Physicochemical Properties
| Molecular Formula | C21H38O2 |
| Molecular Weight | 322.525227069855 |
| Exact Mass | 322.287 |
| CAS # | 56687-67-3 |
| Related CAS # | DCP-LA;28399-31-7 |
| PubChem CID | 534623 |
| Appearance | Colorless to light yellow liquid |
| LogP | 6.132 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 15 |
| Heavy Atom Count | 23 |
| Complexity | 339 |
| Defined Atom Stereocenter Count | 0 |
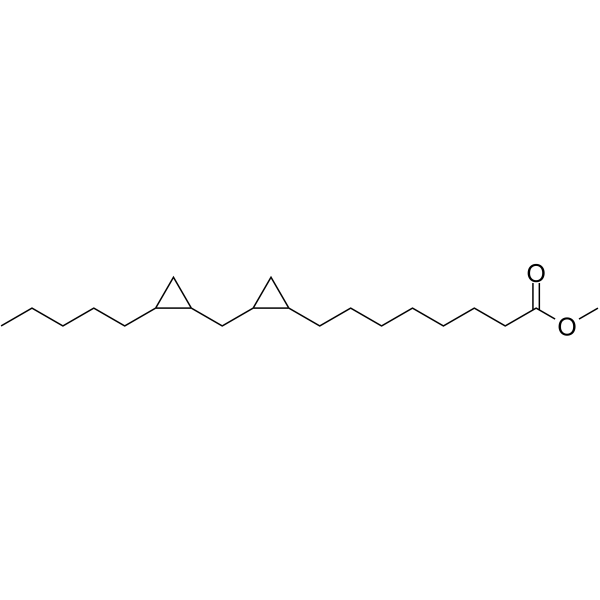
| SMILES | O(C)C(CCCCCCCC1CC1CC1CC1CCCCC)=O |
| InChi Key | WSVAMWHCPDAWFM-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H38O2/c1-3-4-8-11-17-14-19(17)16-20-15-18(20)12-9-6-5-7-10-13-21(22)23-2/h17-20H,3-16H2,1-2H3 |
| Chemical Name | methyl 8-[2-[(2-pentylcyclopropyl)methyl]cyclopropyl]octanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Peroxisome Proliferator-Activated Receptor α (PPARα) (IC50 = 0.8 μM; activates PPARα to regulate anti-inflammatory and neuroprotective signaling pathways) [1] - Nuclear Factor-κB (NF-κB) (No IC50/Ki data available; inhibits NF-κB-mediated inflammatory response as a downstream effect of PPARα activation) [1] |
| ln Vitro |
- In Aβ₂₅₋₃₅-induced PC12 cells (Alzheimer's disease, AD, cell model), DCPLA-ME (0.1-10 μM) exerted neuroprotective effects in a concentration-dependent manner. At 1 μM, it increased cell viability from 52% (Aβ group) to 89% (measured by MTT assay), reduced reactive oxygen species (ROS) production by 62% (DCFH-DA staining), and decreased the levels of pro-inflammatory cytokines TNF-α (from 28 pg/mL to 11 pg/mL) and IL-1β (from 22 pg/mL to 9 pg/mL) (ELISA) [1] - In primary rat cortical neurons, DCPLA-ME (0.3-3 μM) protected against glutamate-induced excitotoxicity. At 3 μM, it reduced neuronal apoptosis by 58% (Annexin V-FITC/PI flow cytometry) and upregulated the expression of synaptic proteins PSD95 (by 2.3-fold) and synaptophysin (by 1.9-fold) (Western blot). It also activated PPARα, as shown by a 3.1-fold increase in PPARα downstream gene ACOX1 expression (PCR) [1] |
| ln Vivo |
- In 6-month-old APP/PS1 transgenic mice (AD model), DCPLA-ME was administered via oral gavage at doses of 10, 30, and 100 mg/kg/day for 8 weeks. At 30 mg/kg/day, it improved spatial memory in the Morris water maze test: escape latency decreased from 58 s to 22 s, and time spent in the target quadrant increased from 25% to 48%. Brain tissue analysis showed: 1) Hippocampal Aβ₄₂ deposition reduced by 45% (immunohistochemistry); 2) Microglial activation (Iba1-positive cells) decreased by 52%; 3) TNF-α and IL-1β levels in the cortex reduced by 38% and 41%, respectively (ELISA); 4) PPARα protein expression in the hippocampus increased by 2.1-fold (Western blot) [1] - In C57BL/6 mice with MPTP-induced Parkinson's disease (PD) model, DCPLA-ME (50 mg/kg/day, intraperitoneal injection for 14 days) protected dopaminergic neurons: Tyrosine Hydroxylase (TH)-positive neurons in the substantia nigra increased from 42% (MPTP group) to 79%, and striatal dopamine levels recovered from 35% to 72% of normal mice (HPLC) [1] |
| Enzyme Assay |
- For PPARα activation assay (luciferase reporter gene assay): HEK293T cells were seeded in 24-well plates and co-transfected with PPARα expression plasmid, PPARα-responsive element (PPRE)-luciferase reporter plasmid, and Renilla luciferase plasmid (internal control). After 24 h of transfection, cells were treated with DCPLA-ME (0.01-10 μM) or PPARα agonist (positive control) for 18 h. Cells were lysed, and luciferase activity was measured using a dual-luciferase assay system. The ratio of firefly luciferase to Renilla luciferase activity was calculated to evaluate PPARα activation; DCPLA-ME showed a concentration-dependent increase in activity, with an IC50 of 0.8 μM [1] - For NF-κB inhibition assay: RAW 264.7 cells transfected with NF-κB-luciferase reporter plasmid were treated with LPS (1 μg/mL) and DCPLA-ME (0.1-10 μM) for 6 h. Luciferase activity was detected as described above. At 1 μM, DCPLA-ME reduced LPS-induced NF-κB activity by 53% [1] |
| Cell Assay |
- For Aβ₂₅₋₃₅-induced PC12 cell experiment: PC12 cells were cultured in RPMI-1640 medium with 10% fetal bovine serum and seeded in 96-well/6-well plates. After 24 h of seeding, cells were pre-treated with DCPLA-ME (0.1-10 μM) for 2 h, then co-incubated with Aβ₂₅₋₃₅ (20 μM) for 48 h. Cell viability was measured by MTT assay (490 nm absorbance). For ROS detection, cells were loaded with DCFH-DA (10 μM) for 30 min, and fluorescence intensity was measured at 488 nm excitation/525 nm emission. For cytokine detection, cell supernatants were collected for TNF-α/IL-1β ELISA [1] - For primary rat cortical neuron experiment: Cortices from E18 rat embryos were dissected, and neurons were isolated by trypsin digestion and plated on poly-L-lysine-coated plates. After 7 days in culture (neuron maturation), neurons were pre-treated with DCPLA-ME (0.3-3 μM) for 1 h, then exposed to glutamate (100 μM) for 24 h. Neuronal apoptosis was detected by Annexin V-FITC/PI staining and flow cytometry. PSD95/synaptophysin expression was analyzed by Western blot (primary antibodies against PSD95/synaptophysin, secondary HRP-conjugated antibody, ECL detection) [1] |
| Animal Protocol |
- For APP/PS1 transgenic mouse AD model experiment: 6-month-old male APP/PS1 mice (n=10 per group) were randomly divided into vehicle and DCPLA-ME treatment groups (10, 30, 100 mg/kg/day). DCPLA-ME was dissolved in dimethyl sulfoxide (DMSO) and diluted with 0.9% saline (final DMSO concentration ≤2%), then administered via oral gavage (10 mL/kg) once daily for 8 weeks; vehicle group received the same volume of solvent. After treatment, Morris water maze test was performed (5 days of training, 1 day of probe trial) to evaluate memory. Mice were then euthanized, brains were harvested, and hippocampus/cortex were dissected for Aβ deposition (immunohistochemistry with anti-Aβ antibody), microglial activation (anti-Iba1 antibody), and protein/gene detection (Western blot/PCR) [1] - For MPTP-induced mouse PD model experiment: 8-week-old male C57BL/6 mice (n=8 per group) were intraperitoneally injected with MPTP (20 mg/kg) once daily for 5 days to establish PD model. From the first day of MPTP injection, DCPLA-ME (50 mg/kg/day) or vehicle was intraperitoneally injected once daily for 14 days. After treatment, mice were euthanized, substantia nigra and striatum were dissected. TH-positive neurons were detected by immunohistochemistry, and striatal dopamine levels were measured by HPLC with electrochemical detection [1] |
| ADME/Pharmacokinetics |
- Oral pharmacokinetics in Sprague-Dawley rats: DCPLA-ME (100 mg/kg, oral gavage) had an oral bioavailability of ~35%. Plasma concentration-time profile showed: Cmax = 2.8 μg/mL (reached at 1.5 h), t1/2 = 4.2 h, AUC₀₋₂₄h = 18.6 μg·h/mL. Tissue distribution: At 2 h post-administration, brain concentration was ~15% of plasma concentration, liver concentration was ~4.3-fold of plasma concentration. Excretion: Within 24 h, ~20% of the dose was excreted in urine (as metabolites) and ~65% in feces (parent drug + metabolites) [1] - Intravenous pharmacokinetics in rats: DCPLA-ME (30 mg/kg, intravenous injection) had a Vd = 1.2 L/kg, Cl = 0.2 L/h/kg, t1/2 = 3.8 h [1] |
| Toxicity/Toxicokinetics |
- Acute toxicity: In ICR mice, oral LD50 of DCPLA-ME was >2000 mg/kg; intraperitoneal LD50 was >1000 mg/kg. No mortality or obvious toxic symptoms (e.g., lethargy, ataxia) were observed at doses up to 2000 mg/kg (oral) [1] - Subacute toxicity: Sprague-Dawley rats were orally administered DCPLA-ME (10, 50, 100 mg/kg/day) for 4 weeks. No significant changes in body weight, food intake, or organ weights (liver, kidney, brain) were observed. Serum biochemistry showed no abnormalities in ALT, AST, BUN, or creatinine. Hematology parameters (RBC, WBC, platelets) were within normal ranges. Plasma protein binding rate of DCPLA-ME was ~82% (measured by ultrafiltration) [1] |
| References |
[1]. Halogenated esters of cyclopropanated unsaturated fatty acids for use in the treatment of neurodegenerative diseases. WO2015058191A1. |
| Additional Infomation |
- DCPLA-ME is a halogenated ester of cyclopropanated unsaturated fatty acids, specifically developed as a PPARα agonist for the potential treatment of neurodegenerative diseases (e.g., Alzheimer's disease, Parkinson's disease) [1] - Its neuroprotective mechanism involves dual actions: 1) Activating PPARα to upregulate anti-inflammatory and antioxidant genes (e.g., ACOX1, SOD1); 2) Inhibiting NF-κB-mediated neuroinflammation and reducing toxic protein aggregation (e.g., Aβ in AD, α-synuclein in PD) [1] - The patent WO2015058191A1 claims DCPLA-ME as a candidate drug for neurodegenerative diseases, with preclinical data supporting its safety and efficacy in AD and PD animal models [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~15.62 mg/mL (~48.43 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.56 mg/mL (4.84 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 15.6 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.56 mg/mL (4.84 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 15.6 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1005 mL | 15.5024 mL | 31.0049 mL | |
| 5 mM | 0.6201 mL | 3.1005 mL | 6.2010 mL | |
| 10 mM | 0.3100 mL | 1.5502 mL | 3.1005 mL |