Physicochemical Properties
| Molecular Formula | C20H22O4 |
| Molecular Weight | 326.3863 |
| Exact Mass | 326.151 |
| CAS # | 2680-81-1 |
| PubChem CID | 5379033 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 452.0±45.0 °C at 760 mmHg |
| Flash Point | 227.2±28.7 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.596 |
| LogP | 4.19 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 24 |
| Complexity | 437 |
| Defined Atom Stereocenter Count | 0 |
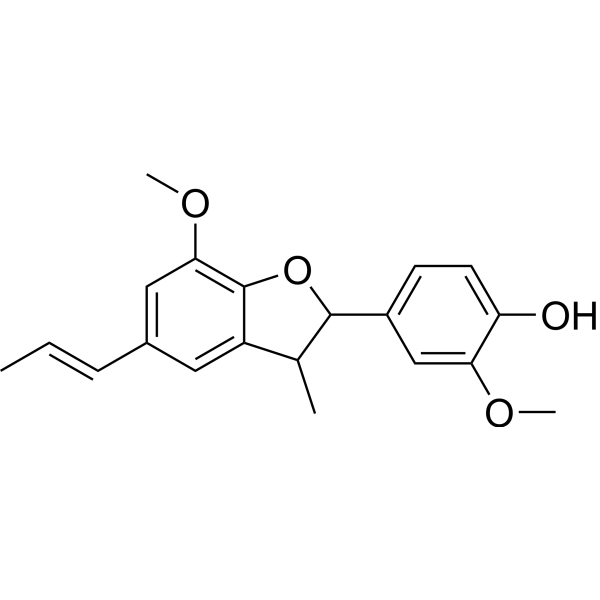
| SMILES | C/C=C/C1=CC2=C(C(=C1)OC)OC(C2C)C3=CC(=C(C=C3)O)OC |
| InChi Key | ITDOFWOJEDZPCF-AATRIKPKSA-N |
| InChi Code | InChI=1S/C20H22O4/c1-5-6-13-9-15-12(2)19(24-20(15)18(10-13)23-4)14-7-8-16(21)17(11-14)22-3/h5-12,19,21H,1-4H3/b6-5+ |
| Chemical Name | 2-methoxy-4-[7-methoxy-3-methyl-5-[(E)-prop-1-enyl]-2,3-dihydro-1-benzofuran-2-yl]phenol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Nuclear factor kappa B (NF-κB) signaling pathway [1] - Cyclooxygenase-2 (COX-2) [1] |
| ln Vitro |
- Dehydrodiisoeugenol exhibits potent anti-inflammatory activity in LPS-stimulated RAW264.7 macrophages. At concentrations of 10, 20, 40 μM, it dose-dependently inhibited NF-κB activation by 35±3%, 58±4%, and 72±5%, respectively [1] - It suppressed COX-2 expression: 40 μM reduced COX-2 mRNA levels by 68±4% and protein levels by 63±3% [1] - It inhibited pro-inflammatory cytokine secretion: 40 μM decreased TNF-α and IL-6 production by 59±4% and 55±3%, respectively [1] - It showed no significant cytotoxicity to RAW264.7 cells at concentrations up to 80 μM, with cell viability >90% [1] - In in vitro metabolic studies using liver microsomes, it was metabolized into 8 main metabolites via oxidation (hydroxylation, epoxidation), reduction, and conjugation reactions [2] |
| Enzyme Assay |
- COX-2 activity assay: LPS-stimulated RAW264.7 macrophages were treated with Dehydrodiisoeugenol (10–40 μM) for 24 hours. Cell supernatants were collected to measure PGE2 concentration by enzyme immunoassay, reflecting COX-2 activity [1] - NF-κB activity assay: RAW264.7 cells were pretreated with the compound (20 μM) for 1 hour, then stimulated with LPS for 30 minutes. Nuclear extracts were prepared, and NF-κB DNA-binding activity was detected by electrophoretic mobility shift assay (EMSA) [1] - Liver microsomal metabolism assay: Dehydrodiisoeugenol (10 μM) was incubated with liver microsomes (from human, rat, mouse) in the presence of cofactors (NADPH, UDP-glucuronic acid). Metabolites were separated by ultra-high performance liquid chromatography (UPLC) and identified by mass spectrometry (MS) [2] |
| Cell Assay |
- Anti-inflammatory cell assay: RAW264.7 macrophages were seeded in 96-well plates (5×10³ cells/well) and 6-well plates (2×10⁵ cells/well). After overnight incubation, cells were pretreated with Dehydrodiisoeugenol (10–40 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. Cytokine (TNF-α, IL-6) levels were measured by ELISA; COX-2 mRNA by RT-PCR; COX-2 and NF-κB pathway proteins by Western blot [1] - Cytotoxicity assay: RAW264.7 cells were treated with Dehydrodiisoeugenol (20–80 μM) for 48 hours, and cell viability was measured by MTT assay [1] - Metabolite identification assay: Cultured hepatocytes were treated with Dehydrodiisoeugenol (20 μM) for 48 hours. Culture media and cell lysates were collected, extracted, and analyzed by UPLC-MS/MS to identify intracellular and extracellular metabolites [2] |
| ADME/Pharmacokinetics |
- Absorption: Oral absorption in rats was moderate, with a bioavailability of ~38% [2] - Distribution: It distributed widely in rat tissues, with the highest concentrations in the liver, kidneys, and lungs [2] - Metabolism: Major metabolic pathways included hydroxylation at the aromatic ring, epoxidation of the alkene chain, reduction of the carbonyl group, and glucuronide/sulfate conjugation. Eight metabolites were identified, with M1 (monohydroxylated derivative) being the most abundant [2] - Excretion: Primarily excreted via feces (62%) and urine (28%) within 72 hours of oral administration in rats [2] - Plasma protein binding: Bound to rat plasma proteins at a rate of 89±2% [2] |
| Toxicity/Toxicokinetics |
- No significant in vitro cytotoxicity: Cell viability of RAW264.7 macrophages and hepatocytes remained above 90% at concentrations up to 80 μM [1][2] - No obvious acute toxicity was observed in preliminary studies, with no abnormal changes in liver/kidney function indices of rats after oral administration [2] |
| References |
[1]. Dehydrodiisoeugenol, an isoeugenol dimer, inhibits lipopolysaccharide-stimulated nuclear factor kappa B activation and cyclooxygenase-2 expression in macrophages. Arch Biochem Biophys. 2005 Feb 15;434(2):326-32. [2]. Metabolic profiling of dehydrodiisoeugenol using xenobiotic metabolomics. J Pharm Biomed Anal. 2017 Oct 25;145:725-733. |
| Additional Infomation |
Dehydrodiisoeugenol has been reported in Myristica fragrans with data available. - Dehydrodiisoeugenol is a natural dimer of isoeugenol, isolated from plant sources [1] - Its anti-inflammatory mechanism involves inhibiting the NF-κB signaling pathway to suppress COX-2 expression and pro-inflammatory cytokine secretion [1] - It is metabolized extensively in the liver via phase I (oxidation, reduction) and phase II (conjugation) reactions, with good tissue distribution and moderate oral bioavailability [2] - It shows potential as an anti-inflammatory agent with favorable metabolic and safety profiles [1][2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~306.38 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.37 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.37 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (6.37 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0638 mL | 15.3191 mL | 30.6382 mL | |
| 5 mM | 0.6128 mL | 3.0638 mL | 6.1276 mL | |
| 10 mM | 0.3064 mL | 1.5319 mL | 3.0638 mL |