol, and octanol. [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Based on the findings of a rat study assessing dermal or percutaneous absorption, only a minor amount of diethylamino hydroxybenzoyl hexyl benzoate will undergo percutaneous absorption and most of the compounds will remain in the upper layers of the stratum corneum. In rat and porcine skin, the percutaneous absorption was 0.10 ± 0.12 μg/cm^2 or 0.04 ± 0.05%. No pharmacokinetic data. No pharmacokinetic data. No pharmacokinetic data. Metabolism / Metabolites In steady-state and transient photolysis experiments _in vitro_, diethylamino hydroxybenzoyl hexyl benzoate may be oxidized by H2O2/UV to form degradation products. Like other UV filters, diethylamino hydroxybenzoyl hexyl benzoate may be subject to photodegradation upon absorption of UV radiation, which results in the compound passing from a ground state to either a singlet or triplet excited state. Biological Half-Life No pharmacokinetic data. |
| Toxicity/Toxicokinetics |
Protein Binding No pharmacokinetic data. |
| References |
[1]. Photophysical properties of hexyl diethylaminohydroxybenzoylbenzoate (Uvinul A Plus), a UV-A absorber. Photochem Photobiol Sci. 2017 Sep 13;16(9):1449-1457. |
| Additional Infomation |
Diethylamino hydroxybenzoyl hexyl benzoate is a member of benzophenones. Diethylamino hydroxybenzoyl hexyl benzoate is a UV filter with high absorption in the UV-A range. Minimizing the overexposure of human skin to ultraviolet radiation that may lead to acute and chronic photodamage, diethylamino hydroxybenzoyl hexyl benzoate is an oil-soluble UV filter that may be incorporated in the oil phase of emulsions. Diethylamino hydroxybenzoyl hexyl benzoate was approved in Europe in 2005, and is also marketed in the U.S., South America, Mexico, Japan and Taiwan. It has a chemical structure similar to the classical benxophoenone drug class, and displays good photostability. It is used in concentrations up to 10% in sunscreen products, either alone or in combination with other UV absorbers. See also: ... View More ... Drug Indication Indicated for use as a sunscreen agent. Mechanism of Action Diethylamino hydroxybenzoyl hexyl benzoate absorbs in the UV-A range with the peak at 354 nm. Pharmacodynamics Diethylamino hydroxybenzoyl hexyl benzoate is an organic UV filter that attenuates the exposure of UV radiation on human skin. _In vitro_, topical application of diethylamino hydroxybenzoyl hexyl benzoate exerted an anti-inflammatory effect on inflammation-evoked mouse ears by inhibiting oedema formation. Diethylamino hydroxybenzoyl hexyl benzoate (trade name Uvinul A Plus) is a widely used UV-A absorber in cosmetic and personal care products. [1] Its primary function is to absorb UV-A radiation, thereby protecting the skin from UV-induced damage such as photoaging and DNA damage. [1] The study focuses on characterizing its photophysical properties (absorption, fluorescence, photostability, solubility) to support its application in sunscreen formulations, as these properties are critical for the efficacy and stability of UV protection products. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~125.78 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5157 mL | 12.5783 mL | 25.1566 mL | |
| 5 mM | 0.5031 mL | 2.5157 mL | 5.0313 mL | |
| 10 mM | 0.2516 mL | 1.2578 mL | 2.5157 mL |