Physicochemical Properties
| Molecular Formula | C23H25NO6 |
| Molecular Weight | 411.44770693779 |
| Exact Mass | 411.168 |
| Elemental Analysis | C, 67.14; H, 6.12; N, 3.40; O, 23.33 |
| CAS # | 72584-75-9 |
| PubChem CID | 11732365 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.244 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 30 |
| Complexity | 656 |
| Defined Atom Stereocenter Count | 1 |
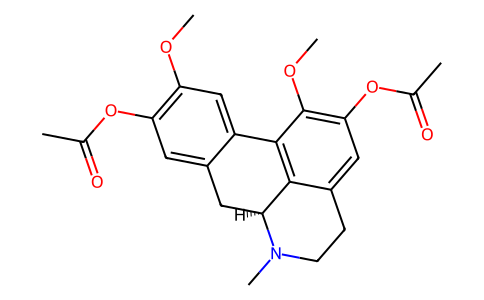
| SMILES | N1(C)[C@]2([H])C3=C(C(OC)=C(OC(=O)C)C=C3CC1)C1=CC(OC)=C(OC(=O)C)C=C1C2 |
| InChi Key | XMEDXTRRSJHOLZ-KRWDZBQOSA-N |
| InChi Code | InChI=1S/C23H25NO6/c1-12(25)29-19-10-15-8-17-21-14(6-7-24(17)3)9-20(30-13(2)26)23(28-5)22(21)16(15)11-18(19)27-4/h9-11,17H,6-8H2,1-5H3/t17-/m0/s1 |
| Chemical Name | [(6aS)-9-acetyloxy-1,10-dimethoxy-6-methyl-5,6,6a,7-tetrahydro-4H-dibenzo[de,g]quinolin-2-yl] acetate |
| Synonyms | Diacetyl boldine; Diacetylboldine; Lumisphere; Lumiskin; 72584-75-9; O,O-Diacetylboldine; UNII-37727Z7M0I; 37727Z7M0I; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural product |
| ln Vivo | 1. Clinical Efficacy and Formulation Composition: - Literature [1] evaluated the efficacy of two compound formulations containing Diacetyl boldine (Formulation A and B) combined with sunscreen in treating facial melasma. Formulation A contained Diacetyl boldine (concentration unspecified), TGF-β1 biomimetic oligopeptide-68, magnesium ascorbyl phosphate, niacinamide, tranexamic acid, and glycolic acid; Formulation B additionally included salicylic acid. - The clinical trial enrolled 120 patients, randomly divided into three groups: Formulation A+B group (combined use of both formulations), 4% hydroquinone group, and control group (sunscreen only). After 12 weeks of treatment, the Formulation A+B group showed significantly better improvement in the Melasma Area and Severity Index (MASI) score compared to the control group (p<0.01) and demonstrated efficacy comparable to the 4% hydroquinone group (p>0.05). Patient self-assessment indicated higher satisfaction in the formulation group regarding spot lightening, skin tone uniformity, and skin texture improvement [1]. 2. Proposed Mechanism of Action: - Diacetyl boldine, as the core component, may reduce melanin synthesis by inhibiting tyrosinase activity. Its mechanism of action is similar to that of hydroquinone, although the literature did not provide specific experimental data to support this. Other components in the formulations (e.g., glycolic acid and salicylic acid) synergistically enhanced the depigmentation effect by promoting keratinocyte metabolism [1]. 3. Safety Assessment: - The formulation groups were well-tolerated, with only 3 cases reporting mild skin dryness and no severe adverse reactions. In the 4% hydroquinone group, 12 cases experienced irritant reactions such as erythema and desquamation, with 2 cases withdrawing from the trial due to intolerance. This suggests that the Diacetyl boldine-containing formulations have a better safety profile compared to traditional hydroquinone therapy [1]. |
| References | [1]. Combined use of two formulations containing diacetyl boldine, TGF-β1 biomimetic oligopeptide-68 with other hypopigmenting/exfoliating agents and sunscreen provides effective and convenient treatment for facial melasma. Either is equal to or is better than 4% hydroquinone on normal skin. J Cosmet Dermatol. 2016 Jun;15(2):131-44. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4304 mL | 12.1521 mL | 24.3043 mL | |
| 5 mM | 0.4861 mL | 2.4304 mL | 4.8609 mL | |
| 10 mM | 0.2430 mL | 1.2152 mL | 2.4304 mL |