Physicochemical Properties
| Molecular Formula | C21H32O6 |
| Molecular Weight | 380.4752 |
| Exact Mass | 380.22 |
| CAS # | 3513-04-0 |
| PubChem CID | 21633061 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 0.83 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 27 |
| Complexity | 720 |
| Defined Atom Stereocenter Count | 8 |
| SMILES | O([H])[C@]12C([H])([H])C([H])([H])[C@@](C(C([H])([H])[H])=O)([C@@]1(C([H])([H])[H])[C@@]([H])(C([H])([H])[C@]1([H])[C@@]3(C([H])([H])[H])C([H])([H])C([H])([H])[C@@]([H])(C([H])([H])C3=C([H])C([H])([H])[C@@]21O[H])O[H])O[H])O[H] |
| InChi Key | ATMZVVNNICECKQ-MNSFQJRNSA-N |
| InChi Code | InChI=1S/C21H32O6/c1-12(22)19(25)8-9-21(27)18(19,3)16(24)11-15-17(2)6-5-14(23)10-13(17)4-7-20(15,21)26/h4,14-16,23-27H,5-11H2,1-3H3/t14-,15+,16+,17-,18+,19+,20-,21+/m0/s1 |
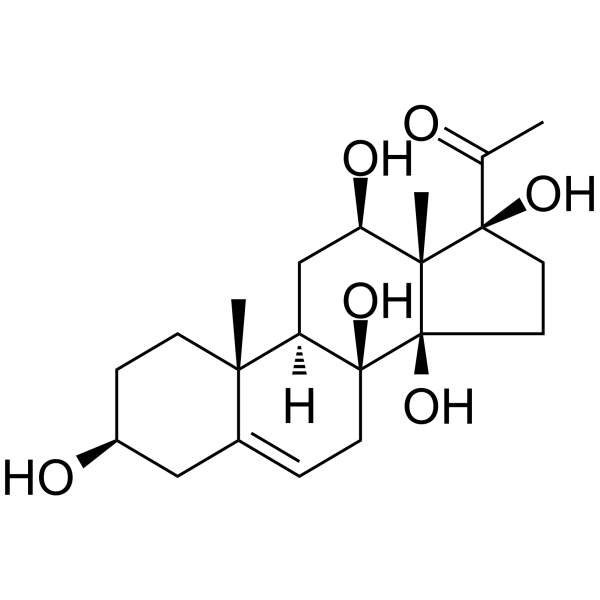
| Chemical Name | 1-[(3S,8S,9R,10R,12R,13S,14R,17S)-3,8,12,14,17-pentahydroxy-10,13-dimethyl-1,2,3,4,7,9,11,12,15,16-decahydrocyclopenta[a]phenanthren-17-yl]ethanone |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. Steroidal glycosides from the aerial part of Asclepias incarnata. Phytochemistry. 2000 Feb;53(4):485-98. |
| Additional Infomation |
Deacylmetaplexigenin has been reported in Asclepias incarnata, Cynanchum viminale, and Cynanchum rostellatum with data available. 1. Source and chemical classification: Deacylmetaplexigenin is a steroidal glycoside (specifically a cardenolide glycoside) isolated from the aerial parts (stems and leaves) of Asclepias incarnata (swamp milkweed), a plant of the Apocynaceae family [1] 2. Isolation and purification process: The aerial parts of Asclepias incarnata were dried and powdered, then extracted with methanol at room temperature for 3 consecutive days. The methanol extract was concentrated under reduced pressure to obtain a crude extract, which was suspended in water and partitioned sequentially with petroleum ether, chloroform, and n-butanol. The chloroform-soluble fraction (enriched in steroidal glycosides) was subjected to silica gel column chromatography, eluted with a gradient of chloroform-methanol (100:0 to 80:20, v/v). Fractions containing Deacylmetaplexigenin were identified by thin-layer chromatography (TLC) (developing solvent: chloroform-methanol-water = 8:2:0.2, v/v; visualization by spraying with 10% sulfuric acid in ethanol and heating at 110°C for 5 minutes). These fractions were further purified using Sephadex LH-20 column chromatography (eluent: methanol) and preparative high-performance liquid chromatography (HPLC) (column: C18 reverse-phase; mobile phase: methanol-water = 70:30, v/v; flow rate: 3 mL/min; detection wavelength: 210 nm) to obtain pure Deacylmetaplexigenin [1] 3. Structural characteristics: Deacylmetaplexigenin has a steroidal aglycone core (cardenolide skeleton) with a 5β,14β-dihydroxy-19-oxo configuration, and a sugar moiety (specifically a digitoxose unit) linked to the C3 hydroxyl group of the aglycone. Its structure was elucidated by spectroscopic methods including nuclear magnetic resonance (¹H-NMR, ¹³C-NMR), mass spectrometry (ESI-MS), and infrared spectroscopy (IR), confirming it as a deacylated derivative of metaplexigenin (lacking the acyl group at the C12 position of the aglycone) [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6283 mL | 13.1413 mL | 26.2826 mL | |
| 5 mM | 0.5257 mL | 2.6283 mL | 5.2565 mL | |
| 10 mM | 0.2628 mL | 1.3141 mL | 2.6283 mL |