Physicochemical Properties
| Molecular Formula | C26H36N2O4 |
| Molecular Weight | 454.611g/mol |
| Exact Mass | 454.283 |
| Elemental Analysis | C, 70.88; H, 8.24; N, 6.36; O, 14.53 |
| CAS # | 38321-02-7 |
| Related CAS # | Verapamil;52-53-9;Verapamil-d3 hydrochloride;Verapamil-d6 hydrochloride;1185032-80-7;Verapamil-d3-1 hydrochloride;2714485-49-9 |
| PubChem CID | 65808 |
| Appearance | White to off-white solid powder |
| Melting Point | 129-131ºC |
| Index of Refraction | 1.529 |
| LogP | 5.093 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 33 |
| Complexity | 606 |
| Defined Atom Stereocenter Count | 1 |
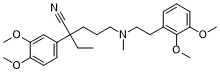
| SMILES | CC(C)[C@@](CCCN(C)CCC1=CC(=C(C=C1)OC)OC)(C#N)C2=CC(=C(C=C2)OC)OC |
| InChi Key | FTZCBMKQDCJEKJ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H36N2O4/c1-7-26(19-27,21-12-13-22(29-3)24(18-21)31-5)15-9-16-28(2)17-14-20-10-8-11-23(30-4)25(20)32-6/h8,10-13,18H,7,9,14-17H2,1-6H3 |
| Chemical Name | (+)-3-(3,4-Dimethoxyphenyl)-6-((5,6-dimethoxyphenethyl)methylamino)hexane-3-carbonitrile |
| Synonyms | Dexverapamil; Dexverapamilum; EINECS 253-878-4 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Calcium channel |
| ln Vitro | Uptake of EverFluor FL Verapamil (EFV) by TR-iBRB2 cells is blocked by the cationic medication Verapamil in a concentration-dependent manner with an IC50 of 98.0 μM [4]. |
| ln Vivo | In addition to controlling the atrioventricular nodal response in atrial fibrillation, oral verapamil can be utilized to prevent atrioventricular reentrant tachycardia [2]. Before ischemia, verapamil was administered intravenously into the femoral vein. Within 45 minutes following coronary artery closure, the incidence of ventricular arrhythmias, such as ventricular tachycardia (VT), ventricular fibrillation (VF), and premature ventricular contractions (PVCs), was considerably reduced by verapamil (1 mg/kg). An ischemic heart resulted in a considerable increase in the total arrhythmia score. The total arrhythmia score's ischemia-induced increase was dramatically inhibited by verapamil (1 mg/kg) [5]. |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Dexverapamil has known human metabolites that include 2-(3,4-dimethoxyphenyl)acetaldehyde, (R)-D-617, arverapamil, and (2R)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(4-hydroxy-3-methoxyphenyl)-2-(propan-2-yl)pentanenitrile. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Limited information indicates that maternal doses of verapamil up to 360 mg daily produce low levels in milk. Newborns may have detectable verapamil serum levels, but levels are low. Verapamil would not be expected to cause any adverse effects in breastfed infants, especially if the infant is older than 2 months. ◉ Effects in Breastfed Infants No adverse reactions have been reported among 3 infants aged 13 days, 8 weeks and 3 months who were exposed to verapamil in breastmilk. ◉ Effects on Lactation and Breastmilk Verapamil can cause hyperprolactinemia and galactorrhea. The clinical relevance of these findings in nursing mothers is not known. The maternal prolactin level in a mother with established lactation may not affect her ability to breastfeed. |
| References |
[1]. Gowarty JL, et al. Verapamil as a culprit of palbociclib toxicity. J Oncol Pharm Pract. 2019 Apr;25(3):743-746. [2]. Krikler DM. Verapamil in arrhythmia. Br J Clin Pharmacol. 1986;21 Suppl 2:183S-189S. [3]. Zhou P, et al. Anti-arrhythmic effect of Verapamil is accompanied by preservation of cx43 protein in rat heart. PLoS One. 2013 Aug 12;8(8):e71567. [4]. Rehnqvist N,et al. Effects of metoprolol vs verapamil in patients with stable angina pectoris. The Angina Prognosis Study in Stockholm (APSIS). Eur Heart J. 1996 Jan;17(1):76-81. [5]. Kubo Y, et al. Blood-to-Retina Transport of Fluorescence-Labeled Verapamil at the Blood-Retinal Barrier. Pharm Res. 2018 Mar 12;35(5):93 |
| Additional Infomation |
Dexverapamil is a 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile that has R configuration. It competitively inhibits the multidrug resistance efflux pump P-glycoprotein (MDR-1, EC 3.6.3.44), thereby potentially increasing the effectiveness of a wide range of antineoplastic drugs which are inactivated by MDR-1 mechanisms. Dexverapamil exhibits lower calcium antagonistic activity and toxicity than racemic verapamil. It has a role as an EC 3.6.3.44 (xenobiotic-transporting ATPase) inhibitor. It is a conjugate base of a dexverapamil(1+). It is an enantiomer of a (S)-verapamil. Dexverapamil is the R-enantiomer of the calcium channel blocker verapamil. Dexverapamil competitively inhibits the multidrug resistance efflux pump P-glycoprotein (MDR-1), thereby potentially increasing the effectiveness of a wide range of antineoplastic drugs which are inactivated by MDR-1 mechanisms. This agent exhibits decreased calcium antagonistic activity and toxicity compared to racemic verapamil. (NCI04) A calcium channel blocker that is a class IV anti-arrhythmia agent. See also: Verapamil (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 10 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5 mg/mL (10.18 mM) (saturation unknown) in 10% DMSO + 90% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.24 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.24 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 4: ≥ 2.08 mg/mL (4.24 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL corn oil and mix evenly. Solubility in Formulation 5: 25 mg/mL (50.91 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |