Physicochemical Properties
| Molecular Formula | C20H28O4 |
| Molecular Weight | 332.43 |
| Exact Mass | 332.198 |
| CAS # | 134418-28-3 |
| PubChem CID | 6473762 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 512.8±50.0 °C at 760 mmHg |
| Flash Point | 178.3±23.6 °C |
| Vapour Pressure | 0.0±3.0 mmHg at 25°C |
| Index of Refraction | 1.584 |
| LogP | 2.97 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 24 |
| Complexity | 605 |
| Defined Atom Stereocenter Count | 5 |
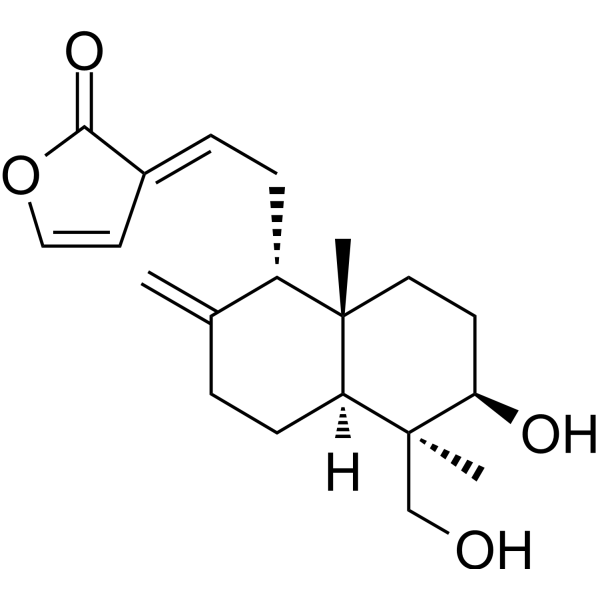
| SMILES | C[C@@]12CC[C@H]([C@@]([C@H]1CCC(=C)[C@H]2C/C=C/3\C=COC3=O)(C)CO)O |
| InChi Key | YIIRVUDGRKEWBV-FZOOCBFYSA-N |
| InChi Code | InChI=1S/C20H28O4/c1-13-4-7-16-19(2,10-8-17(22)20(16,3)12-21)15(13)6-5-14-9-11-24-18(14)23/h5,9,11,15-17,21-22H,1,4,6-8,10,12H2,2-3H3/b14-5+/t15-,16+,17-,19+,20+/m1/s1 |
| Chemical Name | (3E)-3-[2-[(1R,4aS,5R,6R,8aS)-6-hydroxy-5-(hydroxymethyl)-5,8a-dimethyl-2-methylidene-3,4,4a,6,7,8-hexahydro-1H-naphthalen-1-yl]ethylidene]furan-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
14-Deoxy-11,12-didehydroandrographolide demonstrated anti-HIV activity with an EC50 of 57 μg/mL in an assay using H9 cell lines. The compound also showed viricidal activity against herpes simplex virus 1 (HSV-1) without significant cytotoxicity. In RAW 264.7 macrophages transiently transfected with NF-κB reporter plasmids, the compound inhibited NF-κB-dependent transcriptional activation with an IC50 of 17.1 μg/mL. It also inhibited the production of pro-inflammatory cytokines TNF-α (IC50 20.64 μg/mL), IL-6 (IC50 23.6 μg/mL), MIP-2 (IC50 15.03 μg/mL), and nitric oxide (NO, IC50 11.26 μg/mL) in LPS/IFN-γ-stimulated macrophages.[2] |
| ln Vivo |
14-Deoxy-11,12-didehydroandrographolide, isolated from the methanol extract of A. paniculata, significantly reduced mean arterial pressure (MAP) and heart rate in anaesthetized rats after intravenous administration. It also reduced the beating rate of isolated right atria. The compound showed vasorelaxant effects in isolated rat thoracic aorta, potentially through the activation of nitric oxide synthase (NOS) and guanylyl cyclase, leading to NO release from endothelial cells. The hypotensive effects of the A. paniculata hot water extract are suggested to be partly mediated by this compound, acting on vascular smooth muscle.[2] |
| Cell Assay |
An NF-κB-dependent luciferase reporter assay was used to screen for anti-inflammatory activity. RAW 264.7 macrophages were transiently transfected with NF-κB reporter plasmids. Cells were pretreated with the test compound and then stimulated with LPS (100 ng/mL) and IFN-γ (1000 units/mL). Luciferase activity was measured to assess NF-κB trans-activation inhibition. For cytokine and NO measurement, the supernatants from the stimulated macrophages were collected. TNF-α, IL-6, and MIP-2 levels were determined using commercial ELISA kits. Nitric oxide production was measured using the Griess assay.[2] |
| Animal Protocol |
The compound's cardiovascular effects were studied in anaesthetized Sprague-Dawley rats. 14-Deoxy-11,12-didehydroandrographolide was administered intravenously, and mean arterial pressure (MAP) and heart rate were monitored. For studies on isolated tissues, the thoracic aorta and right atria were isolated from rats. The aortic rings were mounted in organ baths containing physiological salt solution, and tension was recorded to assess vasorelaxation. The beating rate of the isolated right atria was also measured.[2] |
| ADME/Pharmacokinetics |
This study investigated the pharmacokinetics of dehydroandrographolide succinate (DAS) injection after single intravenous doses of 80, 160, and 320 mg in healthy Chinese volunteers (n=9, crossover design). The drug was administered as an intravenous infusion, diluted in 250 mL of 5% glucose injection, and infused at a constant rate over 60 minutes. DAS exhibited nonlinear pharmacokinetics over the dose range studied. Mean maximum plasma concentration (Cmax) values were 4.82 mg/L, 12.85 mg/L, and 26.90 mg/L for the 80, 160, and 320 mg dose groups, respectively. Mean time to reach Cmax (Tmax) ranged from 0.94 to 1.0 hours. Mean area under the plasma concentration-time curve from 0 to 12 hours (AUC0-12) values were 6.18 mg·L⁻¹·h, 16.95 mg·L⁻¹·h, and 40.65 mg·L⁻¹·h for the 80, 160, and 320 mg doses, respectively. Mean elimination half-life (t₁/₂) was approximately 1.51 to 1.89 hours. Mean plasma clearance (CLz) decreased with increasing dose: 13.27 L/h (80 mg), 9.60 L/h (160 mg), and 8.07 L/h (320 mg). Urinary excretion of unchanged DAS within 24 hours accounted for 10.1% to 15.5% of the administered dose, with over 90% of the unchanged drug excreted within the first 4 hours. No significant pharmacokinetic differences were observed between male and female subjects.[1] |
| Toxicity/Toxicokinetics |
DAS injection was well tolerated in healthy volunteers at single intravenous doses up to 320 mg. One mild adverse event (stomach ache) was reported in one subject 24 hours after receiving the 320 mg dose; it resolved spontaneously within 2 hours. No serious or unexpected adverse events occurred. No clinically significant changes were observed in laboratory values, vital signs, or electrocardiogram parameters.[1] |
| References |
[1]. Pharmacokinetics and tolerance of dehydroandrographolide succinate injection after intravenous administration in healthy Chinese volunteers. Acta Pharmacol Sin. 2012 Oct;33(10):1332-6. [2]. Isolation and identification of bioactive compounds in Andrographis paniculata (Chuanxinlian). Chin Med. 2010 May 13;5:17. [3]. Inhibition of proprotein convertases-1, -7 and furin by diterpines of Andrographis paniculata and their succinoyl esters. Biochem J. 1999 Feb 15;338 ( Pt 1):107-13. [4]. Dehydroandrographolide succinate inhibits oxidative stress in mice with lipopolysaccharide-induced acute lung injury by inactivating iNOS. Nan Fang Yi Ke Da Xue Xue Bao. 2012 Sep;32(9):1238-41. [5]. Synthesis, structure-activity relationships and biological evaluation of dehydroandrographolide and andrographolide derivatives as novel anti-hepatitis B virus agents. Bioorg Med Chem Lett. 2014 May 15;24(10):2353-9. |
| Additional Infomation |
Dehydroandrographolide has been reported in Andrographis paniculata with data available. Dehydroandrographolide succinate (DAS) is a derivative extracted from the herbal medicine Andrographis paniculata. The DAS injection is used clinically in China for the treatment of viral pneumonia and upper respiratory tract infections due to its reported immunostimulatory, anti-infective, and anti-inflammatory effects. This study concluded that DAS exhibits nonlinear pharmacokinetics, with AUC and Cmax increasing slightly more than dose-proportionally, and clearance decreasing with increasing dose. The authors recommend multiple small doses in clinical regimens to maintain effective concentrations and avoid potential accumulation.[1] 14-Deoxy-11,12-didehydroandrographolide is a diterpenoid compound isolated from the medicinal plant Andrographis paniculata. It is an analogue of andrographolide, the major bioactive component of the plant. The compound is reported to possess multiple pharmacological properties, including immunostimulatory, anti-infective (against HIV and HSV-1), anti-inflammatory (via NF-κB pathway inhibition), and anti-atherosclerotic (vasorelaxant and hypotensive) effects. It is one of the active components contributing to the traditional use of A. paniculata in treating infections, inflammation, and possibly cardiovascular conditions.[2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~752.04 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.26 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.26 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (6.26 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0082 mL | 15.0408 mL | 30.0815 mL | |
| 5 mM | 0.6016 mL | 3.0082 mL | 6.0163 mL | |
| 10 mM | 0.3008 mL | 1.5041 mL | 3.0082 mL |