Physicochemical Properties
| Molecular Formula | C17H12BRF2N3OS |
| Molecular Weight | 424.26 |
| Exact Mass | 422.985 |
| Elemental Analysis | C, 48.13; H, 2.85; Br, 18.83; F, 8.96; N, 9.90; O, 3.77; S, 7.56 |
| CAS # | 497061-48-0 |
| PubChem CID | 1025330 |
| Appearance | White to off-white solid powder |
| LogP | 5.314 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 25 |
| Complexity | 530 |
| Defined Atom Stereocenter Count | 0 |
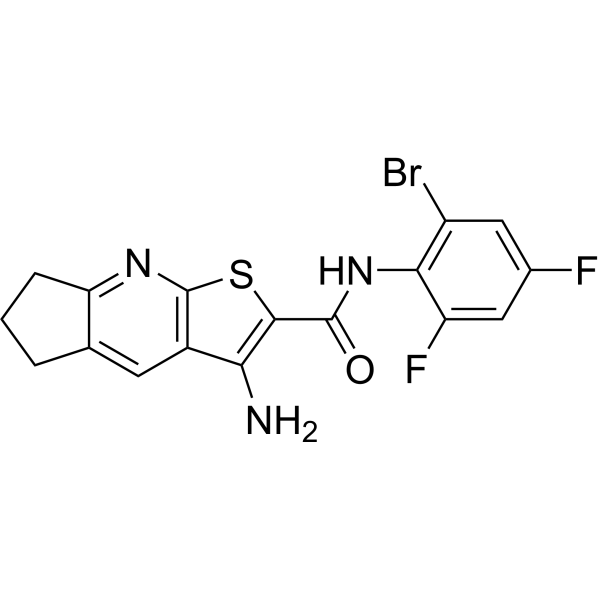
| SMILES | BrC1=C([H])C(=C([H])C(=C1N([H])C(C1=C(C2=C(N=C3C([H])([H])C([H])([H])C([H])([H])C3=C2[H])S1)N([H])[H])=O)F)F |
| InChi Key | DFNOJNBNTVQPCA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H12BrF2N3OS/c18-10-5-8(19)6-11(20)14(10)23-16(24)15-13(21)9-4-7-2-1-3-12(7)22-17(9)25-15/h4-6H,1-3,21H2,(H,23,24) |
| Chemical Name | 6-amino-N-(2-bromo-4,6-difluorophenyl)-4-thia-2-azatricyclo[7.3.0.03,7]dodeca-1,3(7),5,8-tetraene-5-carboxamide |
| Synonyms | DCAC50; DC AC50; DC_AC50 |
| HS Tariff Code | 2934.99.03.00 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In Canine Abrams, Canine D1, human HOS, and human MG63) cells, the IC50 values of DC_AC50 are 9.88 μM, 12.57 μM, 5.96 μM, and 6.68 μM, respectively[1]. Significant mitotic activity was seen in cells treated with DC_AC50 (0-10 μM). DC_AC50 (10 μM) decreased colony formation and increased OSA cells caused by carboplatin [1]. DC_AC50 at dosages of 3 μM and 10 μM. Canine and human OSA cells are inhibited in their migration by DC_AC50 (3 μM) [1]. DC_AC50 (2.5–10 μM) in a way that is depending on dosage. Adenocarcinoma cells (blank K562 cells, MDA-MB-231 breast cancer cells, H1299 human lung cancer cells, and 212LN head and neck cancer cells) are inhibited. Using human normal epithelial lung BEAS-2B or mammary gland cell MCF-10A as control cells, the DC_AC50 cell viability experiment was conducted [1]. |
| Cell Assay |
Cell Viability Assay[1] Cell Types: Canine OSA (Abrams, D1 and human OSA (HOS, MG63) cells. Tested Concentrations: 0-10 μM. Incubation Duration: 72 h. Experimental Results: Dose-dependently diminished viability of OSA cells. Apoptosis analysis[1] Cell Types: Abrams and HOS cells. Tested Concentrations: 1, 3 and 10 μM (10 μM Carboplatin). Incubation Duration: 24 h. Experimental Results: Potentiated carboplatin-induced apoptosis. |
| References |
[1]. Inhibition of copper chaperones sensitizes human and canine osteosarcoma cells to carboplatin chemotherapy. Vet Comp Oncol. 2020 Dec;18(4):559-569. [2]. Inhibition of human copper trafficking by a small molecule significantly attenuates cancer cell proliferation. Nat Chem. 2015 Dec;7(12):968-79. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~117.85 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.89 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.89 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3570 mL | 11.7852 mL | 23.5705 mL | |
| 5 mM | 0.4714 mL | 2.3570 mL | 4.7141 mL | |
| 10 mM | 0.2357 mL | 1.1785 mL | 2.3570 mL |