Physicochemical Properties
| Molecular Formula | C8H4N4O6 |
| Molecular Weight | 252.1406 |
| Exact Mass | 252.013 |
| Elemental Analysis | C, 38.11; H, 1.60; N, 22.22; O, 38.07 |
| CAS # | 2379-57-9 |
| Related CAS # | DNQX disodium salt;1312992-24-7 |
| PubChem CID | 3899541 |
| Appearance | White to off-white solid powder |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 670.7ºC at 760 mmHg |
| Melting Point | >300°C |
| Flash Point | 359.4ºC |
| Vapour Pressure | 1.33E-18mmHg at 25°C |
| Index of Refraction | 1.665 |
| LogP | 1.13 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 18 |
| Complexity | 386 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | RWVIMCIPOAXUDG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H4N4O6/c13-7-8(14)10-4-2-6(12(17)18)5(11(15)16)1-3(4)9-7/h1-2H,(H,9,13)(H,10,14) |
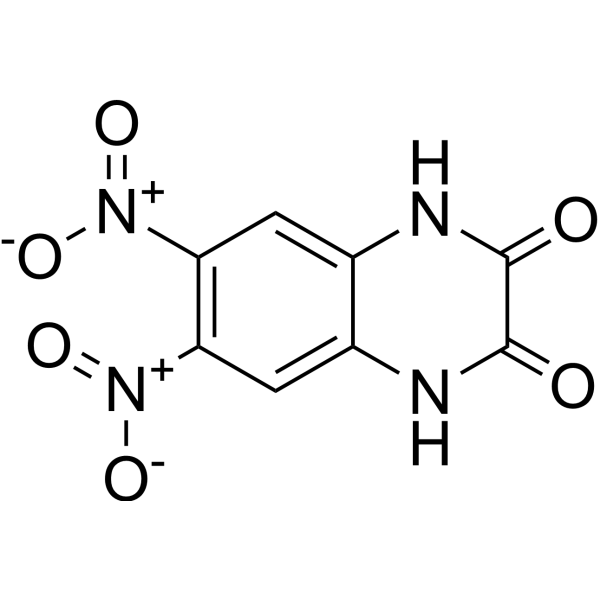
| Chemical Name | 6,7-dinitro-1,4-dihydroquinoxaline-2,3-dione |
| Synonyms | dnqx; 2379-57-9; 6,7-dinitro-1,4-dihydroquinoxaline-2,3-dione; FG-9041; 6,7-Dinitroquinoxaline-2,3(1H,4H)-dione; 6,7-Dinitro-1,4-dihydro-quinoxaline-2,3-dione; 6,7-Dinitroquinoxaline-2,3-dione (DNQX); 2,3-Quinoxalinedione, 1,4-dihydro-6,7-dinitro-; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | non-NMDA glutamate receptor; Kainate Receptor |
| ln Vitro |
Thalamic reticular nucleus (TRN) neurons are selectively depolarized by DNQX [2]. The quinoxaline derivative 6,7-dinitroquinoxaline-2,3-dione (DNQX) selectively depolarizes thalamic reticular nucleus (TRN) neurons. A: DNQX (20 μM) produces a reversible membrane depolarization in a TRN neuron. In contrast, DNQX (20 μM) application does not alter the membrane potential in a ventrobasal (VB) neuron. The transient downward deflections, in this and following figures, are membrane responses to short hyperpolarizing current steps. Graph depicts population data illustrating differential effect of DNQX. B: in a voltage-clamped TRN neuron (Vhold = −70mV), DNQX (20 μM) produces a reversible inward current. DNQX does not alter the holding current of a VB neuron. C: in a different TRN neuron, a lower DNQX concentration (4 μM) produces no change in membrane potential, whereas at a higher concentration, DNQX (100 μM) produces a robust depolarization. Plot illustrates concentration-dependent effects of DNQX on membrane potential of TRN neurons. D: the DNQX-mediated depolarizations persist in TTX (0.5 μM) or a low Ca2+ (0.2 mM)/high Mg2+ (6 mM)-containing physiological solution. [2] |
| ln Vivo | DNQX is a particular AMPA receptor antagonist that considerably lowers the amount of phencyclidine (PCP) that is induced in the posterior cingulate and retrosplenial cortex by 5 mg/kg or 10 mg/kg intraperitoneally or intracerebroventricularly (5 μl, 0.5 mg/ml) (40 mg/kg) and ketamine (80, 100, 120 mg/kg) [3]. |
| Enzyme Assay | Concentrated stock solutions of DNQX, CNQX, 2,3-dioxo-6-nitro-1,2,3,4-tetrahydrobenzo[f]quinoxaline-7-sulfonamide (NBQX), 4-(8-methyl-9H-1,3-dioxolo[4,5-h][2,3]benzodiazepin-5-y l)-benzenamine hydrochloride (GYKI52466), and trichloromethiazide (TCM) were dissolved in DMSO. (2S,4R)-4-methylglutamic acid (SYM2081) was prepared in distilled water, and kynurenic acid was prepared in physiological saline at final concentration. Stock solutions were diluted in physiological saline to a final concentration just prior to application. DNQX, CNQX, and NBQX were applied by injecting a bolus into the input line of the chamber over 60 s using a motorized syringe pump. Based on the rate of syringe pump and chamber perfusion, the final bath concentration of drugs was estimated to one-eighth of the concentration introduced in the flow line (Cox et al. 1995). In a subpopulation of neurons, CNQX was bath applied for 5 min. Control injections of physiological saline or vehicle (DMSO) did not alter membrane potential/input resistance during voltage recordings. GYKI52466, SYM2081, kynurenic acid, and TCM were bath-applied for ≥7 min before subsequent application of other agents. [2] |
| Cell Assay | Quinoxaline derivatives [e.g., 6,7-dinitroquinoxaline-2,3-dione (DNQX), 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX)] have routinely been used as non-NMDA receptor antagonists over the last two decades. In this study, we examined whether quinoxaline derivatives alter the intrinsic properties of thalamic neurons in light of recent findings indicating that these compounds can alter neuronal excitability in hippocampal and cerebellar neurons via transmembrane AMPA receptor (AMPAR) regulatory proteins (TARPs). Whole cell recordings were obtained from TRN and ventrobasal (VB) thalamic relay neurons in vitro. DNQX and CNQX produced a consistent depolarization in all TRN neurons tested. The depolarization persisted in tetrodotoxin and low Ca²+/high Mg²+ conditions, suggesting a postsynaptic site of action. In contrast, DNQX and CNQX produced little or no change in VB thalamocortical relay neurons. The nonspecific ionotropic glutamate receptor antagonist, kynurenic acid, and the selective AMPAR antagonist, 4-(8-methyl-9H-1,3-dioxolo[4,5-h][2,3]benzodiazepin-5-yl)-benzenamine hydrochloride, blocked the DNQX-mediated depolarizations. Our results indicate that the DNQX- and CNQX-mediated depolarizations are mediated by AMPAR but not kainate receptors in TRN neurons. The AMPAR-positive allosteric modulator, trichloromethiazide, potentiated the DNQX-mediated depolarization in TRN neurons but did not unmask any excitatory actions of DNQX/CNQX in relay neurons. This selective action may not only reveal a differential TARP distribution among thalamic neurons but also may provide insight into distinct characteristics of AMPA receptors of thalamic neurons that could be exploited by future pharmacological development. Furthermore, these data suggest that quinoxaline derivatives could modulate synaptic transmission and alter neuronal excitability[2]. |
| Animal Protocol | The present study shows that DNQX, a specific AMPA receptor antagonist, given as either a 5 mg/kg or 10 mg/kg intraperitoneal dose or into the lateral cerebral ventricle (5 microliters of 0.5 mg/ml) significantly diminished PCP (40 mg/kg) and ketamine (80, 100, 120 mg/kg) hsp70 induction in the posterior cingulate and retrosplenial cortex. The most dramatic decrease of hsp70 induction was seen with the intraventricular dose of DNQX. Present findings show that the AMPA receptor has a role in PCP/ketamine induction of hsp70 in the cortex. DNQX inhibition of PCP/ketamine hsp70 induction was likely related to AMPA receptor antagonism which prevented excess calcium influx via voltage-gated calcium channels.[3] |
| References |
[1]. Quinoxalinediones: potent competitive non-NMDA glutamate receptor antagonists. Science. 1988;241(4866):701-703. [2]. Selective excitatory actions of DNQX and CNQX in rat thalamic neurons. J Neurophysiol. 2010;103(4):1728-1734. [3]. DNQX inhibits phencyclidine (PCP) and ketamine induction of the hsp70 heat shock gene in the rat cingulate and retrosplenial cortex. Brain Res. 1995;687(1-2):114-124. |
| Additional Infomation |
6,7-dinitro-1,4-dihydroquinoxaline-2,3-dione is a quinoxaline derivative. In the hippocampus and cerebellum, CNQX has been found to increase inhibitory activity in excitatory neurons by an increase in excitatory response in GABAergic interneurons (Maccaferri and Dingledine 2002; McBain et al. 1992; Menuz et al. 2007). Our findings are consistent in that we find CNQX and DNQX can produce membrane depolarization of GABA-containing TRN neurons; this would in turn alter inhibitory activity onto thalamic relay neurons. Furthermore the quinoxalinedione-induced depolarization is blocked by AMPAR antagonists, leading to the conclusion that DNQX and CNQX are acting as partial agonists on AMPARs. We also found that the AMPAR allosteric modulator trichloromethiazide potentiated the excitatory action of DNQX on TRN neurons. In CA1 pyramidal neurons, the allosteric modulator unmasked an excitatory action of CNQX that was not present in the absence of the modulator (Menuz et al. 2007); however, our results clearly indicate that the excitatory actions of quinoxaline derivatives cannot be generalized to all excitatory neurons because in thalamocortical relay neurons, the allosteric modulator did not unmask an excitatory response to the quinoxaline derivatives.[2] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 35 mg/mL (~138.81 mM) H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (8.25 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (8.25 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.9661 mL | 19.8303 mL | 39.6605 mL | |
| 5 mM | 0.7932 mL | 3.9661 mL | 7.9321 mL | |
| 10 mM | 0.3966 mL | 1.9830 mL | 3.9661 mL |