Physicochemical Properties
| Molecular Formula | C22H33NO2 |
| Molecular Weight | 343.5029 |
| Exact Mass | 343.251 |
| CAS # | 26166-37-0 |
| PubChem CID | 24721564 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 488.2±45.0 °C at 760 mmHg |
| Melting Point | 248-249℃ |
| Flash Point | 242.3±27.4 °C |
| Vapour Pressure | 0.0±2.8 mmHg at 25°C |
| Index of Refraction | 1.620 |
| LogP | 3.07 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 25 |
| Complexity | 662 |
| Defined Atom Stereocenter Count | 9 |
| SMILES | O([H])[C@@]1([H])[C@@]2([H])C(=C([H])[H])[C@]([H])([C@@]3(C([H])([H])C2([H])[H])[C@@]2([H])C([H])([H])[C@]4([H])C5(C([H])([H])[H])C([H])([H])C([H])([H])C([H])([H])C4([C@]2([H])N(C([H])([H])C([H])([H])[H])C5([H])[H])[C@@]31[H])O[H] |
| InChi Key | OVXLNQAYPUEDSI-ZBNNHZLTSA-N |
| InChi Code | InChI=1S/C22H33NO2/c1-4-23-11-20(3)7-5-8-22-15(20)10-14(18(22)23)21-9-6-13(12(2)19(21)25)16(24)17(21)22/h13-19,24-25H,2,4-11H2,1,3H3/t13-,14+,15-,16+,17-,18?,19-,20+,21+,22+/m1/s1 |
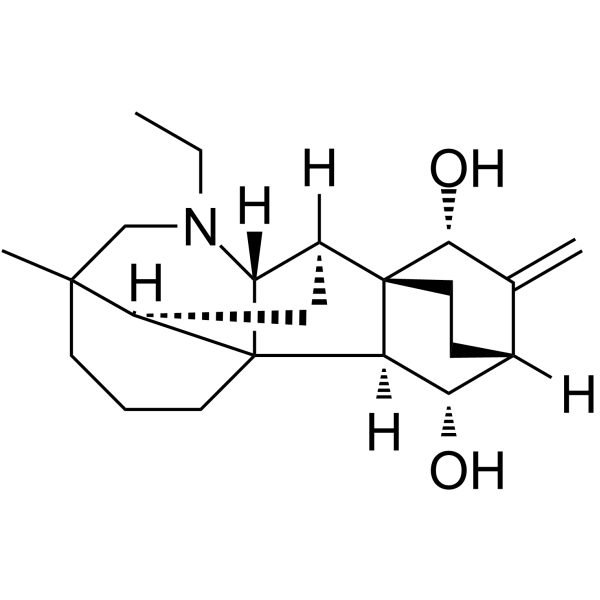
| Chemical Name | (1S,5R,9R,10S,11R,13R,14S,15S,16R)-7-ethyl-5-methyl-12-methylidene-7-azahexacyclo[7.6.2.210,13.01,8.05,16.010,15]nonadecane-11,14-diol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Denudatine altered the action potential of isolated rabbit ventricular fibers in vitro, specifically prolonging the action potential duration (APD) and reducing the maximum depolarization rate (Vmax) of the fibers [2] It significantly inhibited the arrhythmogenic action induced by aconitine in rabbit ventricular fibers, preventing the occurrence of abnormal electrical activity (such as premature depolarizations and sustained arrhythmic patterns) caused by aconitine [2] |
| Cell Assay |
Rabbit ventricular muscle was isolated under sterile and physiological conditions, and cut into thin ventricular fiber strips with appropriate dimensions to maintain structural and functional integrity [2] The ventricular fiber strips were mounted in an organ bath filled with oxygenated physiological solution, maintained at a constant temperature (37°C) to mimic in vivo physiological environment [2] Intracellular action potentials were recorded using glass microelectrodes connected to an electrophysiological recording system, and stable baseline recordings were obtained for at least 30 minutes before drug treatment [2] For action potential measurement: Denudatine was added to the organ bath at a predetermined concentration, and action potential parameters (APD, Vmax, resting membrane potential) were continuously recorded for 60 minutes after drug administration, with data analyzed by electrophysiological software [2] For anti-arrhythmic assay: Aconitine was first added to the organ bath to induce arrhythmic action potentials, and after the arrhythmia was stabilized, Denudatine was added at a specific concentration; changes in arrhythmic patterns and action potential parameters were recorded and compared with the aconitine-only group [2] |
| References |
[1]. Synthesis of Cardiopetaline via a Wagner-Meerwein Rearrangement without Preactivation of the Pivotal Hydroxy Group. Org Lett. 2017 Nov 3;19(21):5833-5835. [2]. Effects of denudatine on action potential of ventricular fibers and its inhibition on arrhythmogenic action of aconitine (author's transl). Zhongguo Yao Li Xue Bao. 1982 Mar;3(1):32-4. |
| Additional Infomation |
(1S,5R,9R,10S,11R,13R,14S,15S,16R)-7-ethyl-5-methyl-12-methylidene-7-azahexacyclo[7.6.2.210,13.01,8.05,16.010,15]nonadecane-11,14-diol has been reported in Aconitum carmichaelii and Aconitum kusnezoffii with data available. Denudatine is a natural alkaloid with cardiovascular-related biological activity [2] Its biological effects are mainly focused on regulating the electrical activity of ventricular muscle fibers and antagonizing aconitine-induced arrhythmias [2] Literature [1] focuses on the synthesis of Cardiopetaline and does not contain any relevant information about Denudatine [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9112 mL | 14.5560 mL | 29.1121 mL | |
| 5 mM | 0.5822 mL | 2.9112 mL | 5.8224 mL | |
| 10 mM | 0.2911 mL | 1.4556 mL | 2.9112 mL |