Physicochemical Properties
| Molecular Formula | C16H14CL2O4 |
| Molecular Weight | 341.19 |
| Exact Mass | 340.026 |
| CAS # | 51338-27-3 |
| PubChem CID | 39985 |
| Appearance | Colorless crystals |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 408.0±45.0 °C at 760 mmHg |
| Melting Point | 39-41°C |
| Flash Point | 150.3±27.7 °C |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.562 |
| LogP | 4.24 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 22 |
| Complexity | 358 |
| Defined Atom Stereocenter Count | 0 |
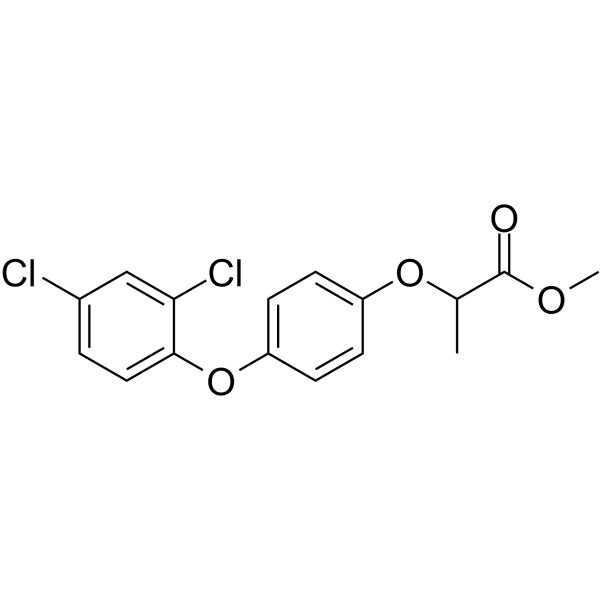
| SMILES | CC(C(=O)OC)OC1=CC=C(C=C1)OC2=C(C=C(C=C2)Cl)Cl |
| InChi Key | BACHBFVBHLGWSL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H14Cl2O4/c1-10(16(19)20-2)21-12-4-6-13(7-5-12)22-15-8-3-11(17)9-14(15)18/h3-10H,1-2H3 |
| Chemical Name | methyl 2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | One micromolar of diclofop-methyl dramatically decreased the steady-state H(+) gradient produced in the presence of ATP[1]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion ESTERS NORMALLY EXHIBIT GREATER HERBICIDAL ACTIVITY THAN PARENT ACIDS, BECAUSE OF IMPROVED ABSORPTION BY TARGET PLANTS. /PHENOXYALKANOIC ACIDS/ The chlorophenoxy compounds are absorbed across the gut wall, lung, and skin. They are not significantly fat storable. Excretion occurs almost entirely by way of the urine. /Chlorphenoxy herbicides/ Metabolism / Metabolites Diclofop-methyl undergoes extensive hydrolysis to its corresponding carboxylic acid in plants in soil. It is then decarboxylated to phenyl ethyl ether and transformed to the phenol in soil. The phenol may undergo complexing or binding to soil and/or be subjected to further degradative processes involving splitting of the ether linkage to give such products as m-dichlorobenzene or 2,4-dichlorophenol. Ring hydroxylation at the 5-position of the 2,4-dichlorophenol moiety was observed but not the other positions of the phenyl rings in plants and soil. This herbicide was applied to wheat at the rate of 1 kg/ha. Analyses of plant material indicated the presence of unchanged herbicide in addition to a number of metabolites. The main metabolites were identified with GC-MS as the free acid, 5'-OH-analog, and 3'-OH- and 6'-OH-analogs of the free acid as plant conjugates. The breakdown of diclofop-methyl in moist nonsterile clays and sandy loam was investigated. Rapid hydrolysis to the acid occurred initially. This bound tightly to the soils. Subsequently, there was decarboxylation to the ethyl analog and cleavage of the ethyl moiety to produce 2,4-dichlorophenoxy-p-phenol. In soils, diclofop-methyl ester bond was rapidly hydrolyzed and the substituted propionic acid was aerobically oxidized to carbon dioxide. An intermediate was identified as 4-(2,4-dichlorophenoxy)phenol. Under anaerobic conditions, only a small degree of degradation occurred. |
| Toxicity/Toxicokinetics |
Toxicity Data LC50 (rat) = 8,300 mg/m3/4h Non-Human Toxicity Values LD50 Rat percutaneous >5000 mg/kg LD50 Rat female dermal >5000 mg/kg LD50 Rat acute oral 580 mg/kg |
| References |
[1]. Ratterman DM, et al. Diclofop-methyl increases the proton permeability of isolated oat-root tonoplast. Plant Physiol. 1989;91(2):756-765. [2]. Li X, et al. The interactive effects of diclofop-methyl and silver nanoparticles on Arabidopsis thaliana: Growth, photosynthesis and antioxidant system. Environ Pollut. 2018;232:212-219. |
| Additional Infomation |
Diclofop-Methyl can cause cancer and developmental toxicity according to The Environmental Protection Agency (EPA). Diclofop methyl appears as colorless crystals. Decomposed by either strong acid or base. Used as a selective herbicide. Methyl 2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate is a methyl ester resulting from the formal condensation of the carboxylic acid group of 2-[4-(2,4-dichlorophenoxy)phenoxy]propanoic acid with methanol. It is an aromatic ether, a dichlorobenzene, a diether and a methyl ester. Herbicide for control of wild oat & foxtails in cereal crops. Diclofop-methyl is a polycyclic alkanoic acid herbicide. Diclofop-methyl undergoes hydrolysis to form diclofop-acid a compound that also exhibits herbicidal properties. Diclofop-methyl was registered in Canada in 1977 and is sold under the tradename Hoe-Grass. Diclofop-methyl is used for postemergence control of wild oats, wild millets, and other annual grass weeds in wheat, barley, rye, red fescue, and broad-leaved crops such as soya beans, sugar beet, flax, legumes, potatoes, and cucumbers. Diclofop-methyl is a selective systemic herbicide that is used primarily in the prairies. It destroys the cell membrane, prevents the translocation of assimilates to the roots, reduces the chlorophyll content, and inhibits photosynthesis and meristem activity. Mechanism of Action In plants, these chemicals /chlorophenoxy compounds/ mimic the action of auxins, hormones chemically related to indoleacetic acid that stimulate growth. No hormonal activity is observed in mammals and other species, and beyond target organ toxicity that can be associated with the pharmacokinetics, biotransformation, and/or elimination of these chemicals, their mechanism(s) of toxic action are poorly understood. /Chlorophenoxy compounds/ |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9309 mL | 14.6546 mL | 29.3092 mL | |
| 5 mM | 0.5862 mL | 2.9309 mL | 5.8618 mL | |
| 10 mM | 0.2931 mL | 1.4655 mL | 2.9309 mL |