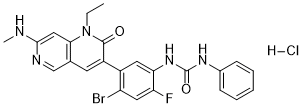
Ripretinib HCl (DCC2618; DCC-2618; Qinlock), the hydrochloride salt of DCC-2618, is an orally bioactive pan KIT and PDGFRA inhibitor approved in 2020 for the treatment of adults with advanced gastrointestinal stromal tumor. It functions by obstructing several additional kinase targets associated with systemic mastocytosis, including KIT D816V.
Physicochemical Properties
| Related CAS # | 1442472-39-0; 1225278-16-9 |
| Appearance | Solid |
| Synonyms | DCC-2618 hydrochloride; DCC 2618 HCl; DCC2618; Ripretinibhydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
KIT (wild-type IC50=14 nM; V560G mutant IC50=12 nM; D816V mutant IC50=16 nM; V560G/D816V double mutant IC50=15 nM); PDGFRα (wild-type IC50=21 nM; D842V mutant IC50=19 nM) [1] KIT and PDGFRα kinases ( focuses on activity against drug-resistant mutants) [2] |
||
| ln Vitro |
|
||
| ln Vivo |
|
||
| Enzyme Assay |
In order to assess KIT and BTK signaling, ROSAKIT WT, ROSAKIT D816V, HMC-1.1, and HMC-1.2 cells were incubated for 4 hours at 37°C in either control medium or DCC-2618 (0.5–5 μM). Western blotting was done essentially according to other instructions. In order to assess the downstream signaling pathways of KIT, HMC-1.1, HMC-1.2, ROSAKIT WT, and ROSAKIT D816V cells were initially pre-cultured for an entire night in Iscove-modified Dulbecco medium that was devoid of stem cell factor and fetal calf serum. DCC-2618 (0.001–10 μM) was then applied to 106 cells from each line for 90 minutes at 37°C. Following treatment, 10% stem cell factor-containing supernatants of Chinese hamster ovary cells transfected with the murine scf (kl) gene (CHO-KL) were added to ROSAKIT WT cells and incubated for 10 minutes at room temperature. After that, Western blotting was carried out essentially as previously mentioned. Recombinant KIT kinase activity assay: Purified recombinant KIT kinase domains (wild-type, V560G, D816V, V560G/D816V) were incubated with ATP (10 μM) and a biotinylated peptide substrate in the presence of serial dilutions of DCC-2618 HCl (0.1 nM-1 μM). Phosphorylated substrate was detected using a homogeneous time-resolved fluorescence (HTRF) assay, with streptavidin-conjugated europium and anti-phosphotyrosine antibody. IC50 values were calculated by nonlinear regression analysis of dose-response curves [1] - Recombinant PDGFRα kinase activity assay: The same HTRF-based method was used for PDGFRα kinase domains (wild-type, D842V). Reaction conditions included ATP (10 μM) and PDGFRα-specific biotinylated peptide substrate. DCC-2618 HCl was tested at concentrations ranging from 0.1 nM to 1 μM, and IC50 values were determined as described for KIT [1] - Kinase selectivity assay: DCC-2618 HCl (1 μM) was screened against a panel of 403 human kinases using a radiometric ATP incorporation assay. Inhibition rates were calculated relative to vehicle control, and selectivity was defined as the ratio of IC50 for off-target kinases to IC50 for KIT D816V [1] |
||
| Cell Assay |
The survival and proliferation of drug-exposed cells—primary or cell line—were examined. In the Online Supplementary Methods, the bioassays used are described. Cell viability assay: KIT/PDGFRα-mutant cell lines (HMC-1.1, HMC-1.2, PDGFRα D842V Ba/F3) and control cell lines (MV4-11, HL-60) were seeded in 96-well plates (5×10^3 cells/well) and treated with DCC-2618 HCl (0.1 nM-10 μM) for 72 hours. Cell viability was measured using a colorimetric MTT assay, and IC50 values were calculated from three independent experiments (mean ± SD) [1] - Western blot for signaling analysis: HMC-1.2 cells were seeded in 6-well plates (2×10^5 cells/well) and serum-starved for 16 hours. Cells were treated with DCC-2618 HCl (10-100 nM) for 2 hours, then harvested and lysed in RIPA buffer. Protein lysates (30 μg/lane) were separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-KIT (Y703, Y721), total KIT, p-AKT (S473), total AKT, p-ERK1/2 (T202/Y204), total ERK1/2, and GAPDH (loading control). Chemiluminescent detection was used to visualize bands, and densitometric analysis was performed [1] - Apoptosis assay: HMC-1.2 cells (1×10^6 cells/mL) were treated with DCC-2618 HCl (50-200 nM) for 48 hours. Cells were washed with PBS, stained with Annexin V-FITC and propidium iodide (PI) for 15 minutes at room temperature, and analyzed by flow cytometry. The percentage of apoptotic cells (Annexin V-positive, PI-negative or positive) was quantified [1] - Colony formation assay: HMC-1.2 cells were seeded in 6-well plates (200 cells/well) and treated with DCC-2618 HCl (10-50 nM). Medium was changed every 3 days, and after 14 days, colonies were fixed with methanol, stained with crystal violet, and counted. Colony formation efficiency was calculated as (number of colonies in treatment group / number of colonies in vehicle group) × 100% [1] - Primary patient cell assay: Bone marrow-derived mast cells from SM patients (n=3) were isolated and cultured in medium containing cytokines. Cells were treated with DCC-2618 HCl (10-100 nM) for 72 hours, and cell viability was measured using a trypan blue exclusion assay [1] |
||
| Animal Protocol |
|
||
| ADME/Pharmacokinetics |
Oral bioavailability: In rats, oral administration of DCC-2618 HCl (10 mg/kg) resulted in an oral bioavailability of 45%, with a Cmax of 1.2 μg/mL and AUC0-24h of 8.5 μg·h/mL [1] - Terminal half-life: Intravenous administration of DCC-2618 HCl (5 mg/kg) in rats showed a terminal half-life (t1/2) of 6.2 hours, with total body clearance of 1.8 mL/min/kg [1] - Plasma protein binding: DCC-2618 HCl exhibited high plasma protein binding (92-94%) in human, rat, and dog plasma, as determined by equilibrium dialysis [1] |
||
| Toxicity/Toxicokinetics |
In vitro toxicity: DCC-2618 HCl showed no significant cytotoxicity in normal human peripheral blood mononuclear cells (PBMCs) at concentrations up to 1 μM (cell viability >85% vs vehicle control) [1] - In vivo toxicity: Rats treated with DCC-2618 HCl (30 mg/kg/day, oral) for 28 days showed no significant weight loss, hematological abnormalities (white blood cell count, red blood cell count, platelets), or histopathological changes in major organs (liver, kidney, heart, lung, spleen) [1] - No overt toxicity in xenograft studies: Mice treated with DCC-2618 HCl (30 mg/kg/day, oral) for 21-32 days maintained normal body weight and showed no signs of acute toxicity (lethargy, diarrhea, hair loss) [1] |
||
| References |
[1]. Haematologica . 2018 May;103(5):799-809. [2]. Cancer Discov . 2017 Feb;7(2):121-122. |
||
| Additional Infomation |
DCC-2618 HCl is an orally bioavailable, potent, and selective small-molecule inhibitor of KIT and PDGFRα kinases, with specific activity against clinically relevant drug-resistant mutations (e.g., KIT D816V, PDGFRα D842V) [1][2] The compound targets oncogenic KIT/PDGFRα signaling, which drives the pathogenesis of systemic mastocytosis (SM), mast cell leukemia (MCL), acute myeloid leukemia (AML), and gastrointestinal stromal tumors (GIST) [1][2] DCC-2618 HCl blocks downstream signaling pathways (PI3K/AKT, RAS/ERK) critical for cell proliferation and survival, leading to growth inhibition and apoptosis in mutant cells [1] Preclinical data support the clinical development of DCC-2618 HCl for the treatment of SM-related hematologic malignancies, including MCL and AML, which are often refractory to conventional therapies [2] DCC-2618 HCl exhibits favorable pharmacokinetic properties (good oral bioavailability, moderate half-life, high plasma protein binding) and a favorable toxicity profile, supporting its use as an oral therapeutic agent [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |