Physicochemical Properties
| Molecular Formula | C26H39CLN4O8 |
| Molecular Weight | 571.068 |
| Exact Mass | 570.246 |
| Elemental Analysis | C, 54.68; H, 6.88; Cl, 6.21; N, 9.81; O, 22.41 |
| CAS # | 155204-81-2 |
| Related CAS # | 150612-55-8;150611-74-8 (HCl);155204-81-2 (HCl hydrate);201933-30-4 (racemic); 155204-80-1 (R-isomer); |
| PubChem CID | 122128 |
| Appearance | Typically exists as solid at room temperature |
| Boiling Point | 632.9ºC at 760mmHg |
| Flash Point | 336.6ºC |
| Vapour Pressure | 6.9E-17mmHg at 25°C |
| LogP | 5.303 |
| Hydrogen Bond Donor Count | 10 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 39 |
| Complexity | 720 |
| Defined Atom Stereocenter Count | 2 |
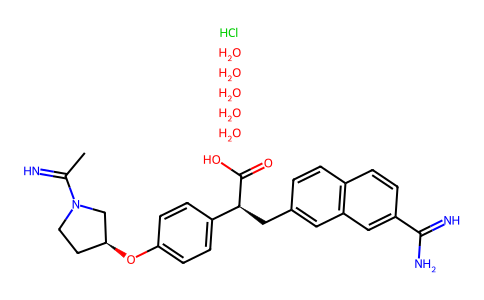
| SMILES | CC(=N)N1CC[C@@H](C1)OC2=CC=C(C=C2)[C@H](CC3=CC4=C(C=C3)C=CC(=C4)C(=N)N)C(=O)O.Cl.O.O.O.O.O |
| InChi Key | LJCBAPRMNYSDOP-LVCYMWGESA-N |
| InChi Code | InChI=1S/C26H28N4O3.ClH.5H2O/c1-16(27)30-11-10-23(15-30)33-22-8-6-19(7-9-22)24(26(31)32)13-17-2-3-18-4-5-20(25(28)29)14-21(18)12-17;;;;;;/h2-9,12,14,23-24,27H,10-11,13,15H2,1H3,(H3,28,29)(H,31,32);1H;5*1H2/t23-,24-;;;;;;/m0....../s1 |
| Chemical Name | (2S)-3-(7-carbamimidoylnaphthalen-2-yl)-2-[4-[(3S)-1-ethanimidoylpyrrolidin-3-yl]oxyphenyl]propanoic acid;pentahydrate;hydrochloride |
| Synonyms | DX9065A; DX 9065A; dx-9065a; 155204-81-2; DX 9065-a; DX-9065-A HCl hydrate; QXQVEPEVI2; 155204-81-2 (HCl, hydrate); (2S)-2-(4-(((3S)-1-acetimidoyl-3-pyrrolidinyl)oxy)phenyl)-3-(7-amidino-2-naphtyl)propanoic acid; (2S)-3-(7-Carbamimidoylnaphthalen-2-yl)-2-[4-[(3S)-1-ethanimidoylpyrrolidin-3-yl]oxyphenyl]propanoic acid;pentahydrate;hydrochloride; DX-9065A |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Factor Xa (FXa) (Ki = 41 nM) |
| ln Vitro |
The synthetic compound DX-9065a represents a low molecular weight, direct, competitive inhibitor of factor Xa (FXa) with a high affinity and selectivity for the enzyme. Under experimental conditions DX-9065a exerts strong anticoagulant actions in vitro and is antithrombotically effective in various thrombosis models. It inhibits proliferation of vascular smooth muscle cells in cell culture systems. As a small molecule inhibitor, DX-9065a inactivates both free and fibrin-bound FXa. By this mechanism it effectively affects the clot-associated procoagulant activity which might be responsible for the propagation of intravascular thrombi as well as for recurrent thrombosis and thrombotic reocclusion after lysis. [1] The synthetic compound DX-9065a represents a low molecular weight, direct inhibitorof FXa with a high affinity and selectivity for the enzyme. The first kinetic studies re-vealed that DX-9065a inhibited competitively human FXa with an inhibition constant Ki of 41 nM, whereas other serine proteases are not or only weakly affected (Ki values in ìM:thrombin > 2000, trypsin = 0.62, chymotrypsin > 2000, plasmin = 23, tPA = 21, plasmakallikrein = 2.3 and tissue kallikrein = 1000). In following studies slightly different Kivalues for the inhibition of human FXa were found ranging from 3.1 and 7.8 nM (14)to 24 nM. [1] A potent and selective FXa inhibitor such as DX-9065a does not influence preformedthrombin, but, based on the amplification mechanisms within the coagulation cascade andthe important role of the prothrombinase complex, it is expected to effectively inhibit thegeneration of thrombin which is probably the most important mechanism for the anti-thrombotic effectiveness of these agents. Thrombin-mediated feedback reactions such asthe activation of the cofactors V and VIII that amplify thrombin formation and the effectof thrombin on platelets and other cellular elements will also be altered. The inhibitoryeffect of DX-9065a on the generation of thrombin was demonstrated in human wholeblood where it caused a time- and concentration-dependent delay in or inhibition ofthrombin generation as well as in a biochemical thrombin generation assay whereDX-9065a delayed and reduced the generation of thrombin resulting in a decreasedamount of thrombin measured in plasma. [1] DX-9065a is the first member of a newly developed series of synthetic and selective inhibitors of factor Xa. DX 9065A inhibited in a dose-dependent manner human factor Xa with K iota value of 3.1 +/- 0.5 nM. Steady-state studies revealed that DX 9065A was a competitive inhibitor of factor Xa. DX 9065A inhibited thrombin generation occurring via both the extrinsic and intrinsic pathway in vitro. [2] |
| ln Vivo |
Studies on platelet-derived microparticles which are formed following platelet acti-vation showed that both the prothrombinase activity on the surface of these microparticlesand the resulting prothrombotic effect in vivo are inhibited by direct (DX-9065a) and in-direct (pentasaccharide) FXa inhibitors. The inhibitory effect of these anti-FXa agents isbased on the inhibition of FXa present at the surface of microparticles, on the delay of theburst of thrombin and on the inhibition of the coagulation cascade triggered at the surfaceof microparticles. An important aspect for the development of small molecule inhibitors of FXa is theirability to inactivate the clotting enzyme not only in plasma but also when it is bound tofibrin within a clot, an effect which is not seen with antithrombin and antithrombin-de-pendent inhibitors. Because of the presence of FXa and active prothrombinase complexesin intravascular and mural thrombi (10,11,39), the inactivation of FXa and the resulting in-hibition of thrombin formation may be an effective way to affect clot-associated proco-agulant activity which is considered to be responsible for the propagation of intravascularthrombi and for recurrent thrombosis after successful thrombolysis. As a small molecularweight inhibitor with a high affinity for the enzyme DX-9065a inhibits both free and pro-thrombinase-bound FXa and is able to penetrate into the clot and inhibit the clot-boundenzyme [1]. After i.v. injection to rabbits, DX-9065a displayed prolonged anti-factor Xa activity and inhibition of thrombin generation. Pretreatment of mice with DX-9065a dose-dependently improved the survival rate of mice injected with a lethal dose of tissue factor (ED50 = 1.1 +/- 0.2 mg/kg). After p.o. administration, DX 9065A caused a reduction in tissue factor-induced mortality of mice with ED50 value of 56 +/- 7 mg/kg. When given i.v. to rats, DX 9065A exhibited a dose-dependent antithrombotic effect against factor Xa + stasis-induced venous thrombosis (ED50 = 1.2 +/- 0.7 mg/kg i.v.), but also in an arteriovenous shunt thrombosis model (ED50 = 8.1 +/- 3.5 mg/kg i.v.) without affecting bleeding time significantly. Similar effects were obtained after s.c. or p.o. administration. In rabbits, after i.v., s.c. or p.o. administration, DX 9065A inhibited stasis-induced thrombosis after injection of tissue factor with ED50 values of 0.03 +/- 0.01, 0.3 +/- 0.07 and 50.5 +/- 19 mg/kg, respectively (n = 10). DX 9065A inhibited in a dose-dependent manner endotoxin-induced venous thrombosis in the rabbit (ED50 = 0.25 +/- 0.1 mg/kg i.v.) (n = 5) and reduced the decrease in platelet number and circulating fibrinogen levels in an experimental model of tissue factor-induced disseminated intravascular coagulation. Compared to standard heparin, DX-9065a exhibited a favorable antithrombotic/bleeding ratio, therefore showing that it might be considered as a promising compound in the treatment and prevention of various thrombotic diseases.[2] Anticoagulant Action [1] After parenteral administration there was a predictable quantitative relation betweenthe administered dose of DX-9065a and its anticoagulant and anti-FXa activity. A dose-dependent prolongation of clotting times in the APTT and PT assay was also seen afteroral administration of the inhibitor. The direct comparison between orally and intra-venously effective doses of DX-9065a revealed that for the anticoagulant and/or anti-thrombotic action at least 5 to 10 times higher doses are required by oral than by intrave-nous administration (see Table 1). It was also obvious that experimentalthrombus formation was inhibited with DX-9065a at doses which only modestly increasedthe clotting times APTT and PT measured ex vivo. It has to be mentioned that the anticoagulant and anti-Xa activities of DX-9065a arespecies-dependent. DX-9065a was nearly equally effective in prolonging the PT inhuman and common squirrel monkey plasma. In contrast, in rat plasma the inhibitor was40 times less potent than in human plasma. The anticoagulant response to DX-9065a incynomolgus monkey, dog, rabbit and mouse plasma ranged between those in human andrat plasma. Effect on Platelet Function [1] DX-9065a, at concentrations up to 100 ìM did not inhibit ADP-, collagen-, or throm-bin-induced platelet aggregation. The direct inactivation of factor Xa by DX-9065adid not prevent platelet activation in human whole blood which was induced by agonists such as arachidonic acid, TRAP or ã-thrombin. However, DX-9065a showed a time- andconcentration-dependent inhibitory effect on platelet activation mediated by either tissuefactor or factor Xa. It seems that the antiplatelet effect of DX-9065a is mainly due to theinhibition of generation of thrombin which is known to be the most important physiologicagonist for platelet activation and aggregation. Antithrombotic Action [1] It was clearly demonstrated by various experimental studies that DX-9065a is an effi-cacious antithrombotic drug by either intravenous, subcutaneous or oral administration.DX-9065a inhibits or prevents the development of several types of vascular thrombosissuch as venous and arterial thrombosis, occlusion of vascular shunts and disseminatedintravascular coagulation. Because of the great differences in the experimental models, thethrombogenic stimuli, the kind of drug administration as well as the species used it is dif-ficult to assess the usefulness of DX-9065a for a definite thrombotic disorder. Based onthe mechanism of action of DX-9065a it is expected that the inhibitor might have a greaterinhibitory effect on a thrombotic process where the underlying pathophysiological mech-anism primarily involves the activation of the coagulation cascade with the final con-version of fibrinogen to fibrin such as in venous thrombosis. A summary of experimentalthrombosis studies carried out with DX-9065a is given in Table 1. |
| ADME/Pharmacokinetics |
PHARMACOKINETICS [1] Animal studies Pharmacokinetics and pharmacodynamics of FXa inhibitors are closely related becausethe circulating blood is the primary site of action for a compound that inactivates clottingenzymes. For the long-term clinical use of anticoagulants/antithrombotics the oral admin-istration is the preferred route. DX-9065a is a low molecular weight FXa inhibitor thatwas designed to overcome the limitations of peptide compounds, especially the lack oforal bioavailability. The effectiveness of DX-9065a after oral administration was demon-strated in animal and human studies. DX-9065a caused strong anticoagulant and/or anti-thrombotic effects after either parenteral or oral administration (Table 1). However, com-parative studies revealed that for antithrombotic effects much (at least 10 times) higheroral than intravenous doses are required (Table 1). This is in accordance withpharmacokinetic studies in baboons where the oral bioavailability was estimated to be ap-proximately 5 to 12%. Studies on the time course of action of DX-9065a in ratsshowed that after i.v. injection of the inhibitor the anti-FXa activity was maximal immedi-ately after the injection and persisted for approximately 30 min. After oral administrationthe maximal anti-Xa activity was reached at 15 to 30 min after administration and per-sisted for about 90 min. The same time course was observed fot the antithrombotic action,i.e., the effect was transient after i.v. injection lasting for 10 to 20 min and was observedfor more than 3 h after oral administration. Other authors found that the peak activityof DX-9065a after oral administration to rats occurred at about 1 h after treatment and per-sisted for 4 h. After i.v. injection of DX-9065a to baboons plasma half-lives of6.3 min for the á-phase and 99 min for the â-phase were found. After oral administrationpeak plasma levels of DX-9065a were seen at 30 min and then gradually declined overabout 6 to 8 h. Human studies The pharmacokinetics of DX-9065a in humans differs from that in animals. This couldbe based on species differences which were clearly demonstrated in studies on the antico-agulant/antithrombotic effects of this inhibitor (see above). It is of special interest that theduration of action of DX-9065a in humans is much longer than in rats or baboons. Inregard to the possible clinical use of DX-9065a in the prevention or treatment of thrombo-embolic disorders, its longer duration of action is highly desirable. After a single i.v. bolusinjection (0.625–2.5 mg) of DX-9065a or a 1 h i.v. infusion (total dose 5 to 30 mg) to malehuman volunteers a biexponential or triexponential decrease of DX-9065a plasmaconcentrations was seen. At i.v. bolus injection of 2.5 mg the terminal phase t1/2 was10.7 h, whereas after i.v. infusion for 60 min the t1/2 was in the range of 22.8 to 26.1 h.The plasma protein binding rates ranged from 64.6 to 83%, and cumulative urinary ex-cretion ranged between 32.3 and 40.9%. Concentrations of DX-9065a in feces were belowthe quantification limit. At the dose range studied the pharmacokinetics of DX-9065a inhuman subjects after either i.v. bolus injection or continuous i.v. infusion for 1 h waslinear, independent of the dose administered and there were no statistically significant dif-ferences between the two treatment groups.After a single i.v. dose of 10 mg [14 C]DX-9065a, administered as 1 h infusion to ahealthy male Caucasian volunteer, plasma concentration of the drug decreased in a biexponential manner and was below the detection limit at 48 h after dosing. The half-lifefor the distribution phase was 6.93 h. The major route of excretion was via urine, ac-counting for more than 77.6% of the dose. A renal tubular secretion might contribute tothe urinary excretion of DX-9065a. Biotransformation of DX-9065a does not seem to playa significant role in the elimination of DX-9065a in humans.In the XaNADU-IB (Xa Neutralization for Atherosclerotic Disease Understanding)trial patients with stable coronary artery disease received DX-9065a at 1 mg i.v. bolus, fol-lowed by infusion at 4 different doses (see below) for 72 h. The half-life values ofDX-9065a were: t1/2á = 0.14 to 0.30 h, t1/2â = 1.93 to 3.20 h, t1/2ã = 76.57 to 98.86 h. Inter-individual differences increased at higher doses.In a double-blind, placebo-controlled study DX-9065a, 2.5, 5, or 10 mg, was adminis-tered s.c. to healthy male subjects. The peak plasma levels of the drug were reached at 1 hafter injection and declined to below the detection limit at 4 to 8 h after treatment. |
| Toxicity/Toxicokinetics |
Hemorrhagic Effects in Animal Studies [1] The main side effects of anticoagulant/antithrombotic drugs during their therapeuticuse are hemorrhagic complications. At antithrombotically effective doses the risk ofbleeding should be as low as possible. Data on anti-FXa agents regarding the impairmentof primary hemostasis pointed out that FXa inhibitors might cause less bleeding complica-tions than thrombin inhibitors. As a competitive inhibitor DX-9065a does not suppress theproduction of thrombin completely so that trace amounts of thrombin are still generated.Because of the much higher affinity of thrombin for platelets than for fibrinogen, thesesmall amounts of thrombin are sufficient to form the platelet-dependent hemostatic plugand, thus, to avoid bleeding. In various preclinical studies DX-9065a was shown to sup-press experimental thrombosis without causing hemorrhagic complications. In different models of bleeding time measurement, such as the tail transec-tion or the gastrointestinal hemorrhagic model in rats or the ear bleeding model in rabbits,DX-9065a did not prolong bleeding time at antithrombotically effective doses by eitherparenteral or oral administration. A direct comparison between the thrombin inhibitorargatroban, the low molecular weight heparin fragmin, unfractionated heparin andDX-9065a showed that argatroban as well as the antithrombin III-dependent anticoagu-lants prolonged bleeding time in rats at slightly higher doses than the antithromboticallyeffective doses, whereas DX-9065a at ten times higher doses did not affect the bleedingtime. Side Effects in Humans [1] In human volunteers DX-9065a, either by i.v. injection (0.625 to 2.5 mg) or by infusion(5 to 30 mg/60 min), produced no serious adverse effects either during or after the study. There were no clinically significant changes in serum chemistry, hematology, bleedingtime or urinary testsIn the XaNADU-IB trial DX-9065a was well tolerated in patients with stable coronaryartery disease. There were no discernable adverse effects on renal or hepatic function,platelet count, or hemoglobin. There were no major bleeding complications and only asmall, non-significant, dose-related increase in the incidence of minor bleeding in thehighest dose group as compared with placebo. |
| References |
[1]. Brigitte Kaiser. DX-9065a, a direct inhibitor of factor Xa. Cardiovasc Drug Rev. 2003 Summer;21(2):91-104. [2]. DX 9065A a novel, synthetic, selective and orally active inhibitor of factor Xa: in vitro and in vivo studies. J Pharmacol Exp Ther. 1996 Mar;276(3):1030-8. |
| Additional Infomation | In recent years great progress has been achieved in the development of small molecule,direct inhibitors of coagulation enzymes which interrupt the clotting process at differentsites of the coagulation cascade. Highly potent and selective inhibitors of activated clott-ing factors such as thrombin and FXa are expected to overcome the still existing limita-tions of the currently used antithrombotic regimens with heparin or vitamin K antagonists.The synthetic, low molecular weight FXa inhibitor DX-9065a which inactivates theenzyme without requiring any endogenous cofactor represents a new class of anticoagu-lant/antithrombotic drugs with a promising therapeutic potential. Under experimentalconditions DX-9065a exerts strong anticoagulant actions in vitro and in vivo, is antithrom-botically effective in various thrombosis models and also inhibits the proliferation of vas-cular smooth muscle cells in cell culture systems as well as in in vivo models. As a smallmolecule anti-FXa agent it inhibits both free and clot-bound FXa. This action, togetherwith the inhibition of thrombin generation, may be an effective way to control the clot-as-sociated procoagulant activity. However, despite of the demonstrated effectiveness of DX-9065aa general assessment of its therapeutic potential has to consider various addi-tional aspects.The development of an active-site directed inhibitor of FXa has to focus not only onoptimal binding to the target enzyme but also to its physico-chemical properties which de-termine the pharmacokinetic and pharmacodynamic behavior of the compound. One of themain goals in the development of a synthetic FXa inhibitor is to find a drug which can beused intravenously for acute thrombotic indications and then the therapy can be continuedwith an oral formulation of the same drug for chronic out-patient treatment. However,many of the small-molecule FXa inhibitors are highly basic moieties causing poor phar-macokinetic properties and especially a limited oral bioavailability. The intestinal ab-sorption of synthetic anti-factor Xa agents which is mainly accomplished by passive dif-fusion can be improved by more lipophilic molecules. A good example is the discovery ofDPC423, a highly potent, selective and orally bioavailable inhibitor of factor Xa. In aseries of 3-trifluoromethylpyrazole derivatives the replacement of the highly basic benz-amidine moiety by a less basic benzylamine moiety and the further optimization of themolecule resulted in a compound with a good pharmacokinetic and pharmacodynamicprofile. DPC423 showed an oral bioavailability of 57% and a plasma half-life of 7.5 h indogs and was antithrombotically effective in a rabbit arterio-venous shunt throm-bosis. DX-9065a, which has a basic naphthamidine moiety in its molecule, is oneof the few FXa inhibitors which, in experimental studies, are also effective by oral admin-istration. However, its oral bioavailability is relatively low and may not be adequate for itslong-term therapeutic use. Nevertheless, first clinical trials in healthy volunteers as well asin patients with cardiovascular diseases demonstrated at least predictable pharmacodyna-mic and pharmacokinetic profiles of DX-9065a by i.v. bolus injection and constant in-fusion. Intravenously administered DX-9065a is shown to be an efficacious anticoagu -lant/antithrombotic agent which has the advance of a greater safety than other drugs suchas heparin or warfarin. Furthermore, it was expected that specific FXa inhibitors mightalso be superior to direct thrombin inhibitors, such as hirudin, in providing selective inhi-bition of thrombus formation without compromising the hemostatic responses of platelets.As shown in preclinical and first clinical studies DX-9065a presents a promising alternative for the prevention or treatment of thrombotic events without hemorrhagic side ef-fects. Thus, it might be used as an adjunctive drug for thrombolytic therapy or togetherwith antiplatelet agents without increasing the bleeding risk.Another important point to be discussed with the use of a given FXa inhibitor iswhether and how its effect can be monitored in clinical practice, i.e., is there an easy andreproducible assay. A dose-dependent increase in the plasma concentrations of DX-9065a has been correlated with a prolongation of coagulation times measured in global clottingassays, such as PT and APTT. It is not yet clear whether at therapeutic doses and plasmalevels DX-9065a will prolong coagulation parameters, which marker is most sensitive forthis drug and whether it should be routinely used for monitoring of DX-9065a effec-tiveness. Furthermore, at present nothing is known about a possible way to neutralize theeffect of the compound in case of overdose or occurrence of undesired side effects.Results from experimental studies indicate a role of FXa in the complex pathogenesisof restenosis and atherosclerosis. Both thrombin and FXa appear to affect proliferation ofvascular smooth muscle cells in vivo. However, the precise role of the serine proteases andespecially the significance of their mitogenic activities for restenosis and atherosclerosis,as well as the practical significance of the inactivation of the enzymes by specific inhib-itors, have still to be clarified. For the thrombogenesis in the arterial system not onlyplatelet activation and aggregation but also thrombin generation is critical. Therefore, thedirect inhibition of FXa by DX-9065a appears to be a safe and effective new approach forpreventing the thrombotic complications of atherosclerotic disease.A general assessment of the therapeutic potential of DX-9065a has to consider variousadditional aspects. In addition to pharmacokinetic characteristics, such as oral bioavail-ability, biological half-life, metabolic transformations and excretory routes, interactionswith other drugs or endogenous factors have to be also investigated. The most promisingclinical indications for DX-9065a have to be defined just as the usefulness of combinationwith other drugs with different mechanisms or sites of action. Such a combination mightbe synergistic for the therapeutic effect, but could also enhance undesired side effects suchas bleeding complications.In conclusion, the FXa inhibitor DX-9065a represents a promising drug for the prophy-laxis and/or therapy of various thromboembolic disorders. Further experimental studiesand especially comprehensive clinical trials are likely to demonstrate the inhibitory profileof this compound, its effectiveness and especially its superiority over other drug regimensused for cardiovascular indications. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7511 mL | 8.7555 mL | 17.5110 mL | |
| 5 mM | 0.3502 mL | 1.7511 mL | 3.5022 mL | |
| 10 mM | 0.1751 mL | 0.8755 mL | 1.7511 mL |