Physicochemical Properties
| Molecular Formula | C42H84CLNO2 |
| Molecular Weight | 670.589 |
| Exact Mass | 669.619 |
| Elemental Analysis | C, 75.23; H, 12.63; Cl, 5.29; N, 2.09; O, 4.77 |
| CAS # | 104162-48-3 |
| Related CAS # | 104162-48-3 (Z-isomer chloride);122342-03-4 (Z-isomer cation);104872-42-6 (E-isomer chloride);717090-94-3 (E-isomer cation); |
| PubChem CID | 6438350 |
| Appearance | White to off-white solid powder |
| LogP | 10.173 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 37 |
| Heavy Atom Count | 46 |
| Complexity | 608 |
| Defined Atom Stereocenter Count | 0 |
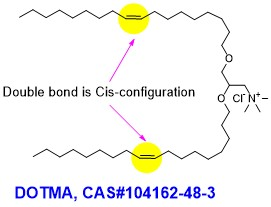
| SMILES | [Cl-].CCCCCCCC/C=C\CCCCCCCCOCC(C[N+](C)(C)C)OCCCCCCCC/C=C\CCCCCCCC |
| InChi Key | LDGWQMRUWMSZIU-LQDDAWAPSA-M |
| InChi Code | InChI=1S/C42H84NO2.ClH/c1-6-8-10-12-14-16-18-20-22-24-26-28-30-32-34-36-38-44-41-42(40-43(3,4)5)45-39-37-35-33-31-29-27-25-23-21-19-17-15-13-11-9-7-2;/h20-23,42H,6-19,24-41H2,1-5H3;1H/q+1;/p-1/b22-20-,23-21-; |
| Chemical Name | N,N,N-trimethyl-2,3-bis(((Z)-octadec-9-en-1-yl)oxy)propan-1-aminium chloride |
| Synonyms | DOTMA; 1,2-di-O-octadecenyl-3-trimethylammonium propane |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References | 1: Gendron F, Grasser M, Le Guennic B. Near-infrared circular dichroism of the ytterbium DOTMA complex: an ab initio investigation. Phys Chem Chem Phys. 2022 Mar 2;24(9):5404-5410. doi: 10.1039/d1cp01675j. PMID: 35170600. 2: Woods M, Payne KM, Valente EJ, Kucera BE, Young VG Jr. Crystal Structures of DOTMA Chelates from Ce3+ to Yb3+ : Evidence for a Continuum of Metal Ion Hydration States. Chemistry. 2019 Jul 25;25(42):9997-10005. doi: 10.1002/chem.201902068. Epub 2019 Jul 3. PMID: 31121070; PMCID: PMC6700027. 3: Mashal M, Attia N, Puras G, Martínez-Navarrete G, Fernández E, Pedraz JL. Retinal gene delivery enhancement by lycopene incorporation into cationic niosomes based on DOTMA and polysorbate 60. J Control Release. 2017 May 28;254:55-64. doi: 10.1016/j.jconrel.2017.03.386. Epub 2017 Mar 24. PMID: 28347807. 4: Kumas C, Fernando WS, Zhao P, Regueiro-Figueroa M, Kiefer GE, Martins AF, Platas-Iglesias C, Sherry AD. Unexpected Changes in the Population of Coordination Isomers for the Lanthanide Ion Complexes of DOTMA-Tetraglycinate. Inorg Chem. 2016 Sep 19;55(18):9297-305. doi: 10.1021/acs.inorgchem.6b01390. Epub 2016 Sep 7. PMID: 27603690; PMCID: PMC5221692. 5: Webber BC, Woods M. Structural analysis of isomeric europium(III) chelates of NB-DOTMA. Inorg Chem. 2012 Aug 6;51(15):8576-82. doi: 10.1021/ic3011597. Epub 2012 Jul 18. PMID: 22809081. 6: Aime S, Botta M, Garda Z, Kucera BE, Tircso G, Young VG, Woods M. Properties, solution state behavior, and crystal structures of chelates of DOTMA. Inorg Chem. 2011 Sep 5;50(17):7955-65. doi: 10.1021/ic2012827. Epub 2011 Aug 5. PMID: 21819052; PMCID: PMC3204394. 7: Matsumoto M, Kishikawa R, Kurosaki T, Nakagawa H, Ichikawa N, Hamamoto T, To H, Kitahara T, Sasaki H. Hybrid vector including polyethylenimine and cationic lipid, DOTMA, for gene delivery. Int J Pharm. 2008 Nov 3;363(1-2):58-65. doi: 10.1016/j.ijpharm.2008.07.010. Epub 2008 Jul 18. PMID: 18687391. 8: Gaucheron J, Santaella C, Vierling P. Transfection with fluorinated lipoplexes based on fluorinated analogues of DOTMA, DMRIE and DPPES. Biochim Biophys Acta. 2002 Aug 31;1564(2):349-58. doi: 10.1016/s0005-2736(02)00469-8. PMID: 12175917. 9: Ren T, Song YK, Zhang G, Liu D. Structural basis of DOTMA for its high intravenous transfection activity in mouse. Gene Ther. 2000 May;7(9):764-8. doi: 10.1038/sj.gt.3301153. PMID: 10822303. 10: Wollenberg B, Kastenbauer, Mundl H, Schaumberg J, Mayer A, Andratschke M, Lang S, Pauli C, Zeidler R, Ihrler S, Löhrs, Naujoks K, Rollston R. Gene therapy --phase I trial for primary untreated head and neck squamous cell cancer (HNSCC) UICC stage II-IV with a single intratumoral injection of hIL-2 plasmids formulated in DOTMA/Chol. Hum Gene Ther. 1999 Jan 1;10(1):141-7. doi: 10.1089/10430349950019273. PMID: 10022539. 11: Konopka K, Davis BR, Düzgüneş N. HIV-1 infection of a non-CD4-expressing variant of HUT-78 cells: lack of inhibition by Leu3A antibodies and enhancement by cationic DOTMA liposomes. Adv Exp Med Biol. 1991;300:97-110. doi: 10.1007/978-1-4684-5976-0_7. PMID: 1781348. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4912 mL | 7.4561 mL | 14.9122 mL | |
| 5 mM | 0.2982 mL | 1.4912 mL | 2.9824 mL | |
| 10 mM | 0.1491 mL | 0.7456 mL | 1.4912 mL |