Physicochemical Properties
| Molecular Formula | C19H35NO2 |
| Exact Mass | 309.267 |
| CAS # | 77-19-0 |
| Related CAS # | Dicyclomine hydrochloride;67-92-5;Dicyclomine-d4 |
| PubChem CID | 3042 |
| Appearance | Typically exists as solid at room temperature |
| Melting Point | 165-166 |
| LogP | 4.402 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 22 |
| Complexity | 326 |
| Defined Atom Stereocenter Count | 0 |
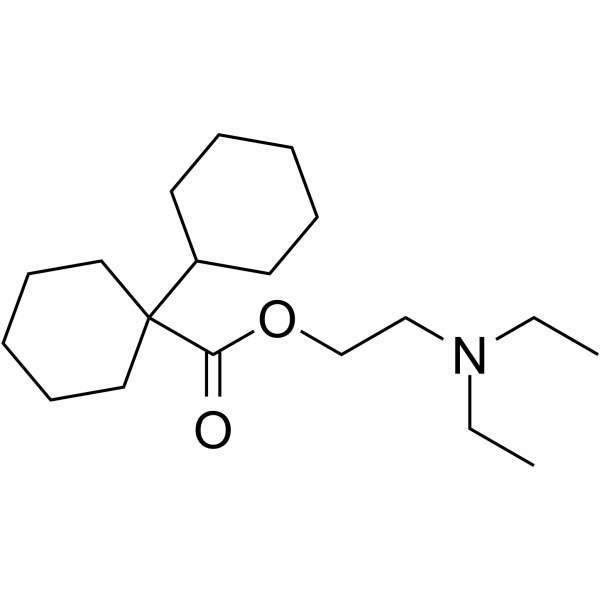
| SMILES | CCN(CC)CCOC(=O)C1(CCCCC1)C2CCCCC2 |
| InChi Key | CURUTKGFNZGFSE-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H35NO2/c1-3-20(4-2)15-16-22-18(21)19(13-9-6-10-14-19)17-11-7-5-8-12-17/h17H,3-16H2,1-2H3 |
| Chemical Name | 2-(diethylamino)ethyl 1-cyclohexylcyclohexane-1-carboxylate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Dicyclomine (diceverine) (ip; 8 mg/kg; daily) increased cognitive impairment in all tests. In addition, memory impairment in dicyclomine-treated 3xTg-AD mice was more severe compared with dicyclomine-treated NonTg mice [2]. Dicyclooverine (ip; 2.0, 4.0, and 8.0 mg/kg; 7 days) produced extremely significant impacts on performance of the paired-associate learning (PAL) test in mice. Lower doses of systemic therapy indicated behavioral impairment in mice during spatial tests [3]. |
| Animal Protocol |
Animal/Disease Models: C57Bl/6 mice [1] Doses: 2.0, 4.0 and 8.0 mg/kg Route of Administration: intraperitoneal (ip) injection; daily; 7 days Experimental Results: Damage due to factors other than the hippocampus. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The bioavailability of dicyclomine has not been determined, though it is likely well absorbed as the primary route of elimination is in the urine. Dicyclomine has a Tmax of 1-1.5h. Dicyclomine is 79.5% eliminated in the urine and 8.4% in the feces. The volume of distribution for a 20mg oral dose is 3.65L/kg. Data regarding the clearance of dicyclomine is not readily available. Metabolism / Metabolites The metabolism of dicyclomine has not been well researched. Biological Half-Life The mean plasma elimination half life is approximately 1.8 hours. |
| Toxicity/Toxicokinetics |
Hepatotoxicity Like other anticholinergic agents, dicyclomine has not been linked to episodes of liver enzyme elevations or clinically apparent liver injury. The metabolism of dicyclomine is not well defined but it is likely metabolized by the liver. References on the safety and potential hepatotoxicity of anticholinergics are given together in the Overview section on Anticholinergic Agents. Drug Class: Anticholinergic Agents Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Dicyclomine has not been well studied during breastfeeding. However, one possible case of apnea has been reported in a breastfed infant that is similar to reactions that have occurred in infants given the drug directly. Dicyclomine should not be used during lactation. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. The manufacturer reported a breastfed infant who developed apnea during maternal therapy with dicyclomine. Dicyclomine is a possible cause of the reaction. ◉ Effects on Lactation and Breastmilk Relevant published information in nursing mothers was not found as of the revision date. Anticholinergics can inhibit lactation in animals, apparently by inhibiting growth hormone and oxytocin secretion. Anticholinergic drugs can also reduce serum prolactin in nonnursing women. The prolactin level in a mother with established lactation may not affect her ability to breastfeed. Protein Binding Data regarding plasma protein binding of dicyclomine is not readily available. Interactions ...ANTICHOLINERGIC AGENTS, SUCH AS...DICYCLOMINE...WOULD BE EXPECTED TO INTERACT WITH DIGOXIN... |
| References |
[1]. M1 Receptors Play a Central Role in Modulating AD-like Pathology in Transgenic Mice. 2006 Mar 2;49(5):671-82.doi: 10.1016/j.neuron.2006.01.020. [2]. M1 Receptors Play a Central Role in Modulating AD-like Pathology in Transgenic Mice. 2006 Mar 2;49(5):671-82.doi: 10.1016/j.neuron.2006.01.020. [3]. A Computer-Automated Touchscreen Paired-Associates Learning (PAL) Task for Mice: Impairments Following Administration of Scopolamine or Dicyclomine and Improvements Following Donepezil.Psychopharmacology (Berl). 2011 Mar;214(2):537-48. |
| Additional Infomation |
Dicyclomine is the ester resulting from the formal condensation of 1-cyclohexylcyclohexanecarboxylic acid with 2-(diethylamino)ethanol. An anticholinergic, it is used as the hydrochloride to treat or prevent spasm in the muscles of the gastrointestinal tract, particularly that associated with irritable bowel syndrome. It has a role as a muscarinic antagonist, an antispasmodic drug and a parasympatholytic. It is a tertiary amine and a carboxylic ester. It is functionally related to a 2-diethylaminoethanol and a 1,1'-bi(cyclohexyl)-1-carboxylic acid. Dicyclomine is a muscarinic M1, M3, and M2 receptor antagonist as well as a non-competitive inhibitor of histamine and bradykinin used to treat spasms of the intestines seen in functional bowel disorder and irritable bowel syndrome. Though it is commonly prescribed, its recommendation may have been based on a small amount of evidence and so its prescription is becoming less favourable. Dicyclomine was granted FDA approval on 11 May 1950. Dicyclomine is an Anticholinergic. The mechanism of action of dicyclomine is as a Cholinergic Antagonist. Dicyclomine is an anticholinergic agent used to treat gastrointestinal conditions such as acid peptic disease and irritable bowel syndrome. Dicyclomine has not been implicated in causing liver enzyme elevations or clinically apparent acute liver injury. Dicyclomine is a carboxylic acid derivative and a selective anticholinergic with antispasmodic activity. Dicyclomine blocks acetylcholine from binding to muscarinic receptors on smooth muscle. This agent has a direct relaxing effect on smooth muscle and therefore prevents spasms in the muscles of the gastrointestinal tract, inhibits gastrointestinal propulsive motility, decreases gastric acid secretion and controls excessive pharyngeal, tracheal and bronchial secretion. A muscarinic antagonist used as an antispasmodic and in urinary incontinence. It has little effect on glandular secretion or the cardiovascular system. It does have some local anesthetic properties and is used in gastrointestinal, biliary, and urinary tract spasms. See also: Dicyclomine Hydrochloride (has salt form). Drug Indication Dicyclomine is indicated for the treatment of functional bowel disorder and irritable bowel syndrome. FDA Label Mechanism of Action Dicyclomine achieves its action partially through direct antimuscarinic activity of the M1, M3, and M2 receptors; and partially through antagonism of bradykinin and histamine. Dicyclomine non-competitively inhibits the action of bradykinin and histamine, resulting in direct action on the smooth muscle, and decreased strength of contractions seen in spasms of the ileum. ...MAJOR ACTION APPEARS TO BE NONSPECIFIC DIRECT RELAXANT ACTION ON SMOOTH MUSLCE RATHER THAN COMPETITIVE ANTAGONISM OF ACH. Therapeutic Uses Muscarinic Antagonists; Parasympatholytics ...OFTEN CLASSIFIED WITH ANTIMUSCARINIC AGENTS AS "ANTISPASMODICS," DO NOT PROPERLY BELONG TO GROUP OF ANTIMUSCARINIC AGENTS. ...DICYCLOMINE HYDROCHLORIDE, VSP-DECR SPASM OF GI TRACT, BILIARY TRACT, URETER, & UTERUS WITHOUT PRODUCING CHARACTERISTIC ATROPINIC EFFECTS ON SALIVARY, SWEAT, OR GI GLANDS, EYE, CV SYSTEM... /HCL/ IT DECR MOTILITY BUT DOES NOT SUPPRESS GASTRIC SECRETION. IT IS USED IN TREATMENT OF IRRITABLE COLON, SPASTIC CONSTIPATION, MUCOUS COLITIS, SPASTIC COLITIS, PYLOROSPASM, & BILIARY DYSKINESIA. IN TREATMENT OF PEPTIC ULCER IT IS USED TO DELAY GASTRIC EMPTYING. /HYDROCHLORIDE/ ANTICHOLINERGIC /HYDROCHLORIDE/ Drug Warnings DICYCLOMINE SHOULD BE USED CAUTIOUSLY IN PT WITH PROSTATIC HYPERTROPHY, BLADDER NECK OBSTRUCTION, PYLORIC OBSTRUCTION, & CARDIOSPASM. EVEN THOUGH IT DOES NOT APPEAR TO RAISE INTRAOCULAR PRESSURE IN NARROW-ANGLE GLAUCOMA, IT IS ADVISABLE TO MONITOR PRESSURE OF SUCH PT. CLINICAL USE OF.../BENTYL/ HAS BEEN DISAPPOINTING. A 3-YR-OLD MALE INGESTED APPROX 100 TABLETS OF BENDECTIN & DEVELOPED TONIC-CLONIC SEIZURES FOLLOWED BY CARDIAC ARREST. ANALYSIS YIELDED HIGH LEVELS OF DOXYLAMINE, DICYCLOMINE & PYRIDOXINE. DOXYLAMINE APPEARS TO BE TOXIC CONSTITUENT. Pharmacodynamics Dicyclomine is an anticholinergic drug used to relax the smooth muscles of the intestines. It's duration of action is not especially long as it is usually taken 4 times daily with individual doses of 20-40mg orally or 10-20mg by intramuscular injection. Dicyclomine should not be administered intravenously. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |