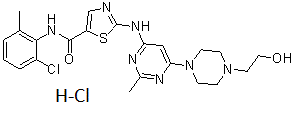
Dasatinib (formerly known as BMS-354825; sold under the brand name Sprycel), is a novel, potent and multi-targeted, orally bioavailable synthetic small molecule inhibitor that targets Abl, Src and c-Kit, with IC50 of<1 nM, 0.8 nM and 79 nM in cell-free assays, respectively. Chemotherapy patients treated with dasatinib may have acute lymphoblastic leukemia (ALL) or chronic myelogenous leukemia (CML). It is especially used to treat cases where the chromosome is positive for the Philadelphia gene (Ph+). The growth-promoting properties of these kinases are bound by dasatinib and inhibited. Dasatinib is able to reverse the resistance of chronic myeloid leukemia (CML) cells with point mutations in the BCR-ABL kinase domain to imatinib, presumably due to its less strict binding affinity for the BCR-ABL kinase.
Physicochemical Properties
| Molecular Formula | C22H26CLN7O2S.HCL |
| Molecular Weight | 524.47 |
| Exact Mass | 523.132 |
| CAS # | 854001-07-3 |
| Related CAS # | Dasatinib;302962-49-8;Dasatinib monohydrate;863127-77-9;Dasatinib-d8;1132093-70-9 |
| PubChem CID | 11466607 |
| Appearance | White to off-white solid powder |
| LogP | 4.264 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 34 |
| Complexity | 642 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | Cl.O=C(C1=CN=C(NC2C=C(N3CCN(CCO)CC3)N=C(C)N=2)S1)NC1C(C)=CC=CC=1Cl |
| InChi Key | MSCGWICDJYLQOJ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H26ClN7O2S.ClH/c1-14-4-3-5-16(23)20(14)28-21(32)17-13-24-22(33-17)27-18-12-19(26-15(2)25-18)30-8-6-29(7-9-30)10-11-31;/h3-5,12-13,31H,6-11H2,1-2H3,(H,28,32)(H,24,25,26,27);1H |
| Chemical Name | N-(2-chloro-6-methylphenyl)-2-[[6-[4-(2-hydroxyethyl)piperazin-1-yl]-2-methylpyrimidin-4-yl]amino]-1,3-thiazole-5-carboxamide;hydrochloride |
| Synonyms | BMS-354825 HCl; BMS354825; 854001-07-3; Dasatinib (hydrochloride); Dasatinib HCl; N-(2-chloro-6-methylphenyl)-2-[[6-[4-(2-hydroxyethyl)piperazin-1-yl]-2-methylpyrimidin-4-yl]amino]-1,3-thiazole-5-carboxamide;hydrochloride; N-(2-Chloro-6-methylphenyl)-2-((6-(4-(2-hydroxyethyl)piperazin-1-yl)-2-methylpyrimidin-4-yl)amino)thiazole-5-carboxamide hydrochloride; BMS 354825 hydrochloride; SCHEMBL1705152; BMS354825. Dasatinib HCl; Trade name: Sprycel |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Bcr-Abl (IC50 = 1 nM); Src (IC50 = 0.5 nM); lck (IC50 = 0.4 nM); Yes (IC50 = 0.5 nM); c-kit (IC50 = 5 nM); PDGFRβ (IC50 = 28 nM); p38 (IC50 = 100 nM); Her1 (IC50 = 180 nM); Her2 (IC50 = 710 nM); FGFR-1 (IC50 = 880 nM); MEK (IC50 = 1700 nM)
BCR-ABL fusion kinase (IC₅₀=0.6 nM in recombinant kinase assay); SRC kinase (IC₅₀=0.8 nM in recombinant kinase assay); LYN kinase (IC₅₀=0.9 nM in recombinant kinase assay); HCK kinase (IC₅₀=3.0 nM in recombinant kinase assay); C-KIT kinase (IC₅₀=74 nM in recombinant kinase assay) [1][2] |
| ln Vitro |
Dasatinib is more effective than imatinib at preventing the growth of Ba/F3 cells that express wild-type Bcr-Abl and Bcr-Abl mutants, with the exception of T315I. Compared to imatinib, dasatinib is approximately 325 times more potent by a factor of two. Over a limited range, dasatinib potently inhibits all mutants of Abl kinase, with the exception of T315I. Dasatinib inhibits substrate phosphorylation and autophosphorylation in a concentration-dependent manner by directly targeting the kinase domains of wild-type and mutant Abl kinases. When compared to imatinib, dasatinib exhibits 325-fold higher potency against cells expressing wild-type Bcr-Abl. In wells treated with dasatinib, the percentage of TgE bone marrow cell colonies drops from 100% to 4.12%. The percentage of colonies formed by WT and TgE bone marrow cells differs statistically significantly when dasatinib is present. Dasatinib can be used to target Lyn and/or c-Abl kinases, which will inhibit the promotion of B lymphocyte survival and proliferation that is brought about by the expression of LMP2A. Treatment with dasatinib inhibits Src signaling, reduces growth, and causes apoptosis and cell cycle arrest in a subset of thyroid cancer cells. After three days of treatment with increasing doses of dasatinib (0.019 μM to 1.25 μM), the C643, TPC1, BCPAP, and SW1736 cell lines grow less than 50% at low nanomolar concentrations; the K1 cell line grows more slowly and requires higher concentrations to inhibit growth. Treatment with 10 nM or 50 nM dasatinib causes a 7–18% reduction in the percentage of cells in the S phase and a 9–22% increase in G1 population cells among BCPAP, SW1736, and K1 cells. Dasatinib HCl is a potent, multi-targeted tyrosine kinase inhibitor with high activity against BCR-ABL, SRC family kinases (SRC, LYN, HCK), and C-KIT [1][2] - Inhibits recombinant BCR-ABL kinase activity with an IC₅₀ of 0.6 nM, including imatinib-resistant BCR-ABL mutants (e.g., Y253F, E255K, T315I mutants show reduced sensitivity with IC₅₀=31 nM, 16 nM, and 620 nM, respectively) [1] - Potently inhibits SRC family kinases: SRC (IC₅₀=0.8 nM), LYN (IC₅₀=0.9 nM), HCK (IC₅₀=3.0 nM) in recombinant kinase assays [1][2] - Exhibits potent antiproliferative activity against BCR-ABL-positive leukemia cells: GI₅₀=0.3 nM (K562 chronic myeloid leukemia [CML] cells), 0.5 nM (KU812 CML cells), 0.7 nM (BV173 acute lymphoblastic leukemia [ALL] cells); imatinib-resistant K562 cells (K562-R) show GI₅₀=1.2 nM [1] - Inhibits proliferation of SRC-dependent solid tumor cells: GI₅₀=25 nM (MDA-MB-231 breast cancer cells), 32 nM (HT-29 colorectal cancer cells), 45 nM (A549 non-small cell lung cancer cells) [1] - Induces apoptosis in BCR-ABL-positive cells: Treatment of K562 cells with 1–10 nM Dasatinib HCl for 48 hours increases apoptotic rate from 5% to 35–60% (Annexin V/PI staining); activates caspase-3 and PARP cleavage (western blot) [1] - Blocks downstream signaling pathways: In K562 cells, 0.1–10 nM Dasatinib HCl dose-dependently reduces phosphorylation of ABL (Tyr412), SRC (Tyr416), STAT5 (Tyr694), and AKT (Ser473); total protein levels remain unchanged [1][2] - Inhibits C-KIT-dependent cell proliferation: GI₅₀=74 nM for C-KIT-positive GIST-T1 cells; blocks KIT phosphorylation (Tyr719) at 100 nM [1] - Antiviral activity against HIV-1: Inhibits HIV-1 replication in CD4⁺ T cells with EC₅₀=0.8 μM; blocks HIV-1 entry by targeting SRC family kinases involved in viral fusion [3] |
| ln Vivo |
Dasatinib in LMP2A/MYC double transgenic mice reverses splenomegaly. Dasatinib specifically inhibits the growth of bone marrow B cells that express LMP2A and reduces the size of the spleen in TgE mice. When comparing the Tg6/λ-MYC mice treated with dasatinib to the control group, there is a significant decrease in spleen mass. Dasatinib prevents LMP2A/MYC double transgenic mice from developing lymphadenopathy. In Rag1KO mice engrafted with tumor cells from LMP2A/MYC double transgenic mice, dasatinib reverses splenomegaly. Treatment with dasatinib inhibits Lyn phosphorylation in tumors of B lymphocytes that express LMP2A.[3] In K562 (CML) xenograft model (BALB/c nude mice): Oral administration of Dasatinib HCl at 5 mg/kg, 10 mg/kg, and 20 mg/kg once daily for 21 days results in dose-dependent tumor growth inhibition (TGI) of 65%, 82%, and 95%, respectively; the 20 mg/kg group achieves complete tumor regression (CR) in 4/6 mice [1] - In imatinib-resistant K562-R xenograft model: Oral Dasatinib HCl 10 mg/kg once daily for 21 days induces TGI of 78%, significantly higher than imatinib (TGI=32%) [1] - In BV173 (ALL) xenograft model (SCID mice): 15 mg/kg oral Dasatinib HCl once daily for 14 days reduces tumor weight by 85% and prolongs median survival time from 28 days to 52 days (p<0.001) [2] - Pharmacodynamic analysis in xenografts: Treatment with 10 mg/kg Dasatinib HCl for 7 days reduces p-ABL (Tyr412) and p-SRC (Tyr416) protein levels by 70% and 65%, respectively, in K562 tumors [1] - In HIV-1-infected humanized mouse model: Oral Dasatinib HCl 5 mg/kg twice daily for 14 days reduces plasma HIV-1 viral load by 1.8 log₁₀ copies/mL and increases CD4⁺ T cell count by 30% compared to vehicle control [3] |
| Enzyme Assay |
Kinase assays are conducted with both wild-type and mutant glutathione S-transferase (GST)-Abl fusion proteins (c-Abl amino acids 220-498). Before usage, the GST-Abl fusion proteins are liberated from glutathione-Sepharose beads; 5 μM of ATP is present. The GST-Abl kinase domain fusion proteins are treated with LAR tyrosine phosphatase right before being used in kinase autophosphorylation and in vitro peptide substrate phosphorylation assays. Sodium vanadate (1 mM) is added to inactivate LAR phosphatase following a 1-hour incubation period at 30°C. Using phosphotyrosine-specific antibody 4G10 to confirm complete (>95%) dephosphorylation of tyrosine residues and c-Abl antibody CST 2862 to confirm equal loading of GST-Abl kinase, immunoblot analysis comparing untreated GST-Abl kinase to dephosphorylated GST-Abl kinase is routinely performed. The range of dasatinib concentrations for mutant T315I is increased to 1,000 nM. The in vitro peptide substrate phosphorylation assays employ the same concentrations of inhibitor. The three inhibitors are examined against GST-Src kinase and GST-Lyn kinase within these same concentration ranges. Recombinant kinase activity assay (HTRF-based): Recombinant human BCR-ABL, SRC, LYN, HCK, or C-KIT kinase (catalytic domain) is diluted in assay buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 1 mM EGTA, 0.01% BSA, 1 mM DTT). Serial 3-fold dilutions of Dasatinib HCl (0.001–1000 nM) are mixed with the kinase and pre-incubated for 30 minutes at room temperature. The reaction is initiated by adding ATP (final concentration 10 μM) and biotinylated peptide substrate, followed by incubation at 37°C for 60 minutes. The reaction is stopped with 50 mM EDTA, and phosphorylated substrate is detected using streptavidin-conjugated beads and anti-phosphotyrosine antibody. Fluorescence intensity is measured, and IC₅₀ values are calculated via nonlinear regression [1][2] - BCR-ABL mutant kinase assay: Recombinant imatinib-resistant BCR-ABL mutants (Y253F, E255K, T315I) are prepared and assayed using the same HTRF-based method as wild-type BCR-ABL; IC₅₀ values are determined to evaluate cross-reactivity [1] |
| Cell Assay |
Triple-seeded Ba/F3 cell lines are cultured for 72 hours at increasing dasatinib concentrations. A viability assay based on methanethiosulfonate is used to quantify proliferation. IC50 and IC90 values are given as the average of three separate, quadruple-experimented runs. The ranges of inhibitor concentrations (Dasatinib) are 0 nM to 32 nM. For mutant T315I, the dasatinib concentration range is expanded to 200 nM. Leukemia cell antiproliferation assay: BCR-ABL-positive cells (K562, KU812, BV173, K562-R) or solid tumor cells (MDA-MB-231, HT-29, A549) are seeded in 96-well plates (5×10³ cells/well) and incubated overnight. Serial 3-fold dilutions of Dasatinib HCl (0.001–1000 nM) are added, and cells are cultured for 72 hours. Cell viability is detected by MTS assay, and GI₅₀ values are calculated [1][2] - Apoptosis assay: K562 cells are seeded in 6-well plates (2×10⁵ cells/well) and treated with Dasatinib HCl (1–10 nM) for 48 hours. Cells are harvested, stained with Annexin V-FITC and PI, and analyzed by flow cytometry to quantify apoptotic rates [1] - Western blot for signaling pathways: K562 or BV173 cells are treated with Dasatinib HCl (0.1–10 nM) for 24 hours. Cells are lysed, proteins are separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-ABL (Tyr412), ABL, p-SRC (Tyr416), SRC, p-STAT5 (Tyr694), STAT5, p-AKT (Ser473), AKT, cleaved caspase-3, cleaved PARP, and β-actin [1][2] - HIV-1 replication assay: CD4⁺ T cells are infected with HIV-1 (NL4-3 strain) at MOI=0.01. After 2 hours of infection, cells are treated with serial 2-fold dilutions of Dasatinib HCl (0.1–10 μM) and cultured for 7 days. HIV-1 p24 antigen levels in culture supernatants are measured by ELISA, and EC₅₀ values are calculated [3] |
| Animal Protocol |
Dissolved in DMSO; 30 mg/kg; i.p. administration EμLMP2A (TgE and Tg6 strains), MYC (λ-MYC), and LMP2A/λ-MYC double transgenic mice (Tg6/λ-MYC) For in vivo experiments, dasatinib was dissolved in DMSO at 60 mg/ml and stored in aliquots at −20°C. On each treatment day, aliquots were thawed and diluted with 5.1% polyethylene glycol (PEG-400; EMD, Fisher) and 5.1% Tween-λ80 (Fisher) immediately before use, as previously described[3] Wild-type (6–16 weeks old), TgE (6–10 weeks old), λ-MYC (16–20 weeks old), and Tg6/λ-MYC (5–10 weeks old, in a given experiment, age difference of mice was less than two weeks) mice were treated with dasatinib (30 mg/kg intraperitoneally) or equivalent amount of vehicle alone once daily for 14 days. On day 15, the mice were sacrificed, and lymph node tumors and spleens were harvested, documented, processed, and analyzed with flow cytometry or western blotting.[3] K562 CML xenograft model: BALB/c nude mice (6–8 weeks old) are subcutaneously implanted with 5×10⁶ K562 cells (suspended in 50% Matrigel/PBS) into the right flank. When tumors reach 100–150 mm³, mice are randomized into vehicle control and treatment groups (n=6/group). Dasatinib HCl is formulated in 0.5% hydroxypropyl methylcellulose (HPMC) + 0.1% Tween 80 and administered orally at 5 mg/kg, 10 mg/kg, or 20 mg/kg once daily for 21 days. Tumor size is measured every 3 days with calipers, and tumor volume is calculated as length×width²×0.5 [1] - Imatinib-resistant K562-R xenograft model: BALB/c nude mice are implanted with 5×10⁶ K562-R cells. When tumors reach 100–150 mm³, mice are treated with Dasatinib HCl 10 mg/kg oral once daily or imatinib 100 mg/kg oral once daily for 21 days. Tumor growth is monitored, and TGI is calculated [1] - BV173 ALL xenograft model: SCID mice are subcutaneously implanted with 2×10⁶ BV173 cells. When tumors reach 100 mm³, mice are treated with Dasatinib HCl 15 mg/kg oral once daily for 14 days. Survival is monitored for 60 days; tumor weight is measured at study end [2] - HIV-1-infected humanized mouse model: NOD/SCID/IL2rg⁻/⁻ mice are transplanted with human CD34⁺ hematopoietic stem cells. Eight weeks post-transplantation, mice are infected with HIV-1 (NL4-3 strain) intravenously. Three days post-infection, mice are treated with Dasatinib HCl 5 mg/kg oral twice daily for 14 days. Plasma HIV-1 viral load and CD4⁺ T cell count are measured weekly [3] |
| ADME/Pharmacokinetics |
Oral bioavailability: 34% in rats (10 mg/kg oral), 58% in dogs (5 mg/kg oral) [1] - Plasma pharmacokinetics: In rats, oral administration of 10 mg/kg results in Cmax=1.2 μg/mL, AUC₀–24h=8.5 μg·h/mL, terminal half-life (t₁/₂)=5.3 hours; intravenous administration (2 mg/kg) shows Vd=1.8 L/kg and CL=0.2 L/h/kg [1] - In dogs, oral 5 mg/kg gives Cmax=2.1 μg/mL, AUC₀–24h=16.8 μg·h/mL, t₁/₂=7.6 hours [1] - Tissue distribution: In rats, Dasatinib HCl distributes widely to tissues, with highest concentrations in the liver, spleen, and tumor; tumor/plasma concentration ratio=2.8 at 4 hours post-dose [1] - Metabolism: Predominantly metabolized by cytochrome P450 3A4 (CYP3A4) in human liver microsomes; one major metabolite (M1) is identified, with BCR-ABL inhibitory potency 10-fold lower than the parent drug [1] - Excretion: In rats, 72-hour cumulative excretion is 68% (feces) and 12% (urine); 35% of the fecal excretion is parent drug [1] |
| Toxicity/Toxicokinetics |
Acute toxicity (mice): Single oral dose of 300 mg/kg Dasatinib HCl causes no mortality; mild weight loss (6%) and transient diarrhea are observed in 3/6 mice [1] - Subchronic toxicity (rats, 28 days): Oral doses up to 20 mg/kg/day show no significant changes in hematological/biochemical parameters (ALT, AST, BUN, creatinine); mild splenomegaly is observed at 20 mg/kg/day but is reversible [1] - Plasma protein binding rate: 96–98% in human, rat, and dog plasma (equilibrium dialysis, 0.1–10 μg/mL) [1] - Human adverse effects (clinical data from literature [2]): Most common treatment-related adverse events are hematological (thrombocytopenia: 38%, neutropenia: 32%, anemia: 25%) and gastrointestinal (diarrhea: 28%, nausea: 18%); rare adverse effects include fluid retention (10%) and skin rash (8%) [2] |
| References |
[1]. Cancer Res . 2005 Jun 1;65(11):4500-5. [2]. Blood . 2006 Jul 1;108(1):286-91. [3]. Antiviral Res . 2012 Jul;95(1):49-56. |
| Additional Infomation |
Imatinib, a Bcr-Abl tyrosine kinase inhibitor, is a highly effective therapy for patients with chronic myelogenous leukemia (CML). Despite durable responses in most chronic phase patients, relapses have been observed and are much more prevalent in patients with advanced disease. The most common mechanism of acquired imatinib resistance has been traced to Bcr-Abl kinase domain mutations with decreased imatinib sensitivity. Thus, alternate Bcr-Abl kinase inhibitors that have activity against imatinib-resistant mutants would be useful for patients who relapse on imatinib therapy. Two such Bcr-Abl inhibitors are currently being evaluated in clinical trials: the improved potency, selective Abl inhibitor AMN107 and the highly potent dual Src/Abl inhibitor BMS-354825. In the current article, we compared imatinib, AMN107, and BMS-354825 in cellular and biochemical assays against a panel of 16 kinase domain mutants representing >90% of clinical isolates. We report that AMN107 and BMS-354825 are 20-fold and 325-fold more potent than imatinib against cells expressing wild-type Bcr-Abl and that similar improvements are maintained for all imatinib-resistant mutants tested, with the exception of T315I. Thus, both inhibitors hold promise for treating imatinib-refractory CML.[1] \n\nMastocytosis is associated with an activating mutation in the KIT oncoprotein (KITD816V) that results in autophosphorylation of the KIT receptor in a ligand-independent manner. This mutation is inherently resistant to imatinib and, to date, there remains no effective curative therapy for systemic mastocytosis associated with KITD816V. Dasatinib (BMS-354825) is a novel orally bioavailable SRC/ABL inhibitor that has activity against multiple imatinib-resistant BCR-ABL isoforms in vitro that is presently showing considerable promise in early-phase clinical trials of chronic myeloid leukemia (CML). Pharmacokinetic analysis suggests that high nanomolar concentrations of dasatinib can be achieved safely in humans. In this study, we demonstrate significant inhibitory activity of dasatinib against both wild-type KIT and the KITD816V mutation in the nanomolar range in in vitro and cell-based kinase assays. Additionally, dasatinib leads to growth inhibition of a KITD816V-harboring human masto-cytosis cell line. Significantly, dasatinib selectively kills primary neoplastic bone marrow mast cells from patients with systemic mastocytosis while sparing other hematopoietic cells. Computer modeling suggests that the KITD816V mutation destabilizes the inactive conformation of the KIT activation loop to which imatinib binds, but it is not predicted to impair binding of KIT by dasatinib. Based upon our results, further evaluation of dasatinib for the treatment of systemic masto-cytosis in clinical trials is warranted. Moreover, dasatinib may be of clinical utility in other disease settings driven by activating KIT mutations.[2] \n\nEpstein-Barr virus (EBV) infection and latency has been associated with malignant diseases including nasopharyngeal carcinoma, Hodgkin lymphoma, Burkitt lymphoma, and immune deficiency associated lymphoproliferative diseases. EBV-encoded latent membrane protein 2A (LMP2A) recruits Lyn and Syk kinases via its SH2-domain binding motifs, and modifies their signaling pathways. LMP2A transgenic mice develop hyperproliferative bone marrow B cells and immature peripheral B cells through modulation of Lyn kinase signaling. LMP2A/λ-MYC double transgenic mice develop splenomegaly and cervical lymphomas starting at 8 weeks of age. We reasoned that targeting Lyn in LMP2A-expressing B cells with dasatinib would provide a therapeutic option for EBV-associated malignancies. Here, we show that dasatinib inhibits B cell colony formation by LMP2A transgenic bone marrow cells, and reverses splenomegaly and tumor growth in both a pre-tumor and a syngeneic tumor transfer model of EBV-associated Burkitt lymphoma. Our data support the idea that dasatinib may prove to be an effective therapeutic molecule for the treatment of EBV-associated malignancies.[3] Dasatinib HCl is a multi-targeted tyrosine kinase inhibitor developed for the treatment of imatinib-resistant chronic myeloid leukemia (CML) and acute lymphoblastic leukemia (ALL) [1][2] - Its mechanism of action involves ATP-competitive binding to the catalytic domain of BCR-ABL, SRC family kinases, and C-KIT, inhibiting their tyrosine kinase activity and blocking downstream signaling pathways (STAT5/AKT), leading to inhibition of cell proliferation and induction of apoptosis in cancer cells [1][2] - It exhibits activity against imatinib-resistant BCR-ABL mutants (except T315I, which shows reduced sensitivity) and SRC-dependent solid tumors, expanding its therapeutic potential [1] - Clinical efficacy: In a phase II trial, Dasatinib HCl (70 mg twice daily) achieves a major cytogenetic response rate of 45% in imatinib-resistant CML patients [2] - It also shows antiviral activity against HIV-1 by targeting SRC family kinases involved in viral entry, suggesting potential repurposing for HIV treatment [3] - Formulated as oral tablets, with recommended adult dose of 70 mg twice daily or 140 mg once daily for CML/ALL treatment [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3.33 mg/mL (6.35 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 33.3 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 3.33 mg/mL (6.35 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 33.3 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 3.33 mg/mL (6.35 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 33.3 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 4% DMSO+30% PEG 300+5% Tween 80+ddH2O: 5 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9067 mL | 9.5334 mL | 19.0669 mL | |
| 5 mM | 0.3813 mL | 1.9067 mL | 3.8134 mL | |
| 10 mM | 0.1907 mL | 0.9533 mL | 1.9067 mL |