Physicochemical Properties
| Molecular Formula | C16H19N2BR |
| Exact Mass | 318.073 |
| Elemental Analysis | C, 60.20; H, 6.00; Br, 25.03; N, 8.78 |
| CAS # | 132-21-8 |
| Related CAS # | 132-21-8;2391-03-9; |
| PubChem CID | 16960 |
| Appearance | Light brown to brown oil |
| Density | 1.265g/cm3 |
| Boiling Point | 403ºC at 760mmHg |
| Melting Point | 113-115 |
| Flash Point | 197.5ºC |
| Vapour Pressure | 1.05E-06mmHg at 25°C |
| Index of Refraction | 1.577 |
| LogP | 3.927 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 19 |
| Complexity | 249 |
| Defined Atom Stereocenter Count | 1 |
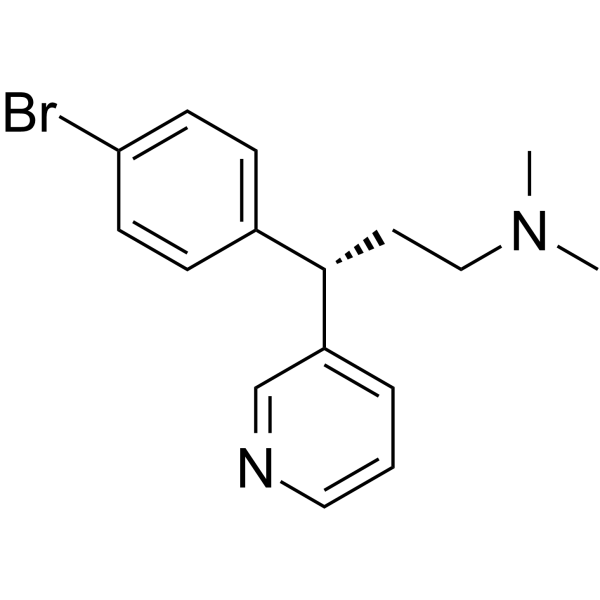
| SMILES | Brc1ccc([C@H](CCN(C)C)c2cccc[n]2)cc1 |
| InChi Key | ZDIGNSYAACHWNL-HNNXBMFYSA-N |
| InChi Code | InChI=1S/C16H19BrN2/c1-19(2)12-10-15(16-5-3-4-11-18-16)13-6-8-14(17)9-7-13/h3-9,11,15H,10,12H2,1-2H3/t15-/m0/s1 |
| Chemical Name | (3S)-3-(4-bromophenyl)-N,N-dimethyl-3-pyridin-2-ylpropan-1-amine |
| Synonyms | Dexbrompheniramine; D-Brompheniramine; 132-21-8; Dexbromfeniramina; Dexbrompheniraminum; (S)-brompheniramine; (+)-brompheniraminum; Brompheniramine d-; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | H1 Receptor |
| ln Vitro | Dexbrompheniramine is an alkylamine derivative with anticholinergic and sedative properties. Dexbrompheniramine is a histamine H1-receptor antagonist that competes with histamine for the H1-receptor sites on effector cells in the gastrointestinal tract, blood vessels and respiratory tract. The antagonistic action of this agent blocks the activities of endogenous histamine, which subsequently leads to temporary relief from the negative histamine-mediated symptoms of allergic reaction such as bronchoconstriction, vasodilation, increased capillary permeability and spasmodic contractions of gastrointestinal smooth muscle. |
| ln Vivo | The goal of these experiments was to determine which histamine receptors are involved in the relationship between drinking and feeding in ruminants. To this end, the effects of the histamine receptor antagonists dexbrompheniramine (H1 receptor antagonist), cimetidine (H2 receptor antagonist), and terfenadine (H1 receptor antagonist) on feeding and drinking patterns of pygmy goats were investigated. Two experiments using dexbrompheniramine [1 and 2 mg/kg of body weight (BW)0.75], two experiments using cimetidine (16 and 32 mg/kg of BW0.75), and two experiments using terfenadine (5 and 11.5 mg/kg of BW0.75) were performed to assess the type and location (periphery or central nervous system) of the histamine receptors involved in the mediation of prandial drinking by pygmy goats. The H1 receptor antagonists dexbrompheniramine (2 mg/kg of BW0.75) and terfenadine (11.5 mg/kg of BW0.75) significantly reduced water intake, but cumulative feed intake did not change. Consequently, the ratio of water intake to feed intake decreased. In contrast, the H2 receptor antagonist did not affect either water or feed intake. Dexbrompheniramine at 2 mg/kg of BW0.75 and terfenadine at 11.5 mg/kg of BW0.75 also decreased draft frequency and decreased the water intake associated with meals. Results showed that blockage of peripheral H1 histamine receptors attenuates the association between water and feed intake in pygmy goats. Therefore, the stimulating effect of feed intake on water intake appears to depend on activation of peripheral H1 histamine receptors[1]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Antihistamines are well absorbed from the gastrointestinal tract after oral administration. Metabolism / Metabolites Hepatic (cytochrome P-450 system), some renal. Biological Half-Life 25 hours |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Small, occasional doses of dexbrompheniramine would not be expected to cause any adverse effects in breastfed infants. Larger doses or more prolonged use may cause effects in the infant or decrease the milk supply, particularly in combination with a sympathomimetic such as pseudoephedrine or before lactation is well established. Single bedtime doses after the last feeding of the day may be adequate for many women and will minimize any effects of the drug. The nonsedating antihistamines are preferred alternatives. ◉ Effects in Breastfed Infants Irritability and disturbed sleep were reported in an 11-week-old breastfed infant whose mother was taking a product containing dexbrompheniramine and etafedrine (d-isoephedrine). These side effects were possibly caused by dexbrompheniramine in breastmilk, but could have been caused by the etafedrine or both drugs. In one telephone follow-up study, mothers reported irritability and colicky symptoms in 10% of infants exposed to various antihistamines and drowsiness was reported in 1.6% of infants. None of the reactions required medical attention and none of the infants were exposed to brompheniramine or dexbrompheniramine. ◉ Effects on Lactation and Breastmilk Antihistamines in relatively high doses given by injection can decrease basal serum prolactin in nonlactating women and in early postpartum women. However, suckling-induced prolactin secretion is not affected by antihistamine pretreatment of postpartum mothers. Whether lower oral doses of antihistamines have the same effect on serum prolactin or whether the effects on prolactin have any consequences on breastfeeding success have not been studied. The prolactin level in a mother with established lactation may not affect her ability to breastfeed. |
| References |
[1]. Effects of histamine H1 receptors on the feeding and drinking patterns in pygmy goats. J Dairy Sci. 1998 Sep;81(9):2369-75. |
| Additional Infomation |
Dexbrompheniramine is the (pharmacologically active) (S)-(+)-enantiomer of brompheniramine. A histamine H1 receptor antagonist, it is used (commonly as its maleate salt) for the symptomatic relief of allergic conditions, including rhinitis and conjunctivitis. It has a role as a H1-receptor antagonist and an anti-allergic agent. Dexbrompheniramine maleate is an antihistamine agent that is used for the treatment of allergic conditions, such as hay fever or urticaria. Dexbrompheniramine is an alkylamine derivative with anticholinergic and sedative properties. Dexbrompheniramine is a histamine H1-receptor antagonist that competes with histamine for the H1-receptor sites on effector cells in the gastrointestinal tract, blood vessels and respiratory tract. The antagonistic action of this agent blocks the activities of endogenous histamine, which subsequently leads to temporary relief from the negative histamine-mediated symptoms of allergic reaction such as bronchoconstriction, vasodilation, increased capillary permeability and spasmodic contractions of gastrointestinal smooth muscle. See also: Dexbrompheniramine Maleate (has salt form). Drug Indication For treatment and relief of symptoms of allergies, hay fever, and colds Mechanism of Action Dexbrompheniramine competitively binds to the histamine H1-receptor. It competes with histamine for the normal H1-receptor sites on effector cells of the gastrointestinal tract, blood vessels and respiratory tract. This blocks the action of endogenous histamine, which subsequently leads to temporary relief of the negative symptoms brought on by histamine. Pharmacodynamics In allergic reactions an allergen interacts with and cross-links surface IgE antibodies on mast cells and basophils. Once the mast cell-antibody-antigen complex is formed, a complex series of events occurs that eventually leads to cell-degranulation and the release of histamine (and other chemical mediators) from the mast cell or basophil. Once released, histamine can react with local or widespread tissues through histamine receptors. Histamine, acting on H1-receptors, produces pruritis, vasodilatation, hypotension, flushing, headache, tachycardia, and bronchoconstriction. Histamine also increases vascular permeability and potentiates pain. Dexbrompheniramine is a histamine H1 antagonist (or more correctly, an inverse histamine agonist) of the alkylamine class. It provides effective, temporary relief of sneezing, watery and itchy eyes, and runny nose due to hay fever and other upper respiratory allergies. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |