Physicochemical Properties
| Molecular Formula | C10H17CL2NOS |
| Molecular Weight | 270.22 |
| Exact Mass | 269.041 |
| CAS # | 2303-16-4 |
| PubChem CID | 5284376 |
| Appearance |
BROWN LIQUID Oily liquid |
| Density | 1.18g/cm3 |
| Boiling Point | 306ºC at 760mmHg |
| Melting Point | 25-30ºC |
| Flash Point | >100 °C |
| Vapour Pressure | 0.000791mmHg at 25°C |
| Index of Refraction | 1.52 |
| LogP | 4.277 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 15 |
| Complexity | 234 |
| Defined Atom Stereocenter Count | 0 |
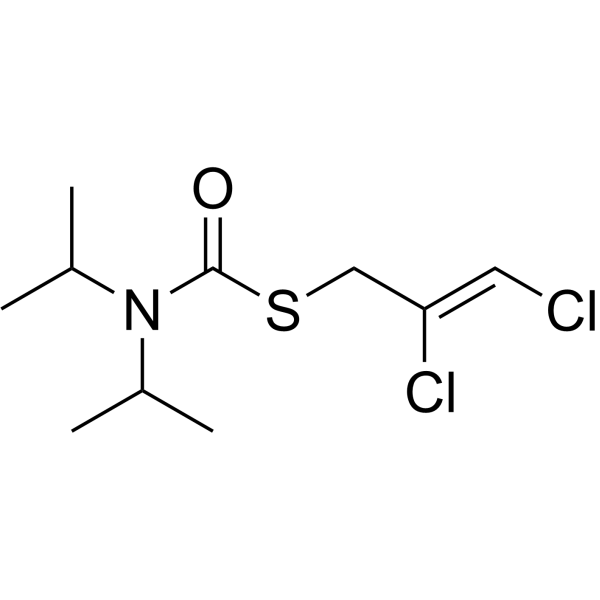
| SMILES | CC(C)N(C(C)C)C(=O)SC/C(=C/Cl)/Cl |
| InChi Key | SPANOECCGNXGNR-UITAMQMPSA-N |
| InChi Code | InChI=1S/C10H17Cl2NOS/c1-7(2)13(8(3)4)10(14)15-6-9(12)5-11/h5,7-8H,6H2,1-4H3/b9-5- |
| Chemical Name | S-[(Z)-2,3-dichloroprop-2-enyl] N,N-di(propan-2-yl)carbamothioate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion ORAL DOSE LARGELY (88%) ELIMINATED IN 48 HR. IN WILD OAT, PRIMARY ABSORPTION IS THROUGH EMERGING COLEOPTILE. EXTREMELY MINOR UPTAKE AT EARLY STAGES OF SEEDLING DEVELOPMENT. ... UPTAKE & TRANSLOCATION OF (14)CARBON FROM LABELED DIALLATE BY ROOTS & COLEOPTILES OF WILD OAT (AVENA FATUA L), WHEAT (TRITICUM AESTIVUM L VAR SELKIRK), BARLEY (HORDEUM VULGARE L VOR TRAIL), FLAX (LINUM USITATISSIMUM L VAR BOLLEY), /WAS STUDIED/ & CONCLUDED ... PATTERN OF ... UPTAKE & MOVEMENT WAS SIMILAR ... Metabolism / Metabolites MOUSE HEPATIC MICROSOMES METABOLIZE CIS- & TRANS-(14)C=O-DIALLATE IN NADPH-DEPENDENT REACTION, YIELDING PRIMARILY (14)CO2 IN ABSENCE OF GLUTATHIONE (GSH) & (14)CO2 & S-(DIISOPROPYLCARBAMOYL)-GSH IN PRESENCE OF GSH. RATS ADMIN EITHER ISOMER EXCRETE S-DIISOPROPYLCARBAMOYL DERIV OF MERCAPTURIC ACID (62%), CYSTEINE (7%) & MERCAPTOACETIC ACID (1.5%) IN ADDN TO (14)CO2 (20%). PATHWAY APPEARS TO INVOLVE SULFOXIDATION, NONENZYMATIC REACTION OF SULFOXIDE WITH GSH & MERCAPTURIC ACID FORMATION. RATS TREATED ORALLY WITH SEVERAL PROMUTAGENS & CARCINOGENS CONTAINING HALOALLYL OR HALOPROPYL SUBSTITUENTS EXCRETE SMALL AMT OF URINARY 2-HALOACRYLIC ACIDS SUCH AS 2-CHLOROACRYLIC ACID & 2,3-DICHLOROACRYLIC ACID FROM DIALLATE. METABOLISM OF DIALLATE ISOMERS PROCEEDS VIA THEIR SULFOXIDES TO FORM 2-CHLOROACROLEIN IN MOUSE HEPATIC MICROSOMAL OXIDASE SYSTEM & 2,3-DICHLORO-2-PROPENE-1-SULFONIC ACID IN MICE & RATS IN VIVO & THEIR LIVER OXIDASE PREPN. The proposed metabolites of diallate, triallate, and sulfallate were identified and quantitated by HPLC and GLC headspace analysis as chloroacroleins and chloroallylthiols. The quantitative relationships indicate that the thiocarbamates yield chloroacroleins on metabolic activation with the mixed function oxidase system alone, while the intermediate S-oxidation products were detoxified on diversion to chloroallylthiols when the glutathione/glutathione-S-transferase system was also present. The major mouse microsomal mixed function oxidase metabolites of three compounds were identified by HPLC cochromatography as the corresponding sulfoxides. It was concluded that the formation of mutagenic chloroacroleins involves primarily sulfoxidation of diallate followed by sigmatropic rearrangement-1,2-elimination reactions and S-methylene hydroxylation of triallate and sulfallate and then decomposition of their alpha-hydroxy intermediates. Competing glutathione-S-transferase catalyzed conjugations with glutathione divert the sulfoxidized intermediates from activation involving chloroacrolein formation to detoxification on chloroallylthiol liberation. As a general rule, thiocarbamates can be absorbed via the skin, mucous membranes, and the respiratory and gastrointestinal tracts. They are eliminated quite rapidly, mainly via expired air and urine. Two major pathways exist for the metabolism of thiocarbamates in mammals. One is via sulfoxidation and conjugation with glutathione. The conjugation product is then cleaved to a cysteine derivative, which is metabolized to a mercapturic acid compound. The second route is oxidation of the sulfur to a sulfoxide, which is then oxidized to a sulfone, or hydroxylation to compounds that enter the carbon metabolic pool. |
| Toxicity/Toxicokinetics |
Toxicity Summary The metabolic products of triallate, diallate and sulfallate appear to be mutagenic or carcinogenic. In particular, the 2-chloro-allyl group is responsible for the mutagenicity of these herbicides. These metabolites likely bind to or disrupt DNA causes base-pair subsitutions. Diallate has been shown to be a carcinogen in mice. Some thiocarbamates (EPTC, Molinate, Pebulate, and Cycloate) share a common mechanism of toxicity, i.e. the inhibition of acetylcholinesterase. An acetylcholinesterase inhibitor suppresses the action of acetylcholine esterase. Because of its essential function, chemicals that interfere with the action of acetylcholine esterase are potent neurotoxins, causing excessive salivation and eye-watering in low doses. Headache, salivation, nausea, vomiting, abdominal pain and diarrhea are often prominent at higher levels of exposure. Acetylcholine esterase breaks down the neurotransmitter acetylcholine, which is released at nerve and muscle junctions, in order to allow the muscle or organ to relax. The result of acetylcholine esterase inhibition is that acetylcholine builds up and continues to act so that any nerve impulses are continually transmitted and muscle contractions do not stop. Interactions Field experiments were conducted in 1979 and 1980 to determine the effect of diallate alone and in combination with aldicarb on Heterodera schachtii population development in sugar beet. Cyst numbers were determined before pesticide treatment in March, after development of white females in July, and at harvest time in October; And egg and larval densities were determined in March and October. Cyst production following diallate treatment increased 3 fold in July 1979, 1.5 fold in July 1980 and 2 fold at harvest time 1980 over control values. The combination of diallate/aldicarb resulted in the greatest reduction in cyst production at all sampling dates. The number of eggs and larvae per 100 cc soil increased in 1979 from March to October 3 fold in untreated plots and 5 fold with diallate, 1.8 fold with aldicarb alone and only 1.1 fold in the diallate/aldicarb combinations. In 1980, egg and larval numbers per 100 cc soil decreased 31% in the untreated control but remained unchanged in diallate treated plots. The diallate/aldicarb combination, however, reduced egg and larval numbers by 52%. Thus, diallate enhances the effectiveness of the nematocide aldicarb against the sugar beet cyst nematode. Non-Human Toxicity Values LD50 Rat oral 395 mg/kg LD50 Dog oral 510 mg/kg LD50 Rabbit percutaneous 2000-2500 mg/kg |
| References |
[1].Evaluation of diallate and triallate herbicides for genotoxic effects in a battery of in vitro and short-term in vivo tests. Mutat Res. 1984 Jun;136(3):173-83. |
| Additional Infomation |
Used as an herbicide. Diallat is a tertiary amine. Diallate is a thiocarbamate herbicide used to control weeds and grasses amongst crops. It can be applied to the soil before planting or to the growing crops. It is used on alfalfa, alsike clover, barley, corn, flax, soybeans, lentils, peas, potatoes, red clover, sugar beets and sweet clover. Thiocarbamates are mainly used in agriculture as insecticides, herbicides, and fungicides. Additional uses are as biocides for industrial or other commercial applications, and in household products. Some are used for vector control in public health. Thiocarbamates are mostly liquids or solids with low melting points. |
Solubility Data
| Solubility (In Vitro) | Typically soluble in DMSO (e.g. 10 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.7007 mL | 18.5034 mL | 37.0069 mL | |
| 5 mM | 0.7401 mL | 3.7007 mL | 7.4014 mL | |
| 10 mM | 0.3701 mL | 1.8503 mL | 3.7007 mL |