DCPIB (an ethacrynic acid derivative) is a novel, potent and selective blocker of the volume-sensitive anion channel (VSAC) on electrical activity and insulin secretion in rat pancreatic beta-cells. DCPIB inhibited whole-cell VSAC currents in beta-cells with IC50 values of 2.2 and 1.7 microM for inhibition of outward and inward currents, respectively. DCPIB also inhibited the VSAC at the single channel level in cells activated by glucose. In intact cells, DCPIB caused a net increase in beta-cell input conductance and evoked an outward current that was sensitive to inhibition by tolbutamide, suggesting KATP channel activation. However, no KATP channel activation was evident under conventional whole-cell conditions, suggesting that the drug might activate the channel in intact cells via an indirect mechanism, possibly involving nutrient metabolism. DCPIB suppressed glucose-induced electrical activity in beta-cells, hyperpolarised the cell membrane potential at a substimulatory glucose concentration and prevented depolarisation when the glucose concentration was raised to stimulatory levels. The suppression of electrical activity by DCPIB was associated with a marked inhibition of glucose-stimulated insulin release from intact islets. It is concluded that DCPIB inhibits electrical and secretory activity in the beta-cell as a combined result of a reciprocal inhibition of VSAC and activation of KATP channel activities, thus producing a marked hyperpolarisation of the beta-cell membrane potential.
Physicochemical Properties
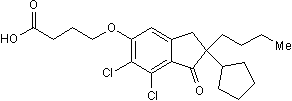
| Molecular Formula | 82749-70-0 | |
| Molecular Weight | 427.37 | |
| Exact Mass | 426.136 | |
| Elemental Analysis | C, 61.83; H, 6.60; Cl, 16.59; O, 14.97 | |
| CAS # | 82749-70-0 | |
| Related CAS # |
|
|
| PubChem CID | 10071166 | |
| Appearance | White to off-white solid powder | |
| Density | 1.263g/cm3 | |
| Boiling Point | 593.835ºC at 760 mmHg | |
| Flash Point | 312.94ºC | |
| Index of Refraction | 1.564 | |
| LogP | 6.342 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 28 | |
| Complexity | 563 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | KHKGTPJPBOQECW-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C22H28Cl2O4/c1-2-3-10-22(15-7-4-5-8-15)13-14-12-16(28-11-6-9-17(25)26)19(23)20(24)18(14)21(22)27/h12,15H,2-11,13H2,1H3,(H,25,26) | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
TRESK (IC50 = 0.14 μM); TASK1 (IC50 = 0.95 μM); TASK3 (IC50 = 50.72 μM) In COS-7 cells, DCPIB (10 μM) increases TRAAK currents and activates TREK1 [1]. With an IC50 of 0.14 μM, DCPIB (10 μM) substantially and reversibly suppresses TRESK currents in COS-7 cells [1]. In CPAE cells, 10 μM had no discernible inhibitory effect on ICl, swell circulation, or ICl, Ca [2]. The effects of DCPIB (10 μM, 5 min) on cardiomyocyte vascular layer edema are insignificant [2]. In BV2 cells, LPS-induced MAPK activation is inhibited by DCPIB (10 μM, 3 h) [3]. |
| ln Vitro |
In COS-7 cells, DCPIB (10 μM) increases TRAAK currents and activates TREK1 [1]. With an IC50 of 0.14 μM, DCPIB (10 μM) substantially and reversibly suppresses TRESK currents in COS-7 cells [1]. In CPAE cells, 10 μM had no discernible inhibitory effect on ICl, swell circulation, or ICl, Ca [2]. The effects of DCPIB (10 μM, 5 min) on cardiomyocyte vascular layer edema are insignificant [2]. In BV2 cells, LPS-induced MAPK activation is inhibited by DCPIB (10 μM, 3 h) [3]. DCPIB (10 μM) potently inhibits native ICL,swell in CPAE cells (83.7% inhibition), Xenopus oocytes (53.3% inhibition), and guinea-pig atrial cardiomyocytes (72.9% inhibition). It blocks ICL,swell in a voltage-independent and reversible manner. It does not affect cell swelling itself, as measured by cell width changes during hypotonic perfusion. It prevents swelling-induced action potential duration (APD) shortening in guinea-pig atrial myocytes without affecting APD under isosmotic conditions.[2] |
| ln Vivo |
Improved neuronal injury and inhibition of astrocyte activation by rMCAO deposition are two benefits of LDN-212854 (intracerebroventricular infusion, 1 mM, 10 μL) [3]. Accumulating evidence indicates that extensive microglia activation-mediated local inflammation contributes to neuronal injury in cerebral ischemia. We have previously shown that 4-(2-butyl-6, 7-dichloro-2-cyclopentyl-indan-1-on-5-yl) oxobutyric acid (DCPIB), a potent volume-regulated anion channel (VRAC) inhibitor, suppresses pathological glutamate release and excitatory neurotoxicity in reversible middle cerebral artery occlusion (rMCAO) model in vivo. In the present study, we sought to determine whether DCPIB also attenuates microglia activation that could contribute to neuronal injury in the cerebral ischemia/reperfusion pathology. We show that oxygen-glucose deprivation (OGD) induced microglia proliferation, migration, and secretion of cytokines and all these pathological changes were effectively inhibited by DCPIB in vitro. In the microglia/neuron co-cultures, OGD induced neuronal damage was reduced markedly in the presence of DCPIB. In rat rMCAO animal model, DCPIB significantly attenuated microglia activation and neuronal death. Activation of mitogen-activated protein kinase (MAPK) signaling pathway is known to be a critical signaling pathway for microglia activation. We further explored a potential involvement of DCPIB in this pathway by western blot analysis. Under the conditions that MAPK pathway was activated either by lipopolysaccharides (LPS) or OGD, the levels of phosphorylated ERK1/2, JNK and p38 were reduced significantly in the presence of DCPIB. Altogether, our study demonstrated that DCPIB inhibits microglia activation potently under ischemic conditions both in vitro and in vivo. The DCPIB effect is likely attributable to both direct inhibition VRAC and indirect inhibition of MAPK pathway in microglia that are beneficial for the survival of neurons in cerebral ischemic conditions[3]. |
| Enzyme Assay |
Mutagenesis[1] QuickChange II mutagenesis kit was used to introduce mutations into TREK1 and TRESK channels. TREK1 Δ1–56 (the first 56 amino acids were deleted) was made by PCR as previously. All clones were verified by further sequencing of the complete open reading frame region. Electrophysiological Recordings[1] Electrophysiology experiments were performed as described earlier. Briefly, wild-type or mutant K2P channels were recorded 24–48 h after transfection by the whole cell patch clamp technique. Currents were measured with a MultiClamp 700B patch-clamp amplifier/Digidata 1550B digitizer and pClamp 10 software. The sampling rate was 20 kHz and digitally filtered at 2 kHz. Series resistance compensation was set to 60–80%. The electrodes were pulled from borosilicate glass capillaries (BF150-110-10) and had a resistance of 2–5 MΩ when filled with intracellular solution. The intracellular solution contained 140 mM KCl, 1 mM MgCl2, 10 mM HEPES, and 5 mM EGTA (pH adjusted to 7.4 with KOH). The extracellular solution was composed of 145 mM NaCl, 2.5 mM KCl, 1 mM CaCl2, 3 mM MgCl2, and 10 mM HEPES (pH adjusted to 7.4 with NaOH). DCPIB and fluoxetine were dissolved in dimethyl sulfoxide (DMSO) to make 100 mM or 20 mM stock solutions that were stored at −20 °C. During the experiments, constant perfusion of bath solution was delivered by a homemade perfusion device allowing rapid solution switches with a flow velocity of approximately 2–3 mL/min. |
| Cell Assay |
Immunofluorescence [3] Cell Types: BV2 Cell Tested Concentrations: 10 μM Incubation Duration: 3 h Experimental Results: Ki67-positive staining of microglia and pro was Dramatically diminished. -Secretion of inflammatory cytokines (TNF-α, IL-1β). Western Blot Analysis[3] Cell Types: BV2 Cells Tested Concentrations: 10 μM Incubation Duration: 3 hrs (hours) Experimental Results: The migration potential of BV2 cells exposed to brief OGD (hypoxia-glucose deprivation) was inhibited. ICL,swell recording in CPAE cells: Cells were bathed in isotonic Cs⁺ solution, then exposed to 25% hypotonic solution to activate ICL,swell. Currents were recorded using whole-cell patch clamp with Cs⁺-based pipette solution. DCPIB was applied after swelling to assess inhibition.[2] ICL,swell recording in Xenopus oocytes: Oocytes were defolliculated and stored in ND 96 solution. Hypotonic solution (ND 48 without mannitol) was used to activate ICL,swell. Currents were recorded via two-electrode voltage clamp. ICL,swell recording in guinea-pig atrial myocytes: Cells were perfused with hypotonic solution (160 mOsmol) to activate ICL,swell. Pipette solution contained Cs⁺ to block K⁺ currents. DCPIB was applied to swollen cells to test blockade. Action potential recording in atrial myocytes: APs were elicited by current injection in current-clamp mode. Cells were exposed to hypotonic solution to induce swelling and APD shortening, followed by DCPIB application to reverse the effect.[2] |
| Animal Protocol |
Animal/Disease Models: Reversible middle cerebral artery occlusion (rMCAO) model [3]: 1 mM, 10 μL Route of Administration: Manual administration via intracerebroventricular infusion for 20 seconds Experimental Results: Pyramidal neuronal damage caused by rMCAO in the CA1 region was diminished. |
| References |
[1]. DCPIB, an Inhibitor of Volume-Regulated Anion Channels, Distinctly Modulates K2P Channels. ACS Chem Neurosci. 2019 Apr 17. [2]. DCPIB is a novel selective blocker of I(Cl,swell) and prevents swelling-induced shortening of guinea-pig atrial action potential duration. Br J Pharmacol. 2001 Dec;134(7):1467-79. [3]. DCPIB, a potent volume-regulated anion channel antagonist, attenuates microglia-mediated inflammatory response and neuronal injury following focal cerebral ischemia. Brain Res. 2014 Jan 13;1542:176-85. |
| Additional Infomation |
4-[(2-butyl-6,7-dichloro-2-cyclopentyl-1-oxo-3H-inden-5-yl)oxy]butanoic acid is a member of indanones. DCPIB is a non-diuretic ethacrynic acid derivative, structurally related to indanyloxyacetic acid (IAA-94). It is proposed as a valuable pharmacological tool to study the role of ICL,swell in cardiac excitability under pathophysiological conditions such as ischemia and reperfusion, where cell swelling occurs. It does not inhibit swelling-enhanced IKs currents, suggesting specificity for ICL,swell rather than general swelling-induced channel activation.[2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.87 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (4.87 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.87 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3399 mL | 11.6995 mL | 23.3989 mL | |
| 5 mM | 0.4680 mL | 2.3399 mL | 4.6798 mL | |
| 10 mM | 0.2340 mL | 1.1699 mL | 2.3399 mL |