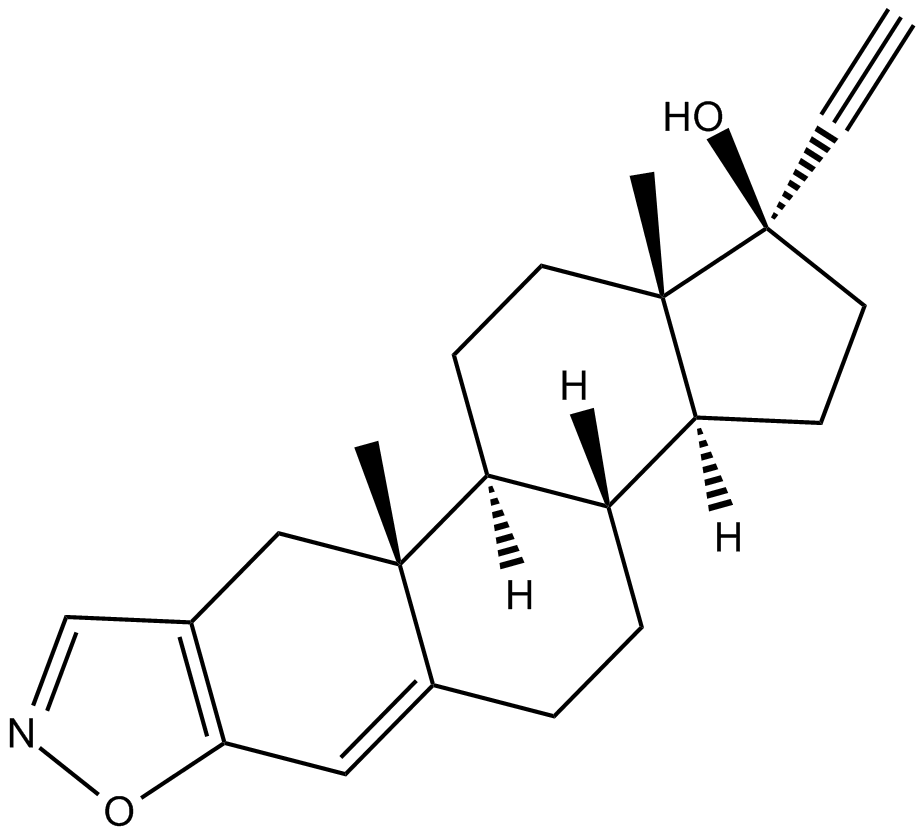
Danazol is a novel and potent analogue of the synthetic steroid ethisterone that functions as a weak androgenic androgen gonadotropin inhibitor.
Physicochemical Properties
| Molecular Formula | C22H27NO2 |
| Molecular Weight | 337.46 |
| Exact Mass | 337.204 |
| Elemental Analysis | C, 78.30; H, 8.06; N, 4.15; O, 9.48 |
| CAS # | 17230-88-5 |
| Related CAS # | 17230-88-5 |
| PubChem CID | 28417 |
| Appearance | White to light yellow crystalline powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 478.2±45.0 °C at 760 mmHg |
| Melting Point | 224.4-226.8ºC |
| Flash Point | 243.0±28.7 °C |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.604 |
| LogP | 4.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 25 |
| Complexity | 677 |
| Defined Atom Stereocenter Count | 6 |
| SMILES | C#C[C@]1(O)CC[C@@]2([H])[C@]3([H])CCC4=CC5=C(C=NO5)C[C@]4(C)[C@@]3([H])CC[C@@]21C |
| InChi Key | POZRVZJJTULAOH-LHZXLZLDSA-N |
| InChi Code | InChI=1S/C22H27NO2/c1-4-22(24)10-8-18-16-6-5-15-11-19-14(13-23-25-19)12-20(15,2)17(16)7-9-21(18,22)3/h1,11,13,16-18,24H,5-10,12H2,2-3H3/t16-,17+,18+,20+,21+,22+/m1/s1 |
| Chemical Name | (1S,2R,13R,14S,17R,18S)-17-ethynyl-2,18-dimethyl-7-oxa-6-azapentacyclo[11.7.0.02,10.04,8.014,18]icosa-4(8),5,9-trien-17-ol |
| Synonyms | Danazol; Danol; Danocrine; Chronogyn; Ladogal; Cyclomen |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Metabolism / Metabolites Hepatic, to principal metabolites, ethisterone and 17-hydroxymethylethisterone. Biological Half-Life Approximately 24 hours. |
| Additional Infomation |
Danazol can cause developmental toxicity according to state or federal government labeling requirements. Danazol is a 17beta-hydroxy steroid and a terminal acetylenic compound. It has a role as an anti-estrogen, an estrogen antagonist and a geroprotector. It derives from a hydride of a pregnane. A synthetic steroid with antigonadotropic and anti-estrogenic activities that acts as an anterior pituitary suppressant by inhibiting the pituitary output of gonadotropins. It possesses some androgenic properties. Danazol has been used in the treatment of endometriosis and some benign breast disorders. Danazol is an Androgen. The mechanism of action of danazol is as an Androgen Receptor Agonist. Danazol is a synthetic androgen derived from ethinyl testosterone. Danazol indirectly reduces estrogen production by decreasing pituitary secretion of follicle-stimulating hormone and luteinizing hormone, and binds to sex hormone receptors in target tissues, thereby exhibiting antiestrogenic, anabolic and weakly androgenic effects. (NCI04) A synthetic steroid with antigonadotropic and anti-estrogenic activities that acts as an anterior pituitary suppressant by inhibiting the pituitary output of gonadotropins. It possesses some androgenic properties. Danazol has been used in the treatment of endometriosis and some benign breast disorders. Drug Indication For the treatment of endometriosis and fibrocystic breast disease (in patients unresponsive to simple measures). Also used for the prophylactic treatment of all types of hereditary angioedema in males and females. Mechanism of Action As a gonadotropin inhibitor, danazol suppresses the pituitary-ovarian axis possibly by inhibiting the output of pituitary gonadotropins. Danazol also depresses the preovulatory surge in output of follicle-stimulating hormone (FSH) and luteinizing hormone (LH), thereby reducing ovarian estrogen production. Danazol may also directly inhibits ovarian steroidogenesis; bind to androgen, progesterone, and glucocorticoid receptors; bind to sex-hormone-binding globulin and corticosteroid-binding globulin; and increases the metabolic clearance rate of progesterone. Another mechanism of action by which danazol may use to facilitate regression of endometriosis is by decreasing IgG, IgM, and IgA concentrations, as well as phospholipid and IgG isotope autoantibodies. In the treatment of endometriosis, as a consequence of suppression of ovarian function, danazol causes both normal and ectopic endometrial tissues to become inactive and atrophic. This leads to anovulation and associated amenorrhea. In fibrocystic breast disease, the exact mechanism of action of danazol is unknown, but may be related to suppressed estrogenic stimulation as a result of decreased ovarian production of estrogen. A direct effect on steroid receptor sites in breast tissue is also possible. This leads to a disappearance of nodularity, relief of pain and tenderness, and possibly changes in the menstrual pattern. In terms of hereditary angioedema, danazol corrects the underlying biochemical deficiency by increasing serum concentrations of the deficient C1 esterase inhibitor, resulting in increased serum concentrations of the C4 component of the complement system. (Source: PharmGKB) Pharmacodynamics Danazol is a derivative of the synthetic steroid ethisterone, a modified testosterone. It was approved by the U.S. Food and Drug Administration (FDA) as the first drug to specifically treat endometriosis, but its role as a treatment for endometriosis has been largely replaced by the gonadotropin-releasing hormone (GnRH) agonists. Danazol has antigonadotropic and anti-estrogenic activities. Danazol acts as an anterior pituitary suppressant by inhibiting the pituitary output of gonadotropins. It possesses some androgenic properties. |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~25 mg/mL (~74.1 mM;) Ethanol: ~20 mg/mL (~59.3 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (7.41 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: ≥ 2 mg/mL (5.93 mM) (saturation unknown) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear EtOH stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 4: ≥ 2 mg/mL (5.93 mM) (saturation unknown) in 10% EtOH + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear EtOH stock solution to 900 μL of corn oil and mix well. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9633 mL | 14.8166 mL | 29.6331 mL | |
| 5 mM | 0.5927 mL | 2.9633 mL | 5.9266 mL | |
| 10 mM | 0.2963 mL | 1.4817 mL | 2.9633 mL |