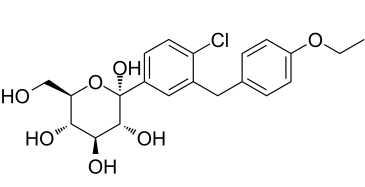
Dapagliflozin impurity is an isomer of Dapagliflozin which is a SGLT2 (sodium-glucose transporter 2) inhibitor approved in 2012 for the treatment of type 2 diabetes.
Physicochemical Properties
| Molecular Formula | C21H25CLO7 |
| Molecular Weight | 424.872006177902 |
| Exact Mass | 424.128 |
| CAS # | 960404-86-8 |
| PubChem CID | 59648701 |
| Appearance | Off-white to light yellow solid powder |
| LogP | 1.6 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 29 |
| Complexity | 515 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | O[C@@]1([C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1)C1C=CC(Cl)=C(CC2C=CC(OCC)=CC=2)C=1 |
| InChi Key | KYDGWGYAUCJZDV-ADAARDCZSA-N |
| InChi Code | InChI=1S/C21H25ClO7/c1-2-28-15-6-3-12(4-7-15)9-13-10-14(5-8-16(13)22)21(27)20(26)19(25)18(24)17(11-23)29-21/h3-8,10,17-20,23-27H,2,9,11H2,1H3/t17-,18-,19+,20-,21+/m1/s1 |
| Chemical Name | (2S,3R,4S,5S,6R)-2-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-6-(hydroxymethyl)oxane-2,3,4,5-tetrol |
| Synonyms | Dapagliflozin impurity; 960404-86-8; Dapagliflozin Impurity 10; Dapagliflozin hydroxy impurity; Hydroxy dapagliflozin; 2GLW9C3QQ3; (2S,3R,4S,5S,6R)-2-(4-Chloro-3-(4-ethoxybenzyl)phenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-2,3,4,5-tetraol; (2S,3R,4S,5S,6R)-2-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-6-(hydroxymethyl)oxane-2,3,4,5-tetrol; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Isomer and metabolite of Dapagliflozin |
| References |
[1]. Lack of pharmacokinetic interaction between dapagliflozin, a novel sodium-glucose transporter 2 inhibitor, and metformin, pioglitazone, glimepiride or sitagliptin in healthy subjects. Diabetes Obes Metab. 2011 Jan;13(1):47-54. |
| Additional Infomation | Aims: Dapagliflozin increases urinary glucose excretion by selectively inhibiting renal sodium-glucose transporter 2, an insulin-independent mechanism of action that may be complementary to that of other oral antidiabetes drugs. The current studies assessed the potential for pharmacokinetic (PK) interaction between dapagliflozin and pioglitazone, metformin, glimepiride or sitagliptin in healthy subjects following single-dose administration. Methods: In open-label, randomized, three-period, three-treatment crossover studies, 24 subjects received 50 mg dapagliflozin, 45 mg pioglitazone or the combination, while 18 subjects received 20 mg dapagliflozin, 1000 mg metformin or the combination. In an open-label, randomized, five-period, five-treatment, unbalanced crossover study, 18 subjects first received 20 mg dapagliflozin, 4 mg glimepiride or the combination, and afterward 100 mg sitagliptin or sitagliptin plus 20 mg dapagliflozin. Blood samples were taken over 72 h of each treatment period. Lack of PK interaction was defined as the ratio of geometric means and 90% confidence interval (CI) for combination:monotherapy being within the range of 0.80-1.25. Results: Co-administration of dapagliflozin with pioglitazone, metformin, glimepiride or sitagliptin had no effect on dapagliflozin maximum plasma concentration (C(max) ) or area under the plasma concentration-time curve (AUC). Similarly, dapagliflozin did not affect the C(max) or AUC for the co-administered drug, except for slight extensions of the 90% CI for the ratio of geometric means for glimepiride AUC (upper limit 1.29) and pioglitazone C(max) (lower limit 0.75). All monotherapies and combination therapies were well tolerated. Conclusion: Dapagliflozin can be co-administered with pioglitazone, metformin, glimepiride or sitagliptin without dose adjustment of either drug. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~58.84 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.43 mg/mL (3.37 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.3 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.43 mg/mL (3.37 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.3 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3537 mL | 11.7683 mL | 23.5366 mL | |
| 5 mM | 0.4707 mL | 2.3537 mL | 4.7073 mL | |
| 10 mM | 0.2354 mL | 1.1768 mL | 2.3537 mL |