Physicochemical Properties
| Molecular Formula | C4H9NO2S |
| Molecular Weight | 135.18476 |
| Exact Mass | 135.035 |
| CAS # | 454-29-5 |
| PubChem CID | 778 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 299.7±35.0 °C at 760 mmHg |
| Melting Point | 232-233 °C(lit.) |
| Flash Point | 135.0±25.9 °C |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.538 |
| LogP | 0.22 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 8 |
| Complexity | 86.1 |
| Defined Atom Stereocenter Count | 0 |
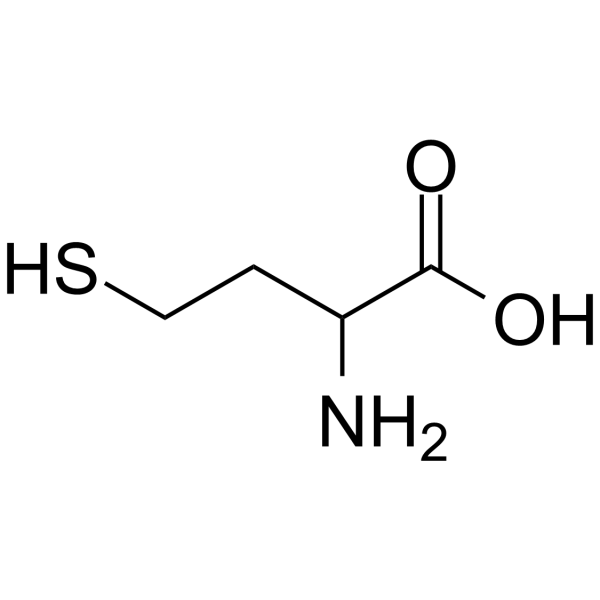
| SMILES | O=C(O)C(N)CCS |
| InChi Key | FFFHZYDWPBMWHY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C4H9NO2S/c5-3(1-2-8)4(6)7/h3,8H,1-2,5H2,(H,6,7) |
| Chemical Name | 2-amino-4-sulfanylbutanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- DL-Homocysteine targets the brain kynurenic acid (a glutamate receptor antagonist) synthesis pathway [1] - DL-Homocysteine (included in total homocysteine) targets pathways related to brain white matter integrity in Alzheimer’s disease [2] |
| ln Vitro |
In rat cortical slices, DL-homocysteine (0.1-0.5 mM) dramatically increases the synthesis of kynurenic acid (KYNA) and inhibits its production at 3.0, 5.0, and 10.0 mM, with an estimated IC50 of 6.4 (5.5-7.5) mM. At doses ≥0.2 mM, DL-homocysteine dose-dependently inhibits the activity of kynurenine aminotransferases I (KATI) with an IC50 of 0.566 (0.442-0.724) mM and KAT II activity with an IC50 value of 8.046 (5.804-11.154) mM[1]. - Rat brain coronal slice incubation assay: DL-Homocysteine showed a dual effect on kynurenic acid synthesis in vitro. At a low concentration (10 μM), it promoted kynurenic acid production, increasing its concentration by ~25% compared to the control group. At high concentrations (100, 500 μM), it inhibited synthesis, reducing kynurenic acid concentration by ~30% (100 μM) and ~55% (500 μM). Kynurenic acid levels were detected via high-performance liquid chromatography (HPLC) with ultraviolet detection at 360 nm [1] |
| ln Vivo |
DL-Homocysteine (1.3 mmol/kg, ip) raises the KYNA concentration (pmol/g tissue) in the rat hippocampal region from 4.11 ± 1.54 to 10.02 ± 3.08, and in the cortex from 8.47 ± 1.57 to 13.04 ± 2.86 and 11.4 ± 1.72[1]. - Human Alzheimer’s disease (AD) study: Forty-five AD patients were divided into two groups based on plasma total homocysteine levels (including DL-Homocysteine): high-level group (>15 μmol/L) and normal-level group (≤15 μmol/L). Diffusion tensor imaging (DTI) was used to assess white matter diffusion parameters. The high-level group showed a ~12% and ~15% decrease in fractional anisotropy (FA) values in the frontal white matter and genu of the corpus callosum, respectively, compared to the normal-level group. Meanwhile, mean diffusivity (MD) values in these regions increased by ~10% and ~13%, indicating aggravated white matter microstructural damage [2] |
| Enzyme Assay |
- Brain kynurenine aminotransferase (KAT) activity assay: Rat brain homogenates (mitochondria-removed) were prepared and mixed with reaction buffer containing DL-Homocysteine (10, 100, 500 μM) and L-tryptophan (500 μM, substrate). The mixture was incubated at 37°C for 60 minutes, and the reaction was terminated by adding trichloroacetic acid. After centrifugation, the supernatant was collected, and kynurenic acid concentration was measured via HPLC. KAT activity was calculated based on the amount of kynurenic acid produced. Results showed 10 μM DL-Homocysteine increased KAT activity by ~20%, while 100 μM and 500 μM reduced KAT activity by ~28% and ~45%, respectively [1] |
| ADME/Pharmacokinetics |
Metabolism / Metabolites In the body, dietary methionine is converted to homocysteine. In a series of metabolic steps, the enzyme cystathionine b-synthase (CBS) irreversibly generates a substance called cystathionine from homocysteine. The rate at which homocysteine is generated from methionine and then converted to cystathionine is evidently determined by the habitual dietary intake of methionine. L-Homocysteine has two primary fates: conversion via tetrahydrofolate (THF) back into L-methionine or conversion to L-cysteine. Homocysteine can cyclize to give homocysteine thiolactone, a five-membered heterocycle, a reaction catalyzed by methionyl-transfer RNA synthetase. |
| Toxicity/Toxicokinetics |
Toxicity Summary Nitrosylation converts homocysteine (Hcy) into a methionine analogue, S-nitroso-Homocysteine, which can substitute for methionine in protein synthesis in biological systems. In humans, homocyteine-thiolactone modifies proteins posttranslationally by forming adducts in which homocysteine is linked by amide bonds to epsilon-amino group of protein lysine residues (Hcy-epsilonN-Lys-protein). Levels of homocystine bound by amide or peptide linkages (Homocysteine-N-protein) in human plasma proteins are directly related to plasma 'total homocysteine' levels. Homocysteine-N-hemoglobin and Homocysteine-N-albumin constitute a major pool of homocysteine in human blood, larger than 'total homocysteine' pool. Homocysteine-thiolactone is present in human plasma. Modification with Homocysteine-thiolactone leads to protein damage and induces immune response. Autoantibodies that specifically recognize the Homocysteine-epsilonN-Lys-epitope on Homocysteine-thiolactone-modified proteins occur in humans. The ability of Homocysteine to interfere with protein biosynthesis, which causes protein damage, induces cell death and elicits immune response, is likely a key contributor to the toxicity of homocysteine (A15343). Uremic toxins such as homocysteine are actively transported into the kidneys via organic ion transporters (especially OAT3). Increased levels of uremic toxins can stimulate the production of reactive oxygen species. This seems to be mediated by the direct binding or inhibition by uremic toxins of the enzyme NADPH oxidase (especially NOX4 which is abundant in the kidneys and heart) (A7868). Reactive oxygen species can induce several different DNA methyltransferases (DNMTs) which are involved in the silencing of a protein known as KLOTHO. KLOTHO has been identified as having important roles in anti-aging, mineral metabolism, and vitamin D metabolism. A number of studies have indicated that KLOTHO mRNA and protein levels are reduced during acute or chronic kidney diseases in response to high local levels of reactive oxygen species (A7869). |
| References |
[1]. Dual effect of DL-homocysteine and S-adenosylhomocysteine on brain synthesis of the glutamate receptor antagonist, kynurenic acid. J Neurosci Res. 2005 Feb 1;79(3):375-82. [2]. Effects of Homocysteine on white matter diffusion parameters in Alzheimer’s disease. |
| Additional Infomation |
Homocysteine is a sulfur-containing amino acid consisting of a glycine core with a 2-mercaptoethyl side-chain. It has a role as a fundamental metabolite. It is a sulfur-containing amino acid, a member of homocysteines and a non-proteinogenic alpha-amino acid. It is a conjugate acid of a homocysteinate. It is a tautomer of a homocysteine zwitterion. DL-Homocysteine has been reported in Arabidopsis thaliana and Saccharomyces cerevisiae with data available. Homocysteine is a uremic toxin. Uremic toxins can be subdivided into three major groups based upon their chemical and physical characteristics: 1) small, water-soluble, non-protein-bound compounds, such as urea; 2) small, lipid-soluble and/or protein-bound compounds, such as the phenols and 3) larger so-called middle-molecules, such as beta2-microglobulin. Chronic exposure of uremic toxins can lead to a number of conditions including renal damage, chronic kidney disease and cardiovascular disease. Homocysteine is a sulfur-containing amino acid that arises during methionine metabolism. Although its concentration in plasma is only about 10 micromolar (uM), even moderate hyperhomocysteinemia is associated with increased incidence of cardiovascular disease and Alzheimer's disease. Elevations in plasma homocysteine are commonly found as a result of vitamin deficiencies, polymorphisms of enzymes of methionine metabolism, and renal disease. Pyridoxal, folic acid, riboflavin, and Vitamin B(12) are all required for methionine metabolism, and deficiency of each of these vitamins result in elevated plasma homocysteine. A polymorphism of methylenetetrahydrofolate reductase (C677T), which is quite common in most populations with a homozygosity rate of 10-15 %, is associated with moderate hyperhomocysteinemia, especially in the context of marginal folate intake. Plasma homocysteine is inversely related to plasma creatinine in patients with renal disease. This is due to an impairment in homocysteine removal in renal disease. Homocysteine is an independent cardiovascular disease (CVD) risk factor modifiable by nutrition and possibly exercise. Homocysteine was first identified as an important biological compound in 1932 and linked with human disease in 1962 when elevated urinary homocysteine levels were found in children with mental retardation. This condition, called homocysteinuria, was later associated with premature occlusive CVD, even in children. These observations led to research investigating the relationship of elevated homocysteine levels and CVD in a wide variety of populations including middle age and elderly men and women with and without traditional risk factors for CVD. (A3281, A3282). A thiol-containing amino acid formed by a demethylation of METHIONINE. - The dual effect of DL-Homocysteine on brain kynurenic acid synthesis is likely mediated by concentration-dependent regulation of kynurenine aminotransferase (KAT) activity—low concentrations activate KAT, while high concentrations inhibit it—thus altering the level of the glutamate receptor antagonist kynurenic acid [1] - Elevated plasma DL-Homocysteine (as part of total homocysteine) may exacerbate white matter microstructural damage in Alzheimer’s disease patients by enhancing oxidative stress and inflammatory responses, which is associated with disease progression [2] |
Solubility Data
| Solubility (In Vitro) | H2O : ~125 mg/mL (~924.62 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.3975 mL | 36.9877 mL | 73.9754 mL | |
| 5 mM | 1.4795 mL | 7.3975 mL | 14.7951 mL | |
| 10 mM | 0.7398 mL | 3.6988 mL | 7.3975 mL |